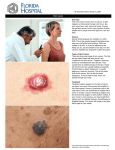
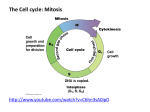
* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Carcinomas with DNA Mismatch Repair Deficiency
Epigenetics wikipedia , lookup
Molecular cloning wikipedia , lookup
Nucleic acid double helix wikipedia , lookup
Saethre–Chotzen syndrome wikipedia , lookup
DNA methylation wikipedia , lookup
Genome evolution wikipedia , lookup
DNA supercoil wikipedia , lookup
Cre-Lox recombination wikipedia , lookup
Epigenetics in stem-cell differentiation wikipedia , lookup
DNA vaccination wikipedia , lookup
Polycomb Group Proteins and Cancer wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Epigenetics of diabetes Type 2 wikipedia , lookup
Extrachromosomal DNA wikipedia , lookup
Epigenetics in learning and memory wikipedia , lookup
Zinc finger nuclease wikipedia , lookup
Epigenetics of neurodegenerative diseases wikipedia , lookup
Genealogical DNA test wikipedia , lookup
Non-coding DNA wikipedia , lookup
Site-specific recombinase technology wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Bisulfite sequencing wikipedia , lookup
Epigenomics wikipedia , lookup
History of genetic engineering wikipedia , lookup
No-SCAR (Scarless Cas9 Assisted Recombineering) Genome Editing wikipedia , lookup
DNA damage theory of aging wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Therapeutic gene modulation wikipedia , lookup
Helitron (biology) wikipedia , lookup
Cell-free fetal DNA wikipedia , lookup
Designer baby wikipedia , lookup
Genome editing wikipedia , lookup
Nutriepigenomics wikipedia , lookup
Microevolution wikipedia , lookup
Microsatellite wikipedia , lookup
Frameshift mutation wikipedia , lookup
Cancer epigenetics wikipedia , lookup
Carcinomas with DNA Mismatch Repair Deficiency Mohammad Hossein Sanei Department of pathology Isfahan university of medical science CRC: HNPCC Autosomal dominant. Mutation in the mismatch repair genes. High pentrance (around 75%). Predilection for right side. Early age of onset. Higher incidence of synchronous & metachronous tumors. 6% of CRC. Inactivation of DNA mismatch repair plays an important role in the pathogenesis of 10% to 15% of colorectal cancers. Although some of these are due to hereditary alterations in one or more of the mismatch repair genes (see Hereditary Non polyposis Colorectal Cancer, later), the majority (about 90%) are sporadic, due primarily to promoter methylation, and silencing, of transcription of MLH1 characteristic MSI-H genetic alteration, these cancers reveal a variety of specific clinical and pathologic associations of importance to surgical pathologists Sporadic mismatch repair–deficient MSI-H colorectal cancers show a marked female predominance (about 4 : 1), increase in prevalence with age, and are particularly common in women older than 70 years.[114] Most (about 90%) sporadic MSI-H colorectal cancers are located in the right colon, where they constitute about 50% of all right-sided cancers in women older than 70 years Precursor lesions often reveal characteristics of sessile serrated polyps (sessile serrated adenoma) in sporadic cases, and conventional adenomas in hereditary cases, such as HNPCC.[116] MSI-H cancers are also more likely to be multiple, have a polypoid or exophytic growth pattern, and show sharply circumscribed pushing margins and marked grossly visible necrosis. Furthermore, MSI-H tumors are more likely to show mucinous or signet ring cell features, and microglandular differentiation Of note, the association between these histologic features and DNA mismatch repair deficiency is high even when less than 50% of the tumor reveals these features. Therefore, the sensitivity and specificity of these histologic features for the diagnosis of MSI-H cancer vary according to the specific criteria used The presence of a component of any one of the associated histologic variants (mucinous, signet ring cell, microglandular, or undifferentiated) or of a prominent tumor-infiltrating lymphocytic response provides a relatively sensitive threshold to identify possible MSI-H cancers. Pathologic Features of High-Frequency Microsatellite Instability Colorectal Carcinomas Pathologic Feature Location in right colon Positive Predictive Value (%)[*] 32 MSI-H (%) 90 82 13 MSI-L/MSS (%) 34 54 5 15 5 35 22 7 36 13 38 14 25 28 13 0.4 3 8 21 Pushing margin 15 Crohn's-like reaction 49 “MSI-H” by pathologist 49 3 8 36 11 55 Exophytic/polypoid Signet ring cell component Mucinous type (>50% extracellular mucin) Mucinous component (>10% extracellular mucin) Cribriform pattern Poor differentiation Medullary type Medullary component (>10%) Lymphocytosis 21 31 34 71 59 25 (NS) 19 (NS) 44 The character of host lymphoid response is another feature that shows a striking difference between MSI-H and MSS cancers (Fig. 23-16). The presence of tumor infiltrating lymphocytes is highly predictive of MSI-H cancer, particularly when present in combination with an undifferentiated tumor (which may or may not be of classic medullary subtype In one study, the presence of at least 5 tumor-infiltrating lymphocytes per 10 highpower fields had a sensitivity of 93% and a specificity of 62% for the detection of MSI-H colorectal cancer.