Chapter 16. Our Atmosphere - Sites @ Suffolk University
... even at low concentrations, and causes respiratory distress when air pollution levels are high. It is not uncommon for Los Angeles school children not to be allowed to play outside because of high ozone levels. But Los Angeles is not the only area with dangerous levels of ozone. Government recommend ...
... even at low concentrations, and causes respiratory distress when air pollution levels are high. It is not uncommon for Los Angeles school children not to be allowed to play outside because of high ozone levels. But Los Angeles is not the only area with dangerous levels of ozone. Government recommend ...
Earth`s Amazing Atmosphere
... 7. The ozone layer is in the upper part of the atmospheric layer that contains most of the atmosphere’s ozone. Use the symbol for ozone to draw in the ozone layer on the diagram. 8. The ozone layer is important because it absorbs ultraviolet radiation. Draw a wavy line coming from space to represent ...
... 7. The ozone layer is in the upper part of the atmospheric layer that contains most of the atmosphere’s ozone. Use the symbol for ozone to draw in the ozone layer on the diagram. 8. The ozone layer is important because it absorbs ultraviolet radiation. Draw a wavy line coming from space to represent ...
notes
... Another form of energy is call enthalpy (H) which is the sum of the internal energy and pV from pV=nRT so H = U + PV or dH = dq –Vdp + Vdp +pdV = Vdp (dq is assumed to be zero for a process that does not have a heat loss) The change in the heat of a mass, per change in a degree centigrade, is ...
... Another form of energy is call enthalpy (H) which is the sum of the internal energy and pV from pV=nRT so H = U + PV or dH = dq –Vdp + Vdp +pdV = Vdp (dq is assumed to be zero for a process that does not have a heat loss) The change in the heat of a mass, per change in a degree centigrade, is ...
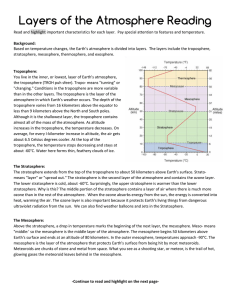
Layers of the Atmosphere Reading
... show a temperature well below zero. Why is that? Temperature is the average amount of energy of motion of each molecule of a substance. The gas molecules in the thermosphere move very rapidly, so the temperature is very high. However, the molecules are spaced far apart in the thin air. There are ...
... show a temperature well below zero. Why is that? Temperature is the average amount of energy of motion of each molecule of a substance. The gas molecules in the thermosphere move very rapidly, so the temperature is very high. However, the molecules are spaced far apart in the thin air. There are ...
Environmental Chemistry (Air)
... b) What is the approximate longitudinal difference in their locations? c) What is the average distance between two latitudes? ...
... b) What is the approximate longitudinal difference in their locations? c) What is the average distance between two latitudes? ...
1 Characteristics of the Atmosphere
... 1,000°C. Therm means “heat.” The temperatures in the thermosphere are so high because it contains a lot of oxygen and nitrogen, which absorb energy from the sun. ...
... 1,000°C. Therm means “heat.” The temperatures in the thermosphere are so high because it contains a lot of oxygen and nitrogen, which absorb energy from the sun. ...
FREE Sample Here - We can offer most test bank and
... The concentration of carbon dioxide (CO2) in the atmosphere is relatively low compared to some of the other constituents. CO2 is important none the less because a. it dissolves in water to form acid rain. b. it is a greenhouse gas. c. it is the main ingredient in photochemical smog. d. it is toxic. ...
... The concentration of carbon dioxide (CO2) in the atmosphere is relatively low compared to some of the other constituents. CO2 is important none the less because a. it dissolves in water to form acid rain. b. it is a greenhouse gas. c. it is the main ingredient in photochemical smog. d. it is toxic. ...
Water in the atmosphere
... The boundary of the polar air cell develops waves so that it dips farther towards the equator in some places and not as far in others. These are the dips and crests of a planetary wave called a Rossby wave, and they circulate around the polar region. They change in number and shape and sometimes pin ...
... The boundary of the polar air cell develops waves so that it dips farther towards the equator in some places and not as far in others. These are the dips and crests of a planetary wave called a Rossby wave, and they circulate around the polar region. They change in number and shape and sometimes pin ...
lecture_17_0 lecture_17_0.
... starting at sea level through the troposphere In the stratosphere starting above about 20 km after an initial isothermal layer, the temperature increases with height This is due to heating within the ozone layer caused by capture of significant ultraviolet radiation from the Sun by the dioxygen and ...
... starting at sea level through the troposphere In the stratosphere starting above about 20 km after an initial isothermal layer, the temperature increases with height This is due to heating within the ozone layer caused by capture of significant ultraviolet radiation from the Sun by the dioxygen and ...
FREE Sample Here - We can offer most test bank and
... The concentration of carbon dioxide (CO2) in the atmosphere is relatively low compared to some of the other constituents. CO2 is important none the less because a. it dissolves in water to form acid rain. b. it is a greenhouse gas. c. it is the main ingredient in photochemical smog. d. it is toxic. ...
