PowerPoint
... 1- How many protons, neutrons, and electrons are in an atom of chlorine-37? Chlorine has an atomic number of 17. Protons = 17 (atomic number) Electron = 17 (atomic number) Neutrons = 20 (neutrons = mass number (37)—atomic number (17) 2- How many protons, neutrons, and electrons are in an atom of Br ...
... 1- How many protons, neutrons, and electrons are in an atom of chlorine-37? Chlorine has an atomic number of 17. Protons = 17 (atomic number) Electron = 17 (atomic number) Neutrons = 20 (neutrons = mass number (37)—atomic number (17) 2- How many protons, neutrons, and electrons are in an atom of Br ...
Matching - hrsbstaff.ednet.ns.ca
... a. Electrons are negatively charged and are the heaviest subatomic particle. b. Protons are positively charged and the lightest subatomic particle. c. Neutrons have no charge and are the lightest subatomic particle. d. The mass of a neutron nearly equals the mass of a proton. ____ 16. All atoms are ...
... a. Electrons are negatively charged and are the heaviest subatomic particle. b. Protons are positively charged and the lightest subatomic particle. c. Neutrons have no charge and are the lightest subatomic particle. d. The mass of a neutron nearly equals the mass of a proton. ____ 16. All atoms are ...
Atoms, Isotopes, and Ions - Science Take-Out
... Teacher Answer Key: Applying What You Learned about Atoms, Isotopes, and Ions 1. Complete the element’s structure diagram on the right. 2. What is the element’s atomic number? ___15___ 3. What is the element’s mass number? ____31___ 4. What is the element’s chemical symbol? ___P____ 5. What is the ...
... Teacher Answer Key: Applying What You Learned about Atoms, Isotopes, and Ions 1. Complete the element’s structure diagram on the right. 2. What is the element’s atomic number? ___15___ 3. What is the element’s mass number? ____31___ 4. What is the element’s chemical symbol? ___P____ 5. What is the ...
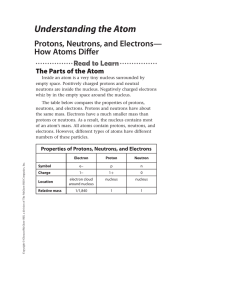
Understanding the Atom
... Alpha Decay An alpha particle is made of two protons and two neutrons. When an atom releases an alpha particle, its atomic number decreases by two. Uranium-238 decays to thorium-234 through the process of alpha decay. Beta Decay In beta decay, a neutron in an atom changes into a proton and a high-e ...
... Alpha Decay An alpha particle is made of two protons and two neutrons. When an atom releases an alpha particle, its atomic number decreases by two. Uranium-238 decays to thorium-234 through the process of alpha decay. Beta Decay In beta decay, a neutron in an atom changes into a proton and a high-e ...
Chapter 3-3—Parts of the Atom - Phoenix Union High School District
... How is an atom arranged? Atoms have a center or core called a nucleus The nucleus contains 2 subatomic particles: Proton that has a positive charge Neutron that has a NEUTRAL charge Protons and neutrons together are called nucleons because they are in the nucleus The nucleus is surrounded by a cl ...
... How is an atom arranged? Atoms have a center or core called a nucleus The nucleus contains 2 subatomic particles: Proton that has a positive charge Neutron that has a NEUTRAL charge Protons and neutrons together are called nucleons because they are in the nucleus The nucleus is surrounded by a cl ...
Agenda/To Do - Perry Local Schools
... The Halogens • Group VIIA • All have 7 valence electrons • All have similar chemical properties • All are highly reactive • Like to combine with group 1 ...
... The Halogens • Group VIIA • All have 7 valence electrons • All have similar chemical properties • All are highly reactive • Like to combine with group 1 ...
Chemistry Chapter 4 (Due October 24) [Test
... ____ 20. Which of the following is correct concerning subatomic particles? a. The electron was discovered by Goldstein in 1886. b. The neutron was discovered by Chadwick in 1932. c. The proton was discovered by Thomson in 1880. d. Cathode rays were found to be made of protons. ____ 21. All atoms are ...
... ____ 20. Which of the following is correct concerning subatomic particles? a. The electron was discovered by Goldstein in 1886. b. The neutron was discovered by Chadwick in 1932. c. The proton was discovered by Thomson in 1880. d. Cathode rays were found to be made of protons. ____ 21. All atoms are ...
atom`s - Hauppauge School District
... light (photons) • In the lowest levels, or the ground state, to the excited state ____________________ ________________________ • When electrons moved from the excited state back to the ground state, ______________ ...
... light (photons) • In the lowest levels, or the ground state, to the excited state ____________________ ________________________ • When electrons moved from the excited state back to the ground state, ______________ ...
