Chemical Reactions
... A decomposition reaction is a reaction has one reactant, and two or more products. CaCO3(s) CaO(s) + CO2(g) ...
... A decomposition reaction is a reaction has one reactant, and two or more products. CaCO3(s) CaO(s) + CO2(g) ...
This article was published in an Elsevier journal. The attached copy
... an easy phase separation. In the subsequent step, Section 3 including reactions (9) and (10), the separation of HI from L − 2, the heavier iodine/iodide–water phase, is the most critical scenario of the cycle [4] and believed to be the most expensive and energy-consuming step [5]. After establishing ...
... an easy phase separation. In the subsequent step, Section 3 including reactions (9) and (10), the separation of HI from L − 2, the heavier iodine/iodide–water phase, is the most critical scenario of the cycle [4] and believed to be the most expensive and energy-consuming step [5]. After establishing ...
Name: Date: ______ 1. Which of the following is a property of both
... A) Sb and SB B) CoBr2 and COBr2 C) TeO2 and TeO3 D) NH4Br and CaSO4 20. The total number of atoms present in one formula unit of Fe2(TeO4)3 is A) 14 B) 15 C) 16 D) 17 21. Which of the following is a property of both the liquid state and the solid state? A) a definite shape B) an indefinite volume C) ...
... A) Sb and SB B) CoBr2 and COBr2 C) TeO2 and TeO3 D) NH4Br and CaSO4 20. The total number of atoms present in one formula unit of Fe2(TeO4)3 is A) 14 B) 15 C) 16 D) 17 21. Which of the following is a property of both the liquid state and the solid state? A) a definite shape B) an indefinite volume C) ...
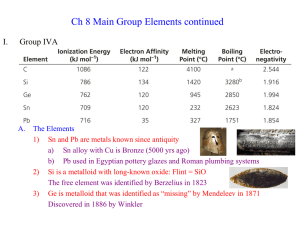
chapter 8 - Denton ISD
... Translated into a sentence, this equation reads, “When heated, solid mercury(II) oxide yields liquid mercury and gaseous oxygen.” It is also possible to write a chemical equation from a sentence describing a reaction. Consider the sentence, “Under pressure and in the presence of a platinum catalyst, ...
... Translated into a sentence, this equation reads, “When heated, solid mercury(II) oxide yields liquid mercury and gaseous oxygen.” It is also possible to write a chemical equation from a sentence describing a reaction. Consider the sentence, “Under pressure and in the presence of a platinum catalyst, ...
CHAPtER 4 Electrolysis
... The anode in an electrolytic cell is positive since the DC source withdraws electrons from it. Electrons are supplied to the positive anode by the oxidation of the copper electrode itself. In an electrolytic cell, the polarity of the electrodes is determined by the external DC source. In contrast, t ...
... The anode in an electrolytic cell is positive since the DC source withdraws electrons from it. Electrons are supplied to the positive anode by the oxidation of the copper electrode itself. In an electrolytic cell, the polarity of the electrodes is determined by the external DC source. In contrast, t ...
Chemistry - Set as Home Page
... In a dry cell, _________ act as the anode. The early Greeks believed that every thing in the universe was made up of three things namely air, earth _________ and water. ...
... In a dry cell, _________ act as the anode. The early Greeks believed that every thing in the universe was made up of three things namely air, earth _________ and water. ...
chapter 8
... Translated into a sentence, this equation reads, “When heated, solid mercury(II) oxide yields liquid mercury and gaseous oxygen.” It is also possible to write a chemical equation from a sentence describing a reaction. Consider the sentence, “Under pressure and in the presence of a platinum catalyst, ...
... Translated into a sentence, this equation reads, “When heated, solid mercury(II) oxide yields liquid mercury and gaseous oxygen.” It is also possible to write a chemical equation from a sentence describing a reaction. Consider the sentence, “Under pressure and in the presence of a platinum catalyst, ...
Summer Study Assignment – Honors Chem 2/AP Chemistry
... 31. Write the electron configuration using the Noble Gas core method for californium. 32. Write a balanced equation for the following double replacement reactions: a. Calcium hydroxide (aq) + nitric acid (aq) b. Chromium (III) sulfite (aq) + sulfuric acid (aq) c. Zinc chloride (aq) + ammonium su ...
... 31. Write the electron configuration using the Noble Gas core method for californium. 32. Write a balanced equation for the following double replacement reactions: a. Calcium hydroxide (aq) + nitric acid (aq) b. Chromium (III) sulfite (aq) + sulfuric acid (aq) c. Zinc chloride (aq) + ammonium su ...
