Biochemistry of Cells
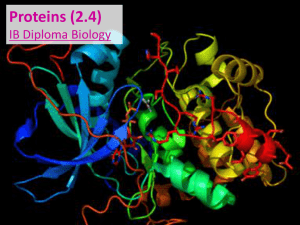
... Secondary protein structures occur when protein chains coil or fold When protein chains called polypeptides join together, the tertiary structure ...
... Secondary protein structures occur when protein chains coil or fold When protein chains called polypeptides join together, the tertiary structure ...
Food: Our Body`s Source of Energy and Structural Materials
... simple sugar such as glucose. Proteins are broken down into amino acids. Fats are broken down into an array of simpler molecules. If food technically can be anything that keeps you alive, what constitutes good nutrition? The phrase good nutrition means ensuring that your body receives what it requir ...
... simple sugar such as glucose. Proteins are broken down into amino acids. Fats are broken down into an array of simpler molecules. If food technically can be anything that keeps you alive, what constitutes good nutrition? The phrase good nutrition means ensuring that your body receives what it requir ...
Emergency Protocol Maple Syrup Urine Disease
... Call metabolic dietician on-call to help with dietary management. Begin enteral therapy if tolerated with a metabolic formula containing all required amino acids except for limited BCAA (Ketonex®-), 2.5-3.5 g/kg/day. Maintain phenylalanine, tyrosine, histidine, methionine, tryptophan, and threonine ...
... Call metabolic dietician on-call to help with dietary management. Begin enteral therapy if tolerated with a metabolic formula containing all required amino acids except for limited BCAA (Ketonex®-), 2.5-3.5 g/kg/day. Maintain phenylalanine, tyrosine, histidine, methionine, tryptophan, and threonine ...
Chapter 12 - Pathways to Biomolecules
... • Enzymes operate effectively within a small pH range. • Not all enzymes have the same optimum oH. • The ionisation of amino acids is depended on pH. • The bonds that determine the tertiary structure of the enzyme are altered as change in pH alter the ionisation of the amino acid residues in the pro ...
... • Enzymes operate effectively within a small pH range. • Not all enzymes have the same optimum oH. • The ionisation of amino acids is depended on pH. • The bonds that determine the tertiary structure of the enzyme are altered as change in pH alter the ionisation of the amino acid residues in the pro ...
Biochemistry: The Chemistry of Life
... The shape is maintained by various types of bonding between the R-groups. Covalent, ionic and hydrogen bonding are all seen Muscles and enzymes take on ...
... The shape is maintained by various types of bonding between the R-groups. Covalent, ionic and hydrogen bonding are all seen Muscles and enzymes take on ...
Nutritional requirements
... Lactose is called Milk sugar Sucrose is called cane sugar. Sports persons take glucose to get instant energy If we eat only rice, only carbohydrates are supplied to the body and body building materials (proteins) will be deficient. If we eat only proteins, body will be built up but for dai ...
... Lactose is called Milk sugar Sucrose is called cane sugar. Sports persons take glucose to get instant energy If we eat only rice, only carbohydrates are supplied to the body and body building materials (proteins) will be deficient. If we eat only proteins, body will be built up but for dai ...
Nutritional Requirements
... Lactose is called Milk sugar Sucrose is called cane sugar. Sports persons take glucose to get instant energy If we eat only rice, only carbohydrates are supplied to the body and body building materials (proteins) will be deficient. If we eat only proteins, body will be built up but for dai ...
... Lactose is called Milk sugar Sucrose is called cane sugar. Sports persons take glucose to get instant energy If we eat only rice, only carbohydrates are supplied to the body and body building materials (proteins) will be deficient. If we eat only proteins, body will be built up but for dai ...
amino acids - El Camino College
... 3. Your body is filled with many types of proteins. Each type has a distinctive sequence of amino acids which determines both its specialized ___________ and its unique ___________. • number, weight B) length, mass C) structure, function D) charge, pH 4. Nucleic acids: A) are the energy source for ...
... 3. Your body is filled with many types of proteins. Each type has a distinctive sequence of amino acids which determines both its specialized ___________ and its unique ___________. • number, weight B) length, mass C) structure, function D) charge, pH 4. Nucleic acids: A) are the energy source for ...
Mutations!
... ◦A sequence of DNA that codes for a specific protein (or proteins) associated with a trait, characteristic, or genetic condition ...