Periodic Table Extra Practice ANSWER KEY 2014
... 1.4 I can describe the charge and location of protons, neutrons, and electrons within the nucleus and shells of an atom. The periodic table is, in many ways, the world’s greatest cheat sheet. The periodic table lists all of the elements (simple substances that make up more complex materials) like go ...
... 1.4 I can describe the charge and location of protons, neutrons, and electrons within the nucleus and shells of an atom. The periodic table is, in many ways, the world’s greatest cheat sheet. The periodic table lists all of the elements (simple substances that make up more complex materials) like go ...
CHEM A Midterm Review
... 1.4 I can describe the charge and location of protons, neutrons, and electrons within the nucleus and shells of an atom. The periodic table is, in many ways, the world’s greatest cheat sheet. The periodic table lists all of the elements (simple substances that make up more complex materials) like go ...
... 1.4 I can describe the charge and location of protons, neutrons, and electrons within the nucleus and shells of an atom. The periodic table is, in many ways, the world’s greatest cheat sheet. The periodic table lists all of the elements (simple substances that make up more complex materials) like go ...
Catalyst
... The mass number or atomic mass: This number tells the mass of one atom, which is approximately the sum of protons and neutrons in the nucleus, since each proton and each neutron has a mass equal to one mass unit, and the electrons ...
... The mass number or atomic mass: This number tells the mass of one atom, which is approximately the sum of protons and neutrons in the nucleus, since each proton and each neutron has a mass equal to one mass unit, and the electrons ...
Henry Moseley, the Atomic Number, and Synthesis
... to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alignment of their chemical properties. Moseley noticed that shooting electrons at elements caused them to release x ...
... to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alignment of their chemical properties. Moseley noticed that shooting electrons at elements caused them to release x ...
Henry Moseley, the Atomic Number, and Synthesis
... to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alignment of their chemical properties. Moseley noticed that shooting electrons at elements caused them to release x ...
... to advance the understanding of the elements and solve the problem with Mendeleev’s periodic table. Explain that organizing the elements by their weight did not always give a periodic alignment of their chemical properties. Moseley noticed that shooting electrons at elements caused them to release x ...
Nuclide, Atomic Number, mass number - Chemwiki
... A nuclide has a measurable amount of energy and lasts for a measurable amount of time. Stable nuclides can exist in the same state indefinitely, but unstable nuclides are radioactive and decay over time. Some unstable nuclides occur in nature, but others are synthesized artificially through nuclear ...
... A nuclide has a measurable amount of energy and lasts for a measurable amount of time. Stable nuclides can exist in the same state indefinitely, but unstable nuclides are radioactive and decay over time. Some unstable nuclides occur in nature, but others are synthesized artificially through nuclear ...
Chem-130 Test Lecture
... The Periodic Table Metals: elements to the left of the periodic table (excluding hydrogen). Generally lustrous, ductile, malleable and conduct heat and electricity. Nonmetals: elements to the right of the periodic table. Lack the properties of metals, are brittle as solids, do not conduct well, ...
... The Periodic Table Metals: elements to the left of the periodic table (excluding hydrogen). Generally lustrous, ductile, malleable and conduct heat and electricity. Nonmetals: elements to the right of the periodic table. Lack the properties of metals, are brittle as solids, do not conduct well, ...
Chapter 18 Comparing Atoms Lab
... 11. Which particles are found in an atom’s nucleus? Which particles are found outside the nucleus? 12. What four elements make up almost all of the mass in your body? 13. What is the atomic number of sodium? ...
... 11. Which particles are found in an atom’s nucleus? Which particles are found outside the nucleus? 12. What four elements make up almost all of the mass in your body? 13. What is the atomic number of sodium? ...
Chapter 2 Law of Conservation of Mass Law of Conservation of Mass
... • Some groups have descriptive names that are commonly used instead of their group numbers. – Halogens (Halides) • Group 17 (VIIA) nonmetals • exist naturally as diatomic molecules – Noble gases • Group 18 (VIIIA) nonmetals • are also called inert gases • are so named because they do not chemically ...
... • Some groups have descriptive names that are commonly used instead of their group numbers. – Halogens (Halides) • Group 17 (VIIA) nonmetals • exist naturally as diatomic molecules – Noble gases • Group 18 (VIIIA) nonmetals • are also called inert gases • are so named because they do not chemically ...
Chemistry 30 Review of Basic Chemistry 20
... Hydrated Compounds When writing hydrated compounds, follow all ionic rules described above. Then use a dot along with the number of water molecules ...
... Hydrated Compounds When writing hydrated compounds, follow all ionic rules described above. Then use a dot along with the number of water molecules ...
