Chapter2. Elements of quantum mechanics
... 3. The angular momentum Pθ of the electron in an orbit is always an integral multiple of Planck’s constant divided by 2π ...
... 3. The angular momentum Pθ of the electron in an orbit is always an integral multiple of Planck’s constant divided by 2π ...
5.2 Quantum Theory and the Atom
... • Electrons move in fixed circular orbits around the nucleus • Each orbital is associated with a specific energy level • As the radius of the shell increases, the energy of the electron increases ...
... • Electrons move in fixed circular orbits around the nucleus • Each orbital is associated with a specific energy level • As the radius of the shell increases, the energy of the electron increases ...
HW 8
... frequency condition the energy of the emitted photon must be equal to the difference in energy between the higher and lower levels. An energy level diagram for the H-atom shows that as the energy levels get higher, the gaps between them converge; of the transitions listed, the two adjacent levels wh ...
... frequency condition the energy of the emitted photon must be equal to the difference in energy between the higher and lower levels. An energy level diagram for the H-atom shows that as the energy levels get higher, the gaps between them converge; of the transitions listed, the two adjacent levels wh ...
5 ELECTRONS IN ATOMS Vocabulary Review Name ___________________________
... 3. the SI unit of frequency ...
... 3. the SI unit of frequency ...
Problem 1. What is the wavelength of a hydrogen atom... sponding to the mean kinetic energy at thermal equilibrium at...
... Problem 1. What is the wavelength of a hydrogen atom moving with a velocity corresponding to the mean kinetic energy at thermal equilibrium at 20 degrees Centigrade? [2 points] Problem 2. If you rewrite the uncertainty relation for a free particle in terms of the uncertainty of its wavelength ∆λ rat ...
... Problem 1. What is the wavelength of a hydrogen atom moving with a velocity corresponding to the mean kinetic energy at thermal equilibrium at 20 degrees Centigrade? [2 points] Problem 2. If you rewrite the uncertainty relation for a free particle in terms of the uncertainty of its wavelength ∆λ rat ...
Quiz 1 Key
... Describe what the emission spectra of a hydrogen atom looks like compared to that of white light and report what this indicated about the energy of the electrons around the hydrogen atom. White light has all of the colors and therefore wavelengths present. The emission spectrum of hydrogen had only ...
... Describe what the emission spectra of a hydrogen atom looks like compared to that of white light and report what this indicated about the energy of the electrons around the hydrogen atom. White light has all of the colors and therefore wavelengths present. The emission spectrum of hydrogen had only ...
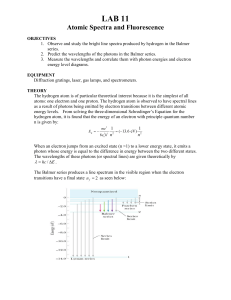
Lab 11: Atomic Spectra
... Fluorescence occurs when an atom absorbs light at one frequency but then emits light at lower frequencies. For example, an atom can absorb a UV photon and jump to a higher energy state. Rather then jumping directly back to the initial energy state (and emitting the same energy UV photon), the atom c ...
... Fluorescence occurs when an atom absorbs light at one frequency but then emits light at lower frequencies. For example, an atom can absorb a UV photon and jump to a higher energy state. Rather then jumping directly back to the initial energy state (and emitting the same energy UV photon), the atom c ...
FYS0460 / FYSZ460 Ohjelmatyö Elektronisuhkulitografia
... Working in laboratory and in cleanroom conditions ...
... Working in laboratory and in cleanroom conditions ...
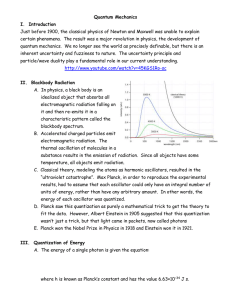
Quantum Mechanics I. Introduction Just before 1900, the classical
... idealized object that absorbs all electromagnetic radiation falling on it and then re-emits it in a characteristic pattern called the blackbody spectrum. B. Accelerated charged particles emit electromagnetic radiation. The thermal oscillation of molecules in a substance results in the emission of ra ...
... idealized object that absorbs all electromagnetic radiation falling on it and then re-emits it in a characteristic pattern called the blackbody spectrum. B. Accelerated charged particles emit electromagnetic radiation. The thermal oscillation of molecules in a substance results in the emission of ra ...
X-ray photoelectron spectroscopy - An introduction
... variations in the band bending and, thus, the work function will vary from point to point. This variation in surface potential produces a broadening of the XPS peaks. -Excitation process such as the shake-up/shake-off processes or vibrational broadening. ...
... variations in the band bending and, thus, the work function will vary from point to point. This variation in surface potential produces a broadening of the XPS peaks. -Excitation process such as the shake-up/shake-off processes or vibrational broadening. ...