[122] A number of complex algorithms have been proposed in an attempt to achieve a high degree of specificity for the presence of MSIH in any single colon carcinoma. Overall, compared with MSI-H HNPCC– associated colon cancer, sporadic MSI-H cancers are also more frequently heterogeneous, poorly differentiated, mucinous, proximally located, and, as mentioned earlier, associated with a more prominent tumor-infiltrating lymphocyte reaction Clinically, patients with MSI-H colorectal cancers are more likely to present at an advanced T stage, but are less likely to show regional lymph node metastases. ESSENTIAL PATHOGENETIC ISSUES It is now known that about 50% of HNPCC cases (defined by Amsterdam I/II criteria) are caused by hereditary defects in one of four DNA mismatch repair genes[210–212] (Table 23-18). Mismatch repair genes function within a molecular complex; MSH2, and its binding partners MSH3 and MSH6, mediate recognition of the DNA mismatch. Subsequent recruitment of MLH1, and its binding partner PMS2, and other enzymes, leads to excision, repolymerization, and finally ligation of the repaired strand of DNA. HighFrequency Microsatellite Gene Instability Mutation Yes (50%) MLH1 (39%) MSH2 (38%) MSH6 (11%) PMS2 (7%) Unknown (5%) No (50%) Yes (as above; 10%) Unknown (90% Germline DNA mismatch repair gene mutations are heterozygous, and involve only one allele. Small frameshift mutations are the most common and result in premature protein truncation, followed by nonsense mutations and larger genomic deletions. Some patients with HNPCC harbor point mutations that result in amino acid substitutions at functionally critical residues Loss of DNA mismatch repair in tumor cells leads to a dramatic increase in the rate of mutations, which is characterized by frameshift mutations in repetitive DNA sequences known as microsatellites; this is referred to as microsatellite instability.[113], [218] Rare patients have also been described who harbor bi allelic inherited mutations in a single [ mismatch repair gene. These individuals have functionally defective DNA mismatch repair in all somatic tissues, which results in a severe, clinically distinctive phenotype (Lynch III) characterized by very early onset (teens and early twenties) and a preponderance of hematologic and brain tumors, in addition to the usual types of Lynch syndrome–associated cancers DNA mismatch repair is also defective in about 12% of all colorectal cancers in the absence of a germ line DNA mismatch repair gene mutation. In the majority of these cases, MLH1 expression is silenced in tumor cells by methylation of CpG islands in the promoter region of the gene. PATHOLOGIC FEATURES Compared with colorectal cancers with MLH1 methylation, HNPCC-Lynch cancers are more likely to occur in men, present at a younger age (generally less than 60 years), be associated with prior colorectal cancers or Lynch II cancers, and be located in the left colon or rectum (about one third overall). HNPCC-Lynch cancers are also typically associated with a residual precursor adenoma, compared with a high association with serrated polyps in cancers that develop in the MLH1 methylation pathway When histologic features are considered, one study also found that lymphocytic infiltration and tumor budding are more common in HNPCC-Lynch cancers, whereas mucin secretion, poor differentiation, tumor heterogeneity, and glandular serration were all more common in MLH1 methylated cancers In a more practical approach, one study attempted to determine optimal pathologic features that can be used to help identify likely HNPCC-Lynch cancers before 60 years of age. Using factors such as patient age, anatomic site, and four pathologic features ( histologic type, grade, Crohn's-like lymphoid reaction, and tumor-infiltrating lymphocytes), an “MsPath” (microsatellite instability by pathology) score is generated that can be used, on a sliding scale, to help predict the probability that a particular cancer is MSI-H ANCILLARY STUDIES USED BY PATHOLOGISTS Microsatellite Instability Testing Frameshift mutations in microsatellites can be identified by extraction of DNA from both normal and tumor tissue (usually paraffin-embedded tissue), amplification of selected microsatellites by PCR, and analysis of fragment size by gel electrophoresis or an automated sequencer (Fig. 23-28). Bethesda consensus–defined panel of microsatellites[222] or a revised panel that uses more mononucleotide markers.[223] The sensitivity of the revised panel of microsatellite instability testing is at least 90% Only occasional tumors from patients with known pathogenic mismatch repair gene mutations are MSS.