Unit 3-The Atom Chapter Packet
... Complete the following table of stable nuclei. Determine the neutron-to-proton ratio of each and then plot the points on the grid provided at the bottom of the page. ...
... Complete the following table of stable nuclei. Determine the neutron-to-proton ratio of each and then plot the points on the grid provided at the bottom of the page. ...
Chem Ch. 4.3
... • Recall that the masses of protons, neutrons, and electrons are VERY small numbers. These numbers are difficult to work with. • Scientists therefore developed a way to measure their mass relative to a standard atom (the carbon-12 atom). • One atomic mass unit (amu) is defined as 1/12 the mass of a ...
... • Recall that the masses of protons, neutrons, and electrons are VERY small numbers. These numbers are difficult to work with. • Scientists therefore developed a way to measure their mass relative to a standard atom (the carbon-12 atom). • One atomic mass unit (amu) is defined as 1/12 the mass of a ...
GEO143_lab_3_atoms_m..
... a full outer shell, atoms will gain or lose electrons. Since most of the outer shells hold eight electrons, this is called the octet rule, because the atom wants to have a full octet (eight) of electrons. This rule, or the potential to gain or lose an electron to maintain a full shell, is what gover ...
... a full outer shell, atoms will gain or lose electrons. Since most of the outer shells hold eight electrons, this is called the octet rule, because the atom wants to have a full octet (eight) of electrons. This rule, or the potential to gain or lose an electron to maintain a full shell, is what gover ...
Intro to the Periodic Table
... a. All atoms with 1 proton will be hydrogen atoms. b. All atoms with 6 protons will be carbon atoms. c. The number of protons in an atom changes very rarely and only with certain substances. 2. Each different type of atom is shown on the periodic table. a. There are 118 different types of atoms, eac ...
... a. All atoms with 1 proton will be hydrogen atoms. b. All atoms with 6 protons will be carbon atoms. c. The number of protons in an atom changes very rarely and only with certain substances. 2. Each different type of atom is shown on the periodic table. a. There are 118 different types of atoms, eac ...
Isotopes
... An unknown element, Q, has an average atomic mass of 73.75 amu. The first isotope of Q has a mass of 72.99 amu and the second isotope has a mass of 74.99 amu. What are the percentages of each isotope? ...
... An unknown element, Q, has an average atomic mass of 73.75 amu. The first isotope of Q has a mass of 72.99 amu and the second isotope has a mass of 74.99 amu. What are the percentages of each isotope? ...
Physical Science
... mixtures of isotopes. Isotopes are atoms of the same element that differ in the number of neutrons. ...
... mixtures of isotopes. Isotopes are atoms of the same element that differ in the number of neutrons. ...
10_Chemistry homework
... ____ Create an “Element Poster” * by including the information listed in the chart below. Create a poster for only one element. Look at the information listed in the "Scoring Criteria" column. If you do not know what a term means or how it applies to chemistry, use the links listed below the chart t ...
... ____ Create an “Element Poster” * by including the information listed in the chart below. Create a poster for only one element. Look at the information listed in the "Scoring Criteria" column. If you do not know what a term means or how it applies to chemistry, use the links listed below the chart t ...
Lawrencium

Lawrencium is a synthetic chemical element with chemical symbol Lr (formerly Lw) and atomic number 103. It is named in honor of Ernest Lawrence, inventor of the cyclotron, a device that was used to discover many artificial radioactive elements. A radioactive metal, lawrencium is the eleventh transuranic element and is also the final member of the actinide series. Like all elements with atomic number over 100, lawrencium can only be produced in particle accelerators by bombarding lighter elements with charged particles. Twelve isotopes of lawrencium are currently known; the most stable is 266Lr with a half-life of 11 hours, but the shorter-lived 260Lr (half-life 2.7 minutes) is most commonly used in chemistry because it can be produced on a larger scale.Chemistry experiments have confirmed that lawrencium indeed behaves as a heavier homolog to lutetium in the periodic table, and is a trivalent element. It thus could also be classified as the first of the 7th-period transition metals: however, its electron configuration is anomalous for its position in the periodic table, having an s2p configuration instead of the s2d configuration of its homolog lutetium. This means that lawrencium may be less volatile than expected for its position in the periodic table and have a volatility comparable to that of lead.In the 1950s, 1960s, and 1970s, many claims of the synthesis of lawrencium of varying quality were made from laboratories in the Soviet Union and the United States. The priority of the discovery and therefore the naming of the element was disputed between Soviet and American scientists, and while the International Union of Pure and Applied Chemistry (IUPAC) established lawrencium as the official name for the element and gave the American team credit for the discovery, this was reevaluated in 1997, giving both teams shared credit for the discovery but not changing the element's name.