Chemical Vapor Deposition (CVD)
... coatings, powders, fibers and monolithic components. • This technique is often used in many thin film applications. • By varying the experimental conditions—substrate material, substrate temperature, composition of the reaction gas mixture, total pressure gas flows, etc.— materials with different pr ...
... coatings, powders, fibers and monolithic components. • This technique is often used in many thin film applications. • By varying the experimental conditions—substrate material, substrate temperature, composition of the reaction gas mixture, total pressure gas flows, etc.— materials with different pr ...
Practice exam - Dynamic Science
... 21) Pure ethanol can be produced from wine by a process best known as: a) b) c) d) ...
... 21) Pure ethanol can be produced from wine by a process best known as: a) b) c) d) ...
Chemical Synthesis Using Earth-Abundant Metal
... (i.e., Pd, Pt, Ru, Rh, Ir, Ag and Au). The problem with precious metals is that they are expensive, steadily rarefying, and are generally non-renewable. Catalysts made from these metals can also be harmful to humans and to the environment. ...
... (i.e., Pd, Pt, Ru, Rh, Ir, Ag and Au). The problem with precious metals is that they are expensive, steadily rarefying, and are generally non-renewable. Catalysts made from these metals can also be harmful to humans and to the environment. ...
A Gas-phase Electrochemical Reactor for Carbon Dioxide
... the main products obtained are C1 (or at most C2) hydrocarbons or oxygenates. The target is in fact the formation of liquid fuels with C≥2 because they are easier to transport and store, preserving the large investments made in the current energy and chemical infrastructures (Ampelli et al., 2011b). ...
... the main products obtained are C1 (or at most C2) hydrocarbons or oxygenates. The target is in fact the formation of liquid fuels with C≥2 because they are easier to transport and store, preserving the large investments made in the current energy and chemical infrastructures (Ampelli et al., 2011b). ...
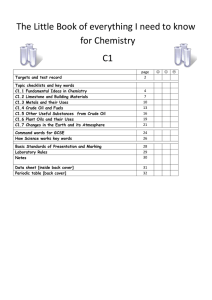
The Big book of C1 chemistry
... produce ash that contains the metal compounds bioleaching uses bacteria to produce leachate solutions that contain metal compounds. Copper can be obtained from solutions of copper salts by electrolysis or by displacement using scrap iron. During electrolysis positive ions move towards the negative ...
... produce ash that contains the metal compounds bioleaching uses bacteria to produce leachate solutions that contain metal compounds. Copper can be obtained from solutions of copper salts by electrolysis or by displacement using scrap iron. During electrolysis positive ions move towards the negative ...
Organometallic Compounds and Catalysis: Synthesis
... Organometallic Compounds and Catalysis: Synthesis and Use of Wilkinson’s Catalyst Organometallic chemistry is the chemistry of compounds which contain a metal carbon bond. Research interest in this area is largely fueled by potential applications of organometallic compounds as catalysts in industria ...
... Organometallic Compounds and Catalysis: Synthesis and Use of Wilkinson’s Catalyst Organometallic chemistry is the chemistry of compounds which contain a metal carbon bond. Research interest in this area is largely fueled by potential applications of organometallic compounds as catalysts in industria ...
Functional Groups
... They have high thermal stability and do not decompose until the temperature exceeds 700°C. The basic structure is two benzene rings connected by an oxygen-containing cyclic hydrocarbon. The benzene rings have from one to eight chlorine atoms substituting for hydrogen giving a total of 75 possible de ...
... They have high thermal stability and do not decompose until the temperature exceeds 700°C. The basic structure is two benzene rings connected by an oxygen-containing cyclic hydrocarbon. The benzene rings have from one to eight chlorine atoms substituting for hydrogen giving a total of 75 possible de ...
g - Santa Rosa Junior College
... • The nitrogen cycle involves a direct interaction between land and sea. • Atmospheric N2 must be fixed to enter the land and sea. – Atmospheric fixation occurs when lightning provides the energy for the reaction between N2(g) and O2(g). – Industrial fixation results from the production of NH3, whic ...
... • The nitrogen cycle involves a direct interaction between land and sea. • Atmospheric N2 must be fixed to enter the land and sea. – Atmospheric fixation occurs when lightning provides the energy for the reaction between N2(g) and O2(g). – Industrial fixation results from the production of NH3, whic ...
ch22 lecture 7e
... • The nitrogen cycle involves a direct interaction between land and sea. • Atmospheric N2 must be fixed to enter the land and sea. – Atmospheric fixation occurs when lightning provides the energy for the reaction between N2(g) and O2(g). – Industrial fixation results from the production of NH3, whic ...
... • The nitrogen cycle involves a direct interaction between land and sea. • Atmospheric N2 must be fixed to enter the land and sea. – Atmospheric fixation occurs when lightning provides the energy for the reaction between N2(g) and O2(g). – Industrial fixation results from the production of NH3, whic ...
Topic 16 Some non-metals and their compounds notes
... The carbon cycle describes the movement of carbon through the environment. Carbon exists in the ground, in the sea, in plants and animals, and in the air. The natural processes shown in the picture below can release carbon dioxide into the atmosphere (by combustion, decay and respiration) or remove ...
... The carbon cycle describes the movement of carbon through the environment. Carbon exists in the ground, in the sea, in plants and animals, and in the air. The natural processes shown in the picture below can release carbon dioxide into the atmosphere (by combustion, decay and respiration) or remove ...
ch22_lecture_6e_final
... • The nitrogen cycle involves a direct interaction between land and sea. • Atmospheric N2 must be fixed to enter the land and sea. – Atmospheric fixation occurs when lightning provides the energy for the reaction between N2(g) and O2(g). – Industrial fixation results from the production of NH3, whic ...