[223] The specificity of the revised panel is also very high. The absence of mutations in up to 20% of HNPCC families with MSI-H tumors is believed to be due to a failure to detect unusual germ line variants TABLE 23-19 -- Bethesda Criteria for Microsatellite Instability Loci with Microsatellite Instability[*] Classification ≥40% High-frequency microsatellite instability 10%-30% Low-frequency microsatellite instability 0% Microsatellite stable Mismatch Repair mmunohistochemistry The DNA mismatch repair proteins are ubiquitously expressed in normal human tissues, particularly proliferating tissues, and nuclear expression in crypt epithelium and lymphocytes serves as an internal positive control for stain quality. In the setting of HNPCC, most hereditary and second-hit tumor alterations are inactivating chainterminating mutations. As a result of instability of the truncated mRNA transcript, or the protein product, these mutations are associated with complete loss of immunohistochemically detectable mismatch repair protein in tumors[224–226] (Fig. 23-29). Immunohistochemical testing is typically performed using antibodies to the four genes that harbor the most common known HNPCC mutations: MLH1, MSH2, MSH6, and PMS2. During normal DNA mismatch repair activation, MLH1 recruits its binding partner PMS2 to the site of DNA repair. If MLH1 is mutated (as in HNPCC) and lost from the DNA mismatch repair complex, then PMS2 will also be absent from the repair protein complex. Therefore, mutation and loss of MLH1 protein is also usually accompanied by loss of PMS2 expression. The sensitivity of DNA mismatch repair immunohistochemistry for identifying loss of protein expression in patients with HNPCC with known mutations is at least 90%. Immunohistochemical staining is often intact in tumors from patients with functionally pathogenic point mutations The specificity of loss of protein expression for an underlying mismatch repair defect is virtually 100%, although up to 10% of these tumors are, in fact, MSS on microsatellite instability testing.[225] TABLE 23-20 -- Interpretation of DNA Mismatch Repair Immunohistochemistry MLH1 + − PMS2 + − MSH2 + + MSH6 Interpretation + Intact DNA + MLH1 mismatch repair; or rare germline point mutation with intact immunohistochemis try; or other gene methylation silencing; or MLH1 germline mutation (HNPCC) + + + + − + − + + − + − MSH2 germline mutation (HNPCC) PMS2 germline mutation (HNPCC) MSH6 germline mutation (HNPCC) BRAF MUTATION TESTING Activating point mutations in the oncogenic signaling protein KRAS are present in about 40% of colorectal adenomas and carcinomas, and are considered a prototypical genetic alteration in these neoplasms. Recently, mutations in another member of the same signaling pathway, BRAF, were identified exclusively in colorectal cancers with wild-type KRAS genes.[228] Interestingly, BRAF mutations are nearly exclusively present in serrated pathway neoplasms, and have not yet been reported in HNPCC-Lynch syndrome tumors with a known mismatch repair defect.[230] Because of pathway specificity, some centers have begun to incorporate BRAF mutation testing into their diagnostic algorithms as a means to differentiate sporadic MLH1-deficient (methylated) cancers from hereditary MLH1-deficient cancers with an underlying germline mutation.[227] In MLH1-deficient cancers, BRAF mutation testing has a sensitivity and specificity of 50% to 70% and 100%, respectively, for somatic MLH1 methylation.[227] CpG Island Methylator Phenotype Testing A subset of colorectal cancers (about 25%) have widespread aberrations in DNA methylation, including promoter silencing of genes that are important to tumor biology.[231] Referred to as the CpG island methylator phenotype (CIMP), this includes most sporadic MSI-H cancers with methylation silencing of MLH1.[ CIMP testing is a method to detect abnormal DNA methylation by using a panel of markers/loci, and has been used in some studies to differentiate sporadic from hereditary MLH1-deficient cancers. Although there has not yet been an international consensus on the correct choice of markers for CIMP testing, several loci have begun to emerge as the most sensitive and specific for this type of application.[230] The goal of a genetic workup of HNPCC families is to identify the underlying germ line mutation. Confirmation of the germ line mutation allows for the most accurate treatment and follow-up recommendations for the patient, and allows predictive testing to be undertaken in interested family members. The initial approach by most laboratories is to analyze the complete coding sequence of the relevant gene or genes (depending on immunohistochemistry results), as well as a portion of the intronic regions important to exon splicing Some laboratories use a variety of rapid screening approaches to find mutations, whereas others undertake a complete sequence analysis. In certain specific geographic/ethnic settings, screening may also begin with testing of founder mutations common to some populations (e.g., the American founder mutation in MSH2).[ If mutations are not detected, testing for mRNA splicing abnormalities and large genomic deletions is indicated, but this may not always be available. One of the biggest challenges in germline testing relates to the interpretation of point mutations.