View PDF
... ____ 17. In the reaction A + B → C + D, if the quantity of B is insufficient to react with all of A, a. A is the limiting reactant. c. there is no limiting reactant. b. B is the limiting reactant. d. no product can be formed. ____ 18. What is the maximum possible amount of product obtained in a che ...
... ____ 17. In the reaction A + B → C + D, if the quantity of B is insufficient to react with all of A, a. A is the limiting reactant. c. there is no limiting reactant. b. B is the limiting reactant. d. no product can be formed. ____ 18. What is the maximum possible amount of product obtained in a che ...
Fundamentals Diagnostic Quiz
... 19. All of the following statements are true regarding the nuclear model of the atom, except: a) The positive charge is densely found in the center of the atom, while the negatively charged electrons exist in a diffuse cloud outside the nucleus. b) Most of the space of an atom is empty space. *c) Th ...
... 19. All of the following statements are true regarding the nuclear model of the atom, except: a) The positive charge is densely found in the center of the atom, while the negatively charged electrons exist in a diffuse cloud outside the nucleus. b) Most of the space of an atom is empty space. *c) Th ...
Granta Design • CES Edupack 2009 • Durability - CORE
... Thus connecting dissimilar metals in either pure water or water with dissolved salts is a bad thing to do: corrosion cells appear that eat up the metal with the lower (more negative) corrosion potential. Worse news is to come: it is not necessary to have two metals: both anodic and cathodic reaction ...
... Thus connecting dissimilar metals in either pure water or water with dissolved salts is a bad thing to do: corrosion cells appear that eat up the metal with the lower (more negative) corrosion potential. Worse news is to come: it is not necessary to have two metals: both anodic and cathodic reaction ...
LaBrake, Fundamentals Diagnostic Questions
... These are questions to be used to help you fully prepare for 1A. While these topics will be covered in the 1ABC series, they will only be covered extremely briefly. It is expected that your chemistry background has prepared you to handle questions of this nature. Various sources can be used to help ...
... These are questions to be used to help you fully prepare for 1A. While these topics will be covered in the 1ABC series, they will only be covered extremely briefly. It is expected that your chemistry background has prepared you to handle questions of this nature. Various sources can be used to help ...
2(g)
... 12.4 g of Hydrogen gas reacts with 12.4 g of Oxygen gas to form liquid water. Determine the excess and limiting reagent. Calculate the maximum amount of water that could be produced by reacting these two gases together. ...
... 12.4 g of Hydrogen gas reacts with 12.4 g of Oxygen gas to form liquid water. Determine the excess and limiting reagent. Calculate the maximum amount of water that could be produced by reacting these two gases together. ...
ANSWERS Problem Set 5a – Chemical Reactions
... 8) The reaction below is balanced, but there are only ten molecules of reactants while there are twelve molecules of products. How can this be balanced? C6H12 + 9 O2 6 CO2 + 6 H2O Chemical reactions are not balanced by molecules or compounds, but only by atomic values. The number of atoms that ar ...
... 8) The reaction below is balanced, but there are only ten molecules of reactants while there are twelve molecules of products. How can this be balanced? C6H12 + 9 O2 6 CO2 + 6 H2O Chemical reactions are not balanced by molecules or compounds, but only by atomic values. The number of atoms that ar ...
Chemistry of METALS
... The refined ore has to be dissolved in cryolite first before electrolysis. Why is this necessary? 1½ mark To lower the melting point of aluminium oxide from about 2015oC to 900oC so as to lower /reduce cost of production Why are the carbon anodes replaced every now and then in the cell for electroly ...
... The refined ore has to be dissolved in cryolite first before electrolysis. Why is this necessary? 1½ mark To lower the melting point of aluminium oxide from about 2015oC to 900oC so as to lower /reduce cost of production Why are the carbon anodes replaced every now and then in the cell for electroly ...
Chapter 11 * Chemical Reactions
... General Equation: Na2SO4 (aq) + Ba(NO3)2 (aq 2NaNO3 (aq) + BaSO4 (s) Ionic Equation: 2Na+1 (aq) + SO4-2 (aq) + Ba+2 (aq) + 2NO3-1 (aq) 2Na+1 (aq) + 2 NO3-1 (aq) + BaSO4 (s) Net Ionic Equation: Ba+2 (aq) + SO4-2 (aq) BaSO4 (s) ...
... General Equation: Na2SO4 (aq) + Ba(NO3)2 (aq 2NaNO3 (aq) + BaSO4 (s) Ionic Equation: 2Na+1 (aq) + SO4-2 (aq) + Ba+2 (aq) + 2NO3-1 (aq) 2Na+1 (aq) + 2 NO3-1 (aq) + BaSO4 (s) Net Ionic Equation: Ba+2 (aq) + SO4-2 (aq) BaSO4 (s) ...