... • The nitrogen cycle involves a direct interaction between land and sea. • Atmospheric N2 must be fixed to enter the land and sea. – Atmospheric fixation occurs when lightning provides the energy for the reaction between N2(g) and O2(g). – Industrial fixation results from the production of NH3, whic ...
Organic Naming Notes
... Geometric Isomers: molecules that contain the same elements in the same amounts but their substituents can be drawn in different geometric arrangements. Trans Configuration: the substituent groups are on opposite sides of the double bond trans-2-Pentene Cis Configuration: the substituent groups are ...
... Geometric Isomers: molecules that contain the same elements in the same amounts but their substituents can be drawn in different geometric arrangements. Trans Configuration: the substituent groups are on opposite sides of the double bond trans-2-Pentene Cis Configuration: the substituent groups are ...
Chemical Engineering Principles of CVD Processes
... The walls of the reactor are cold - usually no deposition occurs on them - with a low wall temp : the risk of contamination from vapor/wall reactions is reduced - Homogeneous reaction is suppressed (CH4 can not be used to reach acceptable deposition rates) Advantage: Flexibility, high cleanliness, h ...
... The walls of the reactor are cold - usually no deposition occurs on them - with a low wall temp : the risk of contamination from vapor/wall reactions is reduced - Homogeneous reaction is suppressed (CH4 can not be used to reach acceptable deposition rates) Advantage: Flexibility, high cleanliness, h ...
transcript - American Chemical Society
... nerve agents. The exact manner in which the array of particles change colors depends on which chemicals are interacting with the nanoparticles. The set of colors associated with a particular chemical is like a fingerprint for that chemical. In its current form, this technology would be useful for de ...
... nerve agents. The exact manner in which the array of particles change colors depends on which chemicals are interacting with the nanoparticles. The set of colors associated with a particular chemical is like a fingerprint for that chemical. In its current form, this technology would be useful for de ...
formula - eduBuzz.org
... • Can you draw a diagram of the experiment you would use to prove the products of combustion of carbon or a hydrocarbon? • What is the word and formula equation for carbon burning in air? • What is carbon dioxide used for? ...
... • Can you draw a diagram of the experiment you would use to prove the products of combustion of carbon or a hydrocarbon? • What is the word and formula equation for carbon burning in air? • What is carbon dioxide used for? ...
Empirical is the
... formulas of the compound. Convert to grams first- milli means 1000 then Remember that a combustion reaction is one with a CH (hydrocarbon) reacting with Oxygen- Carbon dioxide and water are produced. To start this one find the amount of carbon in the carbon dioxide- percent composition! Take that va ...
... formulas of the compound. Convert to grams first- milli means 1000 then Remember that a combustion reaction is one with a CH (hydrocarbon) reacting with Oxygen- Carbon dioxide and water are produced. To start this one find the amount of carbon in the carbon dioxide- percent composition! Take that va ...
13.IVA group. Carbon and Silicon and their compounds.
... Reaction of oxide with bases/alkalis •It is a weakly acidic oxide dissolving sodium hydroxide solution to form sodium carbonate. •CO2(g) + 2NaOH(aq) ==> Na2CO3(aq) + H2O(l) •ionic equation: CO2(g) + 2OH-(aq) ==> CO32-(aq) + H2O(l) •SiO2(s) + 2NaOH(aq) ==> Na2SiO3(aq) + H2O(l) The dioxides react wit ...
... Reaction of oxide with bases/alkalis •It is a weakly acidic oxide dissolving sodium hydroxide solution to form sodium carbonate. •CO2(g) + 2NaOH(aq) ==> Na2CO3(aq) + H2O(l) •ionic equation: CO2(g) + 2OH-(aq) ==> CO32-(aq) + H2O(l) •SiO2(s) + 2NaOH(aq) ==> Na2SiO3(aq) + H2O(l) The dioxides react wit ...
Multiwalled Boron Nitride Nanotubes: Growth, Properties, and
... Another appealing property of BNNTs is piezoelectricity. This phenomenon theoretically originated from the deformation effect due to the rolling of the planar hexagonal BN networks to form tubular structures [13]. Nakhmanson et al. showed that BNNTs could be excellent piezoelectric systems [14]. As ...
... Another appealing property of BNNTs is piezoelectricity. This phenomenon theoretically originated from the deformation effect due to the rolling of the planar hexagonal BN networks to form tubular structures [13]. Nakhmanson et al. showed that BNNTs could be excellent piezoelectric systems [14]. As ...
Silicon Carbide Coating for Carbon Materials Produced by a
... carbon materials. At high temperature gaseous silicon monoxide generated from a Sic-Si02 powders mixture reacts with carbon substrate by converting the outer surfaces into silicon carbide. The correlation between density measurements and thermochemical calculations allows to determine the reaction p ...
... carbon materials. At high temperature gaseous silicon monoxide generated from a Sic-Si02 powders mixture reacts with carbon substrate by converting the outer surfaces into silicon carbide. The correlation between density measurements and thermochemical calculations allows to determine the reaction p ...
1 H NT Ch 12—Stoichiometry I. Review: Chemical Equations a
... ii. Carbon tetrachloride was prepared by reacting 100.0 g of carbon disulfide with 100.0 grams of chlorine gas. Calculate the theoretical and percent yield if 65.0 g of carbon tetrachloride was obtained. U ...
... ii. Carbon tetrachloride was prepared by reacting 100.0 g of carbon disulfide with 100.0 grams of chlorine gas. Calculate the theoretical and percent yield if 65.0 g of carbon tetrachloride was obtained. U ...