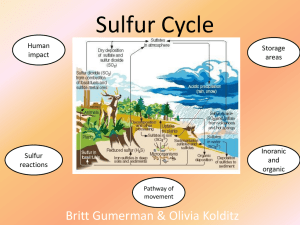
Sulfur Cycle - Walshearthsciences
... Sulfur reacts with fluorine, F2, and burns to form the hexafluoride sulfur(VI) fluoride. S8(s) + 24F2(g) → 8SF6(l) [orange] Reaction of sulfur with acids Sulfur does not react with dilute non-oxidizing acids. Reaction of sulfur with bases Sulfur reacts with hot aqueous potassium hydroxide, KOH, to f ...
... Sulfur reacts with fluorine, F2, and burns to form the hexafluoride sulfur(VI) fluoride. S8(s) + 24F2(g) → 8SF6(l) [orange] Reaction of sulfur with acids Sulfur does not react with dilute non-oxidizing acids. Reaction of sulfur with bases Sulfur reacts with hot aqueous potassium hydroxide, KOH, to f ...
Contents of toothpaste – safety implications
... Toothpaste) and contains 0.304% MFP (400 ppm fluoride). Data from several independent studies indicate that, although a dose-response relationship does exist for fluoride levels in toothpaste and caries, use of a 400 ppm fluoride-containing paste by children under 7 years of age instead of the stand ...
... Toothpaste) and contains 0.304% MFP (400 ppm fluoride). Data from several independent studies indicate that, although a dose-response relationship does exist for fluoride levels in toothpaste and caries, use of a 400 ppm fluoride-containing paste by children under 7 years of age instead of the stand ...
1 chemistry of the nonmetals
... Hydrogen is the most abundant element in the universe, accounting for 90% of the atoms and 75% of the mass of the universe. But hydrogen is much less abundant on earth. Even when the enormous number of hydrogen atoms in the oceans is included, hydrogen makes up less than 1% of the mass of the planet ...
... Hydrogen is the most abundant element in the universe, accounting for 90% of the atoms and 75% of the mass of the universe. But hydrogen is much less abundant on earth. Even when the enormous number of hydrogen atoms in the oceans is included, hydrogen makes up less than 1% of the mass of the planet ...
Title Metabolism of fluoroorganic compounds in microorganisms
... 2006). However, whether due to structural complexity or instability, synthetic modification is often not possible, and as such an alternative strategy is required. Precursor-directed biosynthesis is a useful strategy for producing natural product derivatives, in which the growth medium of a produci ...
... 2006). However, whether due to structural complexity or instability, synthetic modification is often not possible, and as such an alternative strategy is required. Precursor-directed biosynthesis is a useful strategy for producing natural product derivatives, in which the growth medium of a produci ...
A Low-Fluorine Solution with the F/Ba Mole Ratio of 2 for the
... may form prior to YF3 and a starting fluorine content in the precursor solution with the F/Ba mole ratio of 2 might be adequate for the full conversion of the Ba precursor to BaF2 . In the present work, a precursor solution with the F/Ba mole ratio of 2 was developed and thus named after ”F/Ba-2”. T ...
... may form prior to YF3 and a starting fluorine content in the precursor solution with the F/Ba mole ratio of 2 might be adequate for the full conversion of the Ba precursor to BaF2 . In the present work, a precursor solution with the F/Ba mole ratio of 2 was developed and thus named after ”F/Ba-2”. T ...
www.xtremepapers.net
... When a sports medal with a total surface area of 150 cm2 was evenly coated with silver, using electrolysis, its mass increased by 0.216 g. How many atoms of silver were deposited per cm2 on the surface of the medal? ...
... When a sports medal with a total surface area of 150 cm2 was evenly coated with silver, using electrolysis, its mass increased by 0.216 g. How many atoms of silver were deposited per cm2 on the surface of the medal? ...