... ◦A sequence of DNA that codes for a specific protein (or proteins) associated with a trait, characteristic, or genetic condition ...
Background - WordPress.com
... should normally be used in conjunction with bicarbonate for which a relationship between the HEPES and bicarbonate exists for differing CO2 levels, although, HEPES alone can maintain pH in the absence of exogenous CO2. ...
... should normally be used in conjunction with bicarbonate for which a relationship between the HEPES and bicarbonate exists for differing CO2 levels, although, HEPES alone can maintain pH in the absence of exogenous CO2. ...
Biochemistry of life
... heart disease The male hormone testosterone and the female hormone estrogen are also steroids ...
... heart disease The male hormone testosterone and the female hormone estrogen are also steroids ...
Chapter 18
... a. Iron (Fe)—is part of the hemoglobin molecule, catalyzes formation of Vitamin A; is incorporated into a number of enzymes. b. Manganese (Mn)—occurs in enzymes needed for synthesis of fatty acids, and cholesterol, formation of urea, and normal functions of the nervous system. c. Copper (Cu)—essenti ...
... a. Iron (Fe)—is part of the hemoglobin molecule, catalyzes formation of Vitamin A; is incorporated into a number of enzymes. b. Manganese (Mn)—occurs in enzymes needed for synthesis of fatty acids, and cholesterol, formation of urea, and normal functions of the nervous system. c. Copper (Cu)—essenti ...
Food Chemistry
... 2. How many atoms of carbon are present in each molecule? ________________________________________________________________________ 3. Indicate the chemical formula for each sugar. ________________________________________________________________________ 4. What functional groups are found in carbohyd ...
... 2. How many atoms of carbon are present in each molecule? ________________________________________________________________________ 3. Indicate the chemical formula for each sugar. ________________________________________________________________________ 4. What functional groups are found in carbohyd ...
Acid-Base Balance
... Important in ECF Lots of carbon dioxide from metabolic acids It mixes with water & get carbonic acid which dissociates into H+ & HCO3Metabolic acids have H+ ; Our body has “bicarbonate reserve” Bicarbonate reserve = ample supply of bicarb in ECF These combine to form CO2 + H2O CO2 excreted via ...
... Important in ECF Lots of carbon dioxide from metabolic acids It mixes with water & get carbonic acid which dissociates into H+ & HCO3Metabolic acids have H+ ; Our body has “bicarbonate reserve” Bicarbonate reserve = ample supply of bicarb in ECF These combine to form CO2 + H2O CO2 excreted via ...
Lecture 3
... Amino acid monomers held together by covalent bonds (peptide bonds) Polypeptides: thousands or millions of amino acids Have two distinct ends: one terminating in an α amino group (the amino- or N- terminus) and the other is an α carboxylic group (carboxyl or ...
... Amino acid monomers held together by covalent bonds (peptide bonds) Polypeptides: thousands or millions of amino acids Have two distinct ends: one terminating in an α amino group (the amino- or N- terminus) and the other is an α carboxylic group (carboxyl or ...
Chemistry 1010 The Chemistry of Food: Proteins and Water
... How much protein do you need? 0.8 g protein per kg body weight Do most Americans get enough protein? yes – usually one to two times the required amount What kinds of people need extra protein? growing children pregnant and nursing women people with physical trauma (accident, surgery, disease) athle ...
... How much protein do you need? 0.8 g protein per kg body weight Do most Americans get enough protein? yes – usually one to two times the required amount What kinds of people need extra protein? growing children pregnant and nursing women people with physical trauma (accident, surgery, disease) athle ...
Carbon
... Specific heat refers to the amount of heat it takes to raise 1g, 1°C Water has a very high specific heat It takes a lot of energy to change the temperature of water, because of Hbonds ...
... Specific heat refers to the amount of heat it takes to raise 1g, 1°C Water has a very high specific heat It takes a lot of energy to change the temperature of water, because of Hbonds ...
Nutriflex® Specific Product Information may differ from country to
... Lipids may interfere with certain laboratory tests (such as bilirubin, lactate dehydrogenase, oxygen saturation) when the blood sample is taken before the lipids have been eliminated from the bloodstream. In most patients this takes 5 to 6 hours beyond the end of the infusion. The use of fat emulsio ...
... Lipids may interfere with certain laboratory tests (such as bilirubin, lactate dehydrogenase, oxygen saturation) when the blood sample is taken before the lipids have been eliminated from the bloodstream. In most patients this takes 5 to 6 hours beyond the end of the infusion. The use of fat emulsio ...