Chapter 2 Learning Objectives
... Electromagnetic radiation The photoelectric effect Atomic orbitals and quantum numbers Atomic line spectra Multielectron atoms and electron configurations ...
... Electromagnetic radiation The photoelectric effect Atomic orbitals and quantum numbers Atomic line spectra Multielectron atoms and electron configurations ...
1 The Photoelectric Effect 2 Line Spectra and Energy Levels
... by emitting a photon with the same frequency and energy as the one originally absorbed. stimulated emission: an incident photon encounters an atom that’s already in an excited state with the same energy above the ground state as the photon’s energy; by a kind of resonance effect, the incoming photon ...
... by emitting a photon with the same frequency and energy as the one originally absorbed. stimulated emission: an incident photon encounters an atom that’s already in an excited state with the same energy above the ground state as the photon’s energy; by a kind of resonance effect, the incoming photon ...
Chapter 28 notes
... by emitting a photon with the same frequency and energy as the one originally absorbed. stimulated emission: an incident photon encounters an atom that’s already in an excited state with the same energy above the ground state as the photon’s energy; by a kind of resonance effect, the incoming photon ...
... by emitting a photon with the same frequency and energy as the one originally absorbed. stimulated emission: an incident photon encounters an atom that’s already in an excited state with the same energy above the ground state as the photon’s energy; by a kind of resonance effect, the incoming photon ...
Request for Mass Spectrometric Analysis
... Email: [email protected], [email protected] Incomplete forms will cause samples to be rejected. Max turnaround time: 2 weeks. Write legibly. If your sample has special requirements (keep frozen until analysis, sensitive to acid, etc) please describe in Extra Notes. Samp ...
... Email: [email protected], [email protected] Incomplete forms will cause samples to be rejected. Max turnaround time: 2 weeks. Write legibly. If your sample has special requirements (keep frozen until analysis, sensitive to acid, etc) please describe in Extra Notes. Samp ...
Ch.5 VocabReview
... 1. The lowest-energy arrangement of electrons in a subshell is obtained by putting electrons into separate orbitals of the subshell before pairing electrons. ...
... 1. The lowest-energy arrangement of electrons in a subshell is obtained by putting electrons into separate orbitals of the subshell before pairing electrons. ...
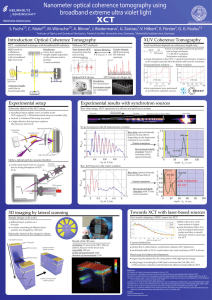
Nanometer optical coherence tomography using
... of the sample materials limit the bandwidth and the resolution ...
... of the sample materials limit the bandwidth and the resolution ...
SESSION 6: ELECTROMAGNETIC RADIATION KEY CONCEPTS: X
... Calculate the frequency of an EM wave with a wavelength of 400 nm. Question 2: What is the wavelength of a photon of light with a frequency of 101.3 kHz? Question 3: Give an example of the use of each type of EM radiation, i.e. gamma rays, X-rays, ultraviolet light, visible light, infrared, microwav ...
... Calculate the frequency of an EM wave with a wavelength of 400 nm. Question 2: What is the wavelength of a photon of light with a frequency of 101.3 kHz? Question 3: Give an example of the use of each type of EM radiation, i.e. gamma rays, X-rays, ultraviolet light, visible light, infrared, microwav ...
4-1. 1 - Riverside Local Schools
... 1. The lowest energy state of an atom is its… 2. A state in which an atom has a higher potential energy than it has in its ground state is an… 3. When an excited atom returns to its ground state, it gives off the energy it gained in the form of… 4. The amount of energy an excited atom gives off as i ...
... 1. The lowest energy state of an atom is its… 2. A state in which an atom has a higher potential energy than it has in its ground state is an… 3. When an excited atom returns to its ground state, it gives off the energy it gained in the form of… 4. The amount of energy an excited atom gives off as i ...
Appendix I.
... Many professionals consider microscopy as the initial method of choice for the characterisation, studying and identification of the various materials they come across throughout their line of work. ...
... Many professionals consider microscopy as the initial method of choice for the characterisation, studying and identification of the various materials they come across throughout their line of work. ...
EDS system
... If the sample is incorrectly focused, no WDS signal may be achieved at all and positioning the sample accurately becomes difficult and timeconsuming ...
... If the sample is incorrectly focused, no WDS signal may be achieved at all and positioning the sample accurately becomes difficult and timeconsuming ...
X-ray fluorescence

X-ray fluorescence (XRF) is the emission of characteristic ""secondary"" (or fluorescent) X-rays from a material that has been excited by bombarding with high-energy X-rays or gamma rays. The phenomenon is widely used for elemental analysis and chemical analysis, particularly in the investigation of metals, glass, ceramics and building materials, and for research in geochemistry, forensic science and archaeology.