Fluoride and Dental Caries
... Effect on dental plaque bacteria F inhibits bacterial adsorption In vitro: 9500 ppm F in solution inhibit bacterial adsorption to hydroxyapatite Clinical: rinses & toothpaste with Sn or amine F reduce plaque deposit ...
... Effect on dental plaque bacteria F inhibits bacterial adsorption In vitro: 9500 ppm F in solution inhibit bacterial adsorption to hydroxyapatite Clinical: rinses & toothpaste with Sn or amine F reduce plaque deposit ...
Unit - 7.pmd
... pπ -p π multiple bonds with itself and with other elements having small size and high electronegativity (e.g., C, O). Heavier elements of this group do not form pπ -pπ bonds as their atomic orbitals are so large and diffuse that they cannot have effective overlapping. Thus, nitrogen exists as a diat ...
... pπ -p π multiple bonds with itself and with other elements having small size and high electronegativity (e.g., C, O). Heavier elements of this group do not form pπ -pπ bonds as their atomic orbitals are so large and diffuse that they cannot have effective overlapping. Thus, nitrogen exists as a diat ...
The p-Block Elements The p-Block Elements
... pπ -p π multiple bonds with itself and with other elements having small size and high electronegativity (e.g., C, O). Heavier elements of this group do not form p π -pπ bonds as their atomic orbitals are so large and diffuse that they cannot have effective overlapping. Thus, nitrogen exists as a dia ...
... pπ -p π multiple bonds with itself and with other elements having small size and high electronegativity (e.g., C, O). Heavier elements of this group do not form p π -pπ bonds as their atomic orbitals are so large and diffuse that they cannot have effective overlapping. Thus, nitrogen exists as a dia ...
File
... halogens? All halogens have seven electrons in their outer shell. This means that: They can easily obtain a full outer shell by gaining one electron. They all gain an electron in reactions to form negative ions with a -1 charge. ...
... halogens? All halogens have seven electrons in their outer shell. This means that: They can easily obtain a full outer shell by gaining one electron. They all gain an electron in reactions to form negative ions with a -1 charge. ...
www.xtremepapers.net
... For each of the questions in this section, one or more of the three numbered statements 1 to 3 may be correct. Decide whether each of the statements is or is not correct (you may find it helpful to put a tick against the statements that you consider to be correct). The responses A to D should be sel ...
... For each of the questions in this section, one or more of the three numbered statements 1 to 3 may be correct. Decide whether each of the statements is or is not correct (you may find it helpful to put a tick against the statements that you consider to be correct). The responses A to D should be sel ...
Histopathology of myocardial damage in experimental fluorosis in
... who noted the replacement of delicate sarcolemma by fibrous structure. During our experiments, fragmentation and degeneration of cardiac muscle fibres were observed. However, the replacement of sarcolemma by fibrous tissue did not occur. Pribilla14 noticed fibrous necrosis, dissolution of nuclei, fi ...
... who noted the replacement of delicate sarcolemma by fibrous structure. During our experiments, fragmentation and degeneration of cardiac muscle fibres were observed. However, the replacement of sarcolemma by fibrous tissue did not occur. Pribilla14 noticed fibrous necrosis, dissolution of nuclei, fi ...
Atomic Structure
... fluorine atom when it reacts with copper. Use the diagram above to help you to answer this question. ...
... fluorine atom when it reacts with copper. Use the diagram above to help you to answer this question. ...
Bonding Web Practice Trupia - Trupia
... ____3. Which symbol represents a particle that has the same total number of electrons as S2–? (3) Se2– (1) O2– (2) Si (4) Ar ____4. Which element has atoms with the greatest attraction for electrons in a chemical bond? (1) beryllium (3) lithium (2) fluorine (4) oxygen ____5. Which atom will form the ...
... ____3. Which symbol represents a particle that has the same total number of electrons as S2–? (3) Se2– (1) O2– (2) Si (4) Ar ____4. Which element has atoms with the greatest attraction for electrons in a chemical bond? (1) beryllium (3) lithium (2) fluorine (4) oxygen ____5. Which atom will form the ...
Q1. This question is about the structure of atoms. (a) Choose words
... Complete the four spaces in the passage. The chemical formula of ammonia is NH3. This shows that there is one atom of .......................................... and three atoms of .................................. in each ......................................... of ammonia. These atoms are joined ...
... Complete the four spaces in the passage. The chemical formula of ammonia is NH3. This shows that there is one atom of .......................................... and three atoms of .................................. in each ......................................... of ammonia. These atoms are joined ...
Production of the Novel Lipopeptide Antibiotic Trifluorosurfactin via
... 2.1 Trifluorosurfactin production Following the method of Moran et al. [9] 4,4,4-trifluoro-DL-valine (10 mg) was added to growing cultures of Bacillus sp. CS93, with little or no inhibition of growth observed. Upon direct analysis of the culture supernatants using electrospray ionisation mass spectr ...
... 2.1 Trifluorosurfactin production Following the method of Moran et al. [9] 4,4,4-trifluoro-DL-valine (10 mg) was added to growing cultures of Bacillus sp. CS93, with little or no inhibition of growth observed. Upon direct analysis of the culture supernatants using electrospray ionisation mass spectr ...
17.2.3 Interhalogen compounds(65-67)
... T-shaped structure of CzV symmetry, consistent with the presence of 10 electrons in the valency shell of the central atom (Fig. 17.7a,b). A notable feature of both structures is the slight deviation from colinearity of the apical F-X-F bonds, the angle being 175.0" for ClF3 and 172.4' for BrF3; this ...
... T-shaped structure of CzV symmetry, consistent with the presence of 10 electrons in the valency shell of the central atom (Fig. 17.7a,b). A notable feature of both structures is the slight deviation from colinearity of the apical F-X-F bonds, the angle being 175.0" for ClF3 and 172.4' for BrF3; this ...
Fluorination Chemistry - Sigma
... The high toxicity/corrosivity of HF renders any fluorination technology that precludes its use or generation highly valuable to synthetic chemists. With this in mind, Aldrich Chemistry provides a versatile toolbox of fluorine reagents for both nucleophilic and electrophilic methods. ...
... The high toxicity/corrosivity of HF renders any fluorination technology that precludes its use or generation highly valuable to synthetic chemists. With this in mind, Aldrich Chemistry provides a versatile toolbox of fluorine reagents for both nucleophilic and electrophilic methods. ...
Day 13 Main Group Pt 1
... dilemma. Hydrogen has atomic properties which resemble both of these elements. The ionization energy for hydrogen is large and similar to that of fluorine, however, the resulting +1 oxidation state is common for hydrogen but not for fluorine. Molecular hydrogen is a diatomic gas like fluorine while ...
... dilemma. Hydrogen has atomic properties which resemble both of these elements. The ionization energy for hydrogen is large and similar to that of fluorine, however, the resulting +1 oxidation state is common for hydrogen but not for fluorine. Molecular hydrogen is a diatomic gas like fluorine while ...
www.XtremePapers.com
... When a sports medal with a total surface area of 150 cm2 was evenly coated with silver, using electrolysis, its mass increased by 0.216 g. How many atoms of silver were deposited per cm2 on the surface of the medal? ...
... When a sports medal with a total surface area of 150 cm2 was evenly coated with silver, using electrolysis, its mass increased by 0.216 g. How many atoms of silver were deposited per cm2 on the surface of the medal? ...
Chemical bonding
... • Carbon is always placed in the middle of the molecule (multiple carbons are bonded together in a chain) ...
... • Carbon is always placed in the middle of the molecule (multiple carbons are bonded together in a chain) ...
lesson 5
... understand how atoms link up to form compounds. Not all atoms form compounds. Only atoms that have outer shells that are not full form compounds. The elements of Group 18 have complete outer shells. These atoms usually do not form compounds. All other atoms have outer shells that are not full. All o ...