Elemental Sulfur Corrosion in Sour Gas and Claus Sulfur Recovery
... Sources: 1. Chesnoy, A.-B., and Pack, D.J., S8 Threatens Natural Gas Operations, Environment, OGJ, V.95, No.17, pp.74-79, Apr. 28, 1997. 2. Courtesy of PG&E, Technological and Ecological Services (TES), San Ramon, California ...
... Sources: 1. Chesnoy, A.-B., and Pack, D.J., S8 Threatens Natural Gas Operations, Environment, OGJ, V.95, No.17, pp.74-79, Apr. 28, 1997. 2. Courtesy of PG&E, Technological and Ecological Services (TES), San Ramon, California ...
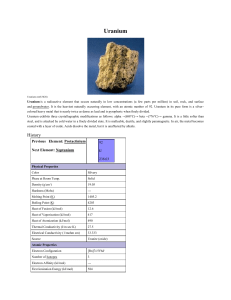
Uranium
... is the reason uranium still exists on the Earth. Three additional isotopes of uranium are not naturally present but can be produced by nuclear transformations. These are uranium-232, uranium-233, and uranium-236. Like the natural uranium isotopes, these three also decay by emitting an alpha particle ...
... is the reason uranium still exists on the Earth. Three additional isotopes of uranium are not naturally present but can be produced by nuclear transformations. These are uranium-232, uranium-233, and uranium-236. Like the natural uranium isotopes, these three also decay by emitting an alpha particle ...
The prebiotic molecules observed in the interstellar gas
... intensity of the molecular radio emission lines in space depends mainly on two factors, both of which can be well determined: the molecular electric dipole moment and the rotational partition function, which depends on the populations of the various levels. For stable molecules, the dipole moment ca ...
... intensity of the molecular radio emission lines in space depends mainly on two factors, both of which can be well determined: the molecular electric dipole moment and the rotational partition function, which depends on the populations of the various levels. For stable molecules, the dipole moment ca ...
Chemistry of CHLORINE
... Electronegativity is the ease/tendency of gaining/ acquiring electrons by an element during chemical reactions. It does not involve use of energy but theoretical arbitrary Pauling’s scale of measurements. (g) (i) 5cm3 of sodium chloride, Sodium bromide and Sodium iodide solutions were put separately ...
... Electronegativity is the ease/tendency of gaining/ acquiring electrons by an element during chemical reactions. It does not involve use of energy but theoretical arbitrary Pauling’s scale of measurements. (g) (i) 5cm3 of sodium chloride, Sodium bromide and Sodium iodide solutions were put separately ...
chem - CBSE Guess
... Rancidity: The oily and fatty food oxidizes and give bad smell and test is called rancidity.Preventatioin:By adding antioxidant which slow down the process of oxidation.2. Vaccum packing,3Flusing N2 gas in chips packets.3.Refrigeration. Q.Explain the various types of reactions with one example of ea ...
... Rancidity: The oily and fatty food oxidizes and give bad smell and test is called rancidity.Preventatioin:By adding antioxidant which slow down the process of oxidation.2. Vaccum packing,3Flusing N2 gas in chips packets.3.Refrigeration. Q.Explain the various types of reactions with one example of ea ...
Chemical Equations and Reactions
... Translated into a sentence, this equation reads, “When heated, solid mercury(II) oxide yields liquid mercury and gaseous oxygen.” It is also possible to write a chemical equation from a sentence describing a reaction. Consider the sentence, “Under pressure and in the presence of a platinum catalyst, ...
... Translated into a sentence, this equation reads, “When heated, solid mercury(II) oxide yields liquid mercury and gaseous oxygen.” It is also possible to write a chemical equation from a sentence describing a reaction. Consider the sentence, “Under pressure and in the presence of a platinum catalyst, ...
Fluorochemical industry
The global market for chemicals from fluorine was about US$16 billion per year as of 2006. The industry was predicted to reach 2.6 million metric tons per year by 2015. The largest market is the United States. Western Europe is the second largest. Asia Pacific is the fastest growing region of production. China in particular has experienced significant growth as a fluorochemical market and is becoming a producer of them as well. Fluorite mining (the main source of fluorine) was estimated in 2003 to be a $550 million industry, extracting 4.5 million tons per year.Mined fluorite is separated into two main grades, with about equal production of each. Acidspar is at least 97% CaF2; metspar is much lower purity, 60–85%. (A small amount of the intermediate, ceramic, grade is also made.) Metspar is used almost exclusively for iron smelting. Acidspar is primarily converted to hydrofluoric acid (by reaction with sulfuric acid). The resultant HF is mostly used to produce organofluorides and synthetic cryolite.