Chapter 12 Handout
... Many chemical reactions occur more rapidly when a ____________________ is present. The biological catalysts that accelerate the rate of chemical reactions in living things are a type of protein called enzymes. Enzymes control the manufacture of complex substances as well as breaking down the _______ ...
... Many chemical reactions occur more rapidly when a ____________________ is present. The biological catalysts that accelerate the rate of chemical reactions in living things are a type of protein called enzymes. Enzymes control the manufacture of complex substances as well as breaking down the _______ ...
What is Food Chemistry
... Carbohydrates make up a group of chemical compounds found in plant and animal cells. They have an empirical formula CnH2nOn or (CH2O)n. Since this formula is essentially a combination of carbon and water these materials are called “hydrates of carbon or carbohydrates”. Carbohydrates are the primary ...
... Carbohydrates make up a group of chemical compounds found in plant and animal cells. They have an empirical formula CnH2nOn or (CH2O)n. Since this formula is essentially a combination of carbon and water these materials are called “hydrates of carbon or carbohydrates”. Carbohydrates are the primary ...
Organic Macromolecules: Biological macromolecules
... The polymer polyethene for example, is made up of many ethene monomers that have been joined into a polymer chain. Polymers form through a process called polymerisation. Two examples of polymerisation reactions are addition and condensation reactions. An addition reaction occurs when unsaturated mon ...
... The polymer polyethene for example, is made up of many ethene monomers that have been joined into a polymer chain. Polymers form through a process called polymerisation. Two examples of polymerisation reactions are addition and condensation reactions. An addition reaction occurs when unsaturated mon ...
Why do we eat
... A vegetarian diet can be healthier than a diet that includes red meat and dairy products, as these contain saturated fats. However, a vegetarian diet is not recommeded for young children because it is so hard to get all the essential amino acids. Eating meat supplies all 20 amino acids. No plant pro ...
... A vegetarian diet can be healthier than a diet that includes red meat and dairy products, as these contain saturated fats. However, a vegetarian diet is not recommeded for young children because it is so hard to get all the essential amino acids. Eating meat supplies all 20 amino acids. No plant pro ...
2_4 Slides
... Polypeptides, or chains of amino acids, are the base (or primary) level of protein structure. But before they are functional, they must fold into specific structures based on the order / structure of their amino acid sequence. Remember, different amino acids have different chemical properties (i.e. ...
... Polypeptides, or chains of amino acids, are the base (or primary) level of protein structure. But before they are functional, they must fold into specific structures based on the order / structure of their amino acid sequence. Remember, different amino acids have different chemical properties (i.e. ...
Biochemistry Review
... uses those amino acids to build new proteins according to the directions of the DNA 57. What is the bond between amino acids called? Peptide bond 58. Recognize picture 59. Each individual is unique because of their _PROTEINS____ which are determined by their __DNA _____. 60. What is an enzyme? a pro ...
... uses those amino acids to build new proteins according to the directions of the DNA 57. What is the bond between amino acids called? Peptide bond 58. Recognize picture 59. Each individual is unique because of their _PROTEINS____ which are determined by their __DNA _____. 60. What is an enzyme? a pro ...
Biochemistry of Cells
... All proteins are made of 20 different amino acids linked in different orders Proteins are used to build cells, act as hormones & enzymes, and do much of the work in a cell ...
... All proteins are made of 20 different amino acids linked in different orders Proteins are used to build cells, act as hormones & enzymes, and do much of the work in a cell ...
Point accepted mutation

A point accepted mutation — also known as a PAM — is the replacement of a single amino acid in the primary structure of a protein with another single amino acid, which is accepted by the processes of natural selection. This definition does not include all point mutations in the DNA of an organism. In particular, silent mutations are not point accepted mutations, nor are mutations which are lethal or which are rejected by natural selection in other ways.A PAM matrix is a matrix where each column and row represents one of the twenty standard amino acids. In bioinformatics, PAM matrices are regularly used as substitution matrices to score sequence alignments for proteins. Each entry in a PAM matrix indicates the likelihood of the amino acid of that row being replaced with the amino acid of that column through a series of one or more point accepted mutations during a specified evolutionary interval, rather than these two amino acids being aligned due to chance. Different PAM matrices correspond to different lengths of time in the evolution of the protein sequence.