surface chemistry - einstein classes
... a true solution is can not be seen. When a same light is passed through a colloidal solution it become visible as a bright streak. This phenomenon is called Tyndall effect and illuminated path (streak of light) is known as Tyndall cone. The appearance of dust particles in a semidarkened room when a ...
... a true solution is can not be seen. When a same light is passed through a colloidal solution it become visible as a bright streak. This phenomenon is called Tyndall effect and illuminated path (streak of light) is known as Tyndall cone. The appearance of dust particles in a semidarkened room when a ...
Solutions
... When a solution contains the maximum amount of solute that will dissolve at a specific temperature it is_________. In this case the amount dissolved will be _________the line of solubility. Solution____________. When a solution contains more than the maximum amount of solute that will dissolve at a ...
... When a solution contains the maximum amount of solute that will dissolve at a specific temperature it is_________. In this case the amount dissolved will be _________the line of solubility. Solution____________. When a solution contains more than the maximum amount of solute that will dissolve at a ...
Phase Rule
... indicates that there is only one set of variables P, T at which all the three phases coexist. If any of the variables is changed, then the number of phases decreases. For example, if the temperature is raised, heat causes more and more of the solid (ice) to melt but no change in temperature or press ...
... indicates that there is only one set of variables P, T at which all the three phases coexist. If any of the variables is changed, then the number of phases decreases. For example, if the temperature is raised, heat causes more and more of the solid (ice) to melt but no change in temperature or press ...
Branham
... The following study deals with the characteristics of the reaction to form BaSO4 and BaCO3 from Na2SO4 and Na2CO3, respectively, using BaS as the other reactant. These reactions would theoretically increase the efficiency of the chemical recovery process in papermaking to near 100% by completely con ...
... The following study deals with the characteristics of the reaction to form BaSO4 and BaCO3 from Na2SO4 and Na2CO3, respectively, using BaS as the other reactant. These reactions would theoretically increase the efficiency of the chemical recovery process in papermaking to near 100% by completely con ...
Porous silicon-based nanostructured microparticles as degradable
... 156.4, 144.0 (2 overlapping signals), 141.3 (2 overlapping signals), 139.2,127.6 (2 overlapping signals), 127.5 (2 overlapping signals) 125.0 (2 overlapping signals), 119.9 (2 overlapping signals), 114.2, 66.5, 47.3, 41.1, 33.8, 30.0, 29.5, 29.4, 29.3, 29.1, 28.9 and 26.7. ESI-MS: predicted C26H33 ...
... 156.4, 144.0 (2 overlapping signals), 141.3 (2 overlapping signals), 139.2,127.6 (2 overlapping signals), 127.5 (2 overlapping signals) 125.0 (2 overlapping signals), 119.9 (2 overlapping signals), 114.2, 66.5, 47.3, 41.1, 33.8, 30.0, 29.5, 29.4, 29.3, 29.1, 28.9 and 26.7. ESI-MS: predicted C26H33 ...
Fig. 3: Particle size distribution of highly active nuclear waste
... there are jet ballasts and air-lifts to maintain the dispersion, some of the dense particulate materials are expected to settle rapidly out at the bottom of the tank and on other pipework. Studies of settled beds of the particulates can provide insight into the permeability and compressibility of an ...
... there are jet ballasts and air-lifts to maintain the dispersion, some of the dense particulate materials are expected to settle rapidly out at the bottom of the tank and on other pipework. Studies of settled beds of the particulates can provide insight into the permeability and compressibility of an ...
Size-Resolved Kinetic Measurements of Aluminum Nanoparticle
... with primary particle size greater than ∼50 nm were not fully oxidized even at 1100 °C (∼4%). The absolute rates observed were found to be consistent with an oxide diffusion controlled rate-limiting step. We also determined the size-dependent diffusion-limited rate constants and Arrehenius parameter ...
... with primary particle size greater than ∼50 nm were not fully oxidized even at 1100 °C (∼4%). The absolute rates observed were found to be consistent with an oxide diffusion controlled rate-limiting step. We also determined the size-dependent diffusion-limited rate constants and Arrehenius parameter ...
Optical Properties of Rutile Single Crystals Grown Under Pressure
... commercially available rutile single crystals grown by the Verneuil method, are generally not good enough to use in optical devices because of their dislocation density due to presence of Magnel phases (TiO2-δ) i.e. oxygen deficient phases. The optical quality of the FZ-grown crystals was superior t ...
... commercially available rutile single crystals grown by the Verneuil method, are generally not good enough to use in optical devices because of their dislocation density due to presence of Magnel phases (TiO2-δ) i.e. oxygen deficient phases. The optical quality of the FZ-grown crystals was superior t ...
Lecture 19 - University of Windsor
... Let’s zoom in and follow the changes along the isopleth from a - e (as the temperature is dropped): a. 1375 °C. The material is above the liquidus line and so the entire mixture is still liquid. ...
... Let’s zoom in and follow the changes along the isopleth from a - e (as the temperature is dropped): a. 1375 °C. The material is above the liquidus line and so the entire mixture is still liquid. ...
Synthesis of Copper (II)-Containing Nickel (II) Hydroxide Particles as
... Chem. Mater., Vol. 10, No. 6, 1998 1633 All solutions were made up using analytical grade reagents and deionