Document
... series, the atomic radii actually ____________again. At the beginning of the series, the increase in __________ _______________ with atomic number pulls in the electron cloud, resulting in a reduction of atomic size. Since electrons are added to an inner d subshell across the series, this adds to th ...
... series, the atomic radii actually ____________again. At the beginning of the series, the increase in __________ _______________ with atomic number pulls in the electron cloud, resulting in a reduction of atomic size. Since electrons are added to an inner d subshell across the series, this adds to th ...
Ch.1-Matter and Change
... Introduction to the Periodic Table The vertical columns of the periodic table are called groups or families. Each group contains elements with similar chemical properties. The horizontal rows of elements in the periodic table are called periods. Physical and chemical properties change somewhat regul ...
... Introduction to the Periodic Table The vertical columns of the periodic table are called groups or families. Each group contains elements with similar chemical properties. The horizontal rows of elements in the periodic table are called periods. Physical and chemical properties change somewhat regul ...
Scandium and Yttrium - Mercyhurst University
... reaction.4 At room temperature, scandium metal turns a light yellow or pink color in air.1 It is not obvious what is causing the color as Sc3+ would be expected to be colorless (white) due to the lack of d-electrons. Purification and Properties The most interesting thing about these two elements is ...
... reaction.4 At room temperature, scandium metal turns a light yellow or pink color in air.1 It is not obvious what is causing the color as Sc3+ would be expected to be colorless (white) due to the lack of d-electrons. Purification and Properties The most interesting thing about these two elements is ...
Chapter 18: The Representative Elements The Representative
... Electron configuration is ns1(n = period number). Lose their valence e- easily (great reducing agents). Most violently reactive of all the metals. React strongly with H2O(l); the vigor of the reaction increases down the group. The alkali metals are all too easily oxidized to be found in thei ...
... Electron configuration is ns1(n = period number). Lose their valence e- easily (great reducing agents). Most violently reactive of all the metals. React strongly with H2O(l); the vigor of the reaction increases down the group. The alkali metals are all too easily oxidized to be found in thei ...
Chapter 18: The Representative Elements
... ns2np5 (n is the period number). In its elemental state, all halogens atoms combine to form diatomic molecules (ex. F2,I2,…). With the exception of F, the halogens can also lose valence electrons and their oxidation states can range from -1 to +7. Chapter 18: The Representative Elements ...
... ns2np5 (n is the period number). In its elemental state, all halogens atoms combine to form diatomic molecules (ex. F2,I2,…). With the exception of F, the halogens can also lose valence electrons and their oxidation states can range from -1 to +7. Chapter 18: The Representative Elements ...
Hydrogen, Alkalis, and Alkaline Earths
... The Hydrogen Economy Hydrogen is an attractive fuel because of its high heat of combustion and zero pollution ...
... The Hydrogen Economy Hydrogen is an attractive fuel because of its high heat of combustion and zero pollution ...
Inorganometallic Chemistry
... direct ionic or covalent localized or delocalized bond between one or more carbon atoms of an organic group or molecule and the atom from the main group, transition, lanthanide or actinide metals. According to the IUPAC rules, organometallic compounds are those in which the carbon atoms are bonded t ...
... direct ionic or covalent localized or delocalized bond between one or more carbon atoms of an organic group or molecule and the atom from the main group, transition, lanthanide or actinide metals. According to the IUPAC rules, organometallic compounds are those in which the carbon atoms are bonded t ...
The s-Block Elements
... reacts slowly with water. This illustrates the importance of the role of kinetic factors in determining the rate of a chemical reaction. Lithium has a higher m.p., this increases the activation energy required for dissolution in aqueous solution. It does not melt during the reaction as Na and K do, ...
... reacts slowly with water. This illustrates the importance of the role of kinetic factors in determining the rate of a chemical reaction. Lithium has a higher m.p., this increases the activation energy required for dissolution in aqueous solution. It does not melt during the reaction as Na and K do, ...
RxnTypesPrednotesIIAP
... There are three compounds that are commonly written in a molecular form, yet they do not exist as molecules. You need to commit these to memory. 1. ammonium hydroxide - NH4OH(aq) - is really an aqueous solution of ammonia gas. 2. sulfurous acid - H2SO3(aq) - is really an aqueous solution of sulfur d ...
... There are three compounds that are commonly written in a molecular form, yet they do not exist as molecules. You need to commit these to memory. 1. ammonium hydroxide - NH4OH(aq) - is really an aqueous solution of ammonia gas. 2. sulfurous acid - H2SO3(aq) - is really an aqueous solution of sulfur d ...
Chemistry of the Non
... 8 SO3–2 (aq) + S8 (s) 8 S2O3–2 (aq) Hypo (used in photography) is the pentahydrated salt of sodium thiosulfate (Na2S2O3 ● 5H2O). • Photographic film is a mixture of AgBr crystals on gelatin. • Exposure to light causes the AgBr to decompose to silver. • When the film is mildly reduced (developed) o ...
... 8 SO3–2 (aq) + S8 (s) 8 S2O3–2 (aq) Hypo (used in photography) is the pentahydrated salt of sodium thiosulfate (Na2S2O3 ● 5H2O). • Photographic film is a mixture of AgBr crystals on gelatin. • Exposure to light causes the AgBr to decompose to silver. • When the film is mildly reduced (developed) o ...
C1a Revision notes - Calthorpe Park Moodle
... a mixture into a number of different parts, called fractions. A tall column is fitted above the mixture, with several condensers coming off at different heights. The column is hot at the bottom and cool at the top. Substances with high boiling points condense at the bottom and substances with low bo ...
... a mixture into a number of different parts, called fractions. A tall column is fitted above the mixture, with several condensers coming off at different heights. The column is hot at the bottom and cool at the top. Substances with high boiling points condense at the bottom and substances with low bo ...
Complete the following equations
... decreases from Be to B and from N to O; in the third period, ionization energy decreases from Mg to Al and from P to S. Explain why the anomaly. (In beryllium electron is removed from 2s orbital, but it is removed from 2p orbital in boron. It is easier to remove an electron from a 2p orbital since i ...
... decreases from Be to B and from N to O; in the third period, ionization energy decreases from Mg to Al and from P to S. Explain why the anomaly. (In beryllium electron is removed from 2s orbital, but it is removed from 2p orbital in boron. It is easier to remove an electron from a 2p orbital since i ...
Topic 4 Chemistry of the Elements of the Main Group
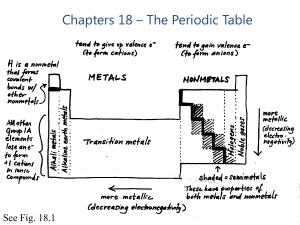
... groups: Metals, Metalloids and non-Metals Non-metals are not electrical conductors, they are characterised by a high electronegativity value (over 2). The valence electrons of non-metals are strongly attracted to their positively charged nucleus and are not available to conduct electricity. Metals ...
... groups: Metals, Metalloids and non-Metals Non-metals are not electrical conductors, they are characterised by a high electronegativity value (over 2). The valence electrons of non-metals are strongly attracted to their positively charged nucleus and are not available to conduct electricity. Metals ...
Elements – (Metals)
... Metals readily lose one or more electrons since electron configuration is such that many inner electrons shield full charge of nucleus and so outer electrons are not as strongly held. For example K forms K+ but Cl is apt to gain electron to become Cl–. Notice that both K+ and Cl– are isoelectronic ( ...
... Metals readily lose one or more electrons since electron configuration is such that many inner electrons shield full charge of nucleus and so outer electrons are not as strongly held. For example K forms K+ but Cl is apt to gain electron to become Cl–. Notice that both K+ and Cl– are isoelectronic ( ...
Preview Sample 1
... 41. When electrons are shared between pairs of atoms rather than donated from one atom to another or mobile across an entire lattice, a(n) ____________________ bond is present. ________________________________________ ...
... 41. When electrons are shared between pairs of atoms rather than donated from one atom to another or mobile across an entire lattice, a(n) ____________________ bond is present. ________________________________________ ...
QA1
... The wire is then ready to be used. If a sample solution is being tested, immerse the wire into the solution and then put the wire into a colourless flame and note the colour of the flame. Caution should be made not to burn the glass part of the platinum wire; otherwise, it will be broken. Sodium com ...
... The wire is then ready to be used. If a sample solution is being tested, immerse the wire into the solution and then put the wire into a colourless flame and note the colour of the flame. Caution should be made not to burn the glass part of the platinum wire; otherwise, it will be broken. Sodium com ...
IT IS ELEMENTARY - the OLLI at UCI Blog
... and animal origin • These elements or their very simple compounds can kill—most commonly by interfering with cellular access to oxygen • Nitrogen N2 • Carbon dioxide CO2 • Carbon monoxide CO • Hydrogen cyanide HCN ...
... and animal origin • These elements or their very simple compounds can kill—most commonly by interfering with cellular access to oxygen • Nitrogen N2 • Carbon dioxide CO2 • Carbon monoxide CO • Hydrogen cyanide HCN ...
Atoms, Elements and Compounds Home
... Over 300 years ago Isaac Newton had shown that white light was a mixture of all the different colours of the rainbow. Scientists had developed this idea into the spectroscope, which allowed them to look in great detail at the spectrum of light from different sources. When some elements are heated th ...
... Over 300 years ago Isaac Newton had shown that white light was a mixture of all the different colours of the rainbow. Scientists had developed this idea into the spectroscope, which allowed them to look in great detail at the spectrum of light from different sources. When some elements are heated th ...
Biologically Important Inorganic Elements Occurrence and Availability
... transition metal in sea water as MoO4 has fairly high solubility in water. Better correlation exists between the abundance of elements in in human body and in sea water than between the human body and the earth's crust. Taken as evidence for the oceans as the site of evolution of life. • Despite th ...
... transition metal in sea water as MoO4 has fairly high solubility in water. Better correlation exists between the abundance of elements in in human body and in sea water than between the human body and the earth's crust. Taken as evidence for the oceans as the site of evolution of life. • Despite th ...
periodic table - Mesa Community College
... Compounds are defined as pure substances that can be broken down to simpler pure substances by a chemical reaction. The smallest particle of an element that can take part in a chemical reaction and form a compound is called an atom. It consists of a center (nucleus) with protons and neutrons and aro ...
... Compounds are defined as pure substances that can be broken down to simpler pure substances by a chemical reaction. The smallest particle of an element that can take part in a chemical reaction and form a compound is called an atom. It consists of a center (nucleus) with protons and neutrons and aro ...
PVS103 - unit 6 notes
... • Boron is unique in the group in that it is clearly a non-metal, we will concentrate on its properties, as it is very interesting. • The molecules boron forms are unique in that they do not conform fully to Lewis theory, for instance BH3 is a stable molecule, but there is no octet of electrons on b ...
... • Boron is unique in the group in that it is clearly a non-metal, we will concentrate on its properties, as it is very interesting. • The molecules boron forms are unique in that they do not conform fully to Lewis theory, for instance BH3 is a stable molecule, but there is no octet of electrons on b ...
Chapters 18 – The Periodic Table
... Some Important Compounds of the Oxygen Family 1. Hydrogen sulfide, H2S. Vile toxic gas formed during anaerobic decomposition of plant and animal matter, in volcanoes, and in deep sea thermal vents. Used in the manufacture of paper. Atmospheric traces cause silver to tarnish through formation of bla ...
... Some Important Compounds of the Oxygen Family 1. Hydrogen sulfide, H2S. Vile toxic gas formed during anaerobic decomposition of plant and animal matter, in volcanoes, and in deep sea thermal vents. Used in the manufacture of paper. Atmospheric traces cause silver to tarnish through formation of bla ...
Periodic Table of Elements
... Metalloids Have some properties of both metals and nonmetals. They are located on the zigzag line between the metals and the nonmetals. Physical Properties: ...
... Metalloids Have some properties of both metals and nonmetals. They are located on the zigzag line between the metals and the nonmetals. Physical Properties: ...