... understand how atoms link up to form compounds. Not all atoms form compounds. Only atoms that have outer shells that are not full form compounds. The elements of Group 18 have complete outer shells. These atoms usually do not form compounds. All other atoms have outer shells that are not full. All o ...
PPTB&W - Gmu - George Mason University
... Group 3A – Boron Family (ns2np1) Relative Basicity of Group 3 oxides ● Recall: A1 oxides (ionic charge +1 and more metallic) are more basic than A2 oxides (ionic charge +2 and less metallic) ● In general, oxides with the element in a lower oxidation state (less positive) are more basic than oxide ...
... Group 3A – Boron Family (ns2np1) Relative Basicity of Group 3 oxides ● Recall: A1 oxides (ionic charge +1 and more metallic) are more basic than A2 oxides (ionic charge +2 and less metallic) ● In general, oxides with the element in a lower oxidation state (less positive) are more basic than oxide ...
PPT - George Mason University
... Other group members are metals – shiny, relatively soft with low melting points Aluminum is more ionic; its low density and 3 valence electrons make it a good electrical conductor Although Aluminum is a metal, its halides exist in the gaseous state as covalent dimers - AL2Cl6 (contrast salts of grou ...
... Other group members are metals – shiny, relatively soft with low melting points Aluminum is more ionic; its low density and 3 valence electrons make it a good electrical conductor Although Aluminum is a metal, its halides exist in the gaseous state as covalent dimers - AL2Cl6 (contrast salts of grou ...
Topic 4 Chemistry of the Elements of the Main Group
... Hydrogen forms ionic hydrides with the reactive s-block metals (groups 1 and 2) and forms covalent hydrides with the p-group metals, e.g. Al and Sn (group 13 and 14). Electronegativity = 2.1. The value is intermediate in the electronegativity scale that spans from 0.7 to 4.0. H can form hydrides ( ...
... Hydrogen forms ionic hydrides with the reactive s-block metals (groups 1 and 2) and forms covalent hydrides with the p-group metals, e.g. Al and Sn (group 13 and 14). Electronegativity = 2.1. The value is intermediate in the electronegativity scale that spans from 0.7 to 4.0. H can form hydrides ( ...
Fluorine
Fluorine is a chemical element with symbol F and atomic number 9. It is the lightest halogen and exists as a highly toxic pale yellow diatomic gas at standard conditions. As the most electronegative element, it is extremely reactive: almost all other elements, including some noble gases, form compounds with fluorine.Among the elements, fluorine ranks 24th in universal abundance and 13th in terrestrial abundance. Fluorite, the primary mineral source of fluorine, was first described in 1529; as it was added to metal ores to lower their melting points for smelting, the Latin verb fluo meaning ""flow"" became associated with it. Proposed as an element in 1810, fluorine proved difficult and dangerous to separate from its compounds, and several early experimenters died or sustained injuries from their attempts. Only in 1886 did French chemist Henri Moissan isolate elemental fluorine using low-temperature electrolysis, a process still employed for modern production. Industrial synthesis of fluorine gas for uranium enrichment, its largest application, began during the Manhattan Project in World War II.Owing to the expense of refining pure fluorine, most commercial applications of the element involve the use of its compounds, with about half of mined fluorite used in steelmaking. The rest is converted into corrosive hydrogen fluoride en route to various organic fluorides, or into cryolite which plays a key role in aluminium refining. Organic fluorides have very high chemical and thermal stability; their major uses are as refrigerants, electrical insulation and cookware, the last as PTFE (Teflon). Pharmaceuticals such as atorvastatin and fluoxetine also contain fluorine, and the fluoride ion inhibits dental cavities, and so finds use in toothpaste and water fluoridation. Global fluorochemical sales amount to over US$15 billion a year.Fluorocarbon gases are generally greenhouse gases with global-warming potentials 100 to 20,000 times that of carbon dioxide. Organofluorine compounds persist in the environment due to the strength of the carbon–fluorine bond. Fluorine has no known metabolic role in mammals; a few plants synthesize organofluorine poisons which deter herbivores.