... The concentration of carbon dioxide (CO2) in the atmosphere is relatively low compared to some of the other constituents. CO2 is important none the less because a. it dissolves in water to form acid rain. b. it is a greenhouse gas. c. it is the main ingredient in photochemical smog. d. it is toxic. ...
The Troposphere The Stratosphere The Mesosphere The
... The Thermosphere Near the top of the atmosphere, the air is very thin. At 80 kilometers above Earth’s surface, the air is only about 0.001 percent as dense as the air at sea level. It’s as though you took a cubic meter of air at sea level and expanded it into 100,000 cubic meters at the top of the m ...
... The Thermosphere Near the top of the atmosphere, the air is very thin. At 80 kilometers above Earth’s surface, the air is only about 0.001 percent as dense as the air at sea level. It’s as though you took a cubic meter of air at sea level and expanded it into 100,000 cubic meters at the top of the m ...
Thermosphere
... ionosphere. It begins 80km above the surface and extends to 400km. • Energy from the sun causes gas molecules in the ionosphere to become electronically charged ...
... ionosphere. It begins 80km above the surface and extends to 400km. • Energy from the sun causes gas molecules in the ionosphere to become electronically charged ...
Margaret Mead, American Anthropologist
... (includes visible light, infrared radiation and ultraviolet light); sun releases vast amounts of radiation, Earth receives about 2 billionths of that energy • The small amount of radiation we receive amounts to a lot of energy; about half of the solar energy ever reaches the Earth’s surface, the res ...
... (includes visible light, infrared radiation and ultraviolet light); sun releases vast amounts of radiation, Earth receives about 2 billionths of that energy • The small amount of radiation we receive amounts to a lot of energy; about half of the solar energy ever reaches the Earth’s surface, the res ...
File - Global Scholars
... reservoir of CFCs in the atmosphere may persist for 50 or 100 years. The largest measurable Antarctic ozone hole was observed in 2006, and since then it appears that ozone loss is stabilizing. However, some studies suggest that it may be 2050 before recovery is well under way. Addressing the depleti ...
... reservoir of CFCs in the atmosphere may persist for 50 or 100 years. The largest measurable Antarctic ozone hole was observed in 2006, and since then it appears that ozone loss is stabilizing. However, some studies suggest that it may be 2050 before recovery is well under way. Addressing the depleti ...
CHAPTER 2. ATMOSPHERIC PRESSURE
... behaves as a homogeneous gas of molecular weight Ma = 29 g mol-1. Dalton’s law stipulates that each component of the air mixture must behave as if it were alone in the atmosphere. One might then expect different components to have different scale heights determined by their molecular weight. In part ...
... behaves as a homogeneous gas of molecular weight Ma = 29 g mol-1. Dalton’s law stipulates that each component of the air mixture must behave as if it were alone in the atmosphere. One might then expect different components to have different scale heights determined by their molecular weight. In part ...
Margaret Mead, American Anthropologist
... (includes visible light, infrared radiation and ultraviolet light); sun releases vast amounts of radiation, Earth receives about 2 billionths of that energy • The small amount of radiation we receive amounts to a lot of energy; about half of the solar energy ever reaches the Earth’s surface, the res ...
... (includes visible light, infrared radiation and ultraviolet light); sun releases vast amounts of radiation, Earth receives about 2 billionths of that energy • The small amount of radiation we receive amounts to a lot of energy; about half of the solar energy ever reaches the Earth’s surface, the res ...
atmosphere - WordPress.com
... • The weather is made up of different elements, which are measured either by special instruments or are observed by a meteorologist. • These measurements are then recorded and used in the making of climate graphs and weather forecasts. Temperature is a physical quantity that is a measure of ...
... • The weather is made up of different elements, which are measured either by special instruments or are observed by a meteorologist. • These measurements are then recorded and used in the making of climate graphs and weather forecasts. Temperature is a physical quantity that is a measure of ...
Causes of Climate Change
... increase or decrease, causing either more or less radiation to be reflected to space rather than to Earth’s surface. Or the amount of infrared radiation from Earth’s atmosphere to space may increase or decrease. Any factor that causes a change to Earth’s energy balance is known as a radiative forcin ...
... increase or decrease, causing either more or less radiation to be reflected to space rather than to Earth’s surface. Or the amount of infrared radiation from Earth’s atmosphere to space may increase or decrease. Any factor that causes a change to Earth’s energy balance is known as a radiative forcin ...
STRUCTURE OF ATMOSPHERE
... amount of ozone is measured (total abundance, or percentage) The percentage of ozone peaks near the top of the stratosphere, and that, in combination with the greater amount of UV at height (less reaches lower altitudes), produces the greatest heating of the middle atmosphere at the stratopause. The ...
... amount of ozone is measured (total abundance, or percentage) The percentage of ozone peaks near the top of the stratosphere, and that, in combination with the greater amount of UV at height (less reaches lower altitudes), produces the greatest heating of the middle atmosphere at the stratopause. The ...
AOSC200_summer_lect1 - UMD | Atmospheric and Oceanic
... • THE CONTINUOUS EXCHANGE OF WATER AMONGST THE ‘RESERVOIRS’ IS KNOWN AS THE HYDROLOGIC CYCLE • THE HYDROLOGIC CYCLE IS POWERED BY ENERGY FROM THE SUN ...
... • THE CONTINUOUS EXCHANGE OF WATER AMONGST THE ‘RESERVOIRS’ IS KNOWN AS THE HYDROLOGIC CYCLE • THE HYDROLOGIC CYCLE IS POWERED BY ENERGY FROM THE SUN ...
Basic Properties of the Atmosphere
... Now the greenhouse effect, as mentioned above, only applies when sunlight is bypassing the atmosphere. This isn’t true in the stratosphere, however, where absorption of UV light by ozone heats it up considerably. In the stratosphere, the temperature gradient reverses due to increased heating by O3. ...
... Now the greenhouse effect, as mentioned above, only applies when sunlight is bypassing the atmosphere. This isn’t true in the stratosphere, however, where absorption of UV light by ozone heats it up considerably. In the stratosphere, the temperature gradient reverses due to increased heating by O3. ...
Atmosphere
... To understand how the energy radiated by earth's surface warms the atmosphere, think about what happens when you turn on a burner on the stove. The hot burner radiates energy much likes earth's surface does. Now, imagine that you place a pot of water on the burner. Through conduction, which is the t ...
... To understand how the energy radiated by earth's surface warms the atmosphere, think about what happens when you turn on a burner on the stove. The hot burner radiates energy much likes earth's surface does. Now, imagine that you place a pot of water on the burner. Through conduction, which is the t ...
ATMOSPHERIC STRUCTURE. The vertical distribution of
... kilometer, but the actual lapse rate in a given situation can be quite different. Sometimes the temperature is constant with height, or it may even increase with height in a shallow layer called an inversion. [See Inversion.] When a dry air parcel rises without mixing heat with the environment, the ...
... kilometer, but the actual lapse rate in a given situation can be quite different. Sometimes the temperature is constant with height, or it may even increase with height in a shallow layer called an inversion. [See Inversion.] When a dry air parcel rises without mixing heat with the environment, the ...
Name
... 9. Name at least 3 things that happen in the stratosphere. 2. How does the atmosphere make life on Earth possible? ...
... 9. Name at least 3 things that happen in the stratosphere. 2. How does the atmosphere make life on Earth possible? ...
Atmosphere of Mars

The atmosphere of Mars is the layer of gases surrounding Mars. It is, like that of Venus, composed mostly of carbon dioxide, but is far thinner. There has been renewed interest in its composition since the detection of traces of methane in 2003 that may indicate life but may also be produced by a geochemical process, volcanic or hydrothermal activity.The atmospheric pressure on the Martian surface averages 600 pascals (0.087 psi), about 0.6% of Earth's mean sea level pressure of 101.3 kilopascals (14.69 psi) and only 0.0065% of Venus's 9.2 megapascals (1,330 psi). It ranges from a low of 30 pascals (0.0044 psi) on Olympus Mons's peak to over 1,155 pascals (0.1675 psi) in the depths of Hellas Planitia. This pressure is well below the Armstrong limit for the unprotected human body. Mars's atmospheric mass of 25 teratonnes compares to Earth's 5148 teratonnes with a scale height of about 11 kilometres (6.8 mi) versus Earth's 7 kilometres (4.3 mi).The Martian atmosphere consists of approximately 96% carbon dioxide, 1.9% argon, 1.9% nitrogen, and traces of free oxygen, carbon monoxide, water and methane, among other gases, for a mean molar mass of 43.34 g/mol. The atmosphere is quite dusty, giving the Martian sky a light brown or orange-red color when seen from the surface; data from the Mars Exploration Rovers indicate that suspended dust particles within the atmosphere are roughly 1.5 micrometres across.On 16 December 2014, NASA reported detecting an unusual increase, then decrease, in the amounts of methane in the atmosphere of the planet Mars; as well as, detecting Martian organic chemicals in powder drilled from a rock by the Curiosity rover. Also, based on deuterium to hydrogen ratio studies, much of the water at Gale Crater on Mars was found to have been lost during ancient times, before the lakebed in the crater was formed; afterwards, large amounts of water continued to be lost.On 18 March 2015, NASA reported the detection of an aurora that is not fully understood and an unexplained dust cloud in the atmosphere of Mars.On 4 April 2015, NASA reported studies, based on measurements by the Sample Analysis at Mars (SAM) instrument on the Curiosity rover, of the Martian atmosphere using xenon and argon isotopes. Results provided support for a ""vigorous"" loss of atmosphere early in the history of Mars and were consistent with an atmospheric signature found in bits of atmosphere captured in some Martian meteorites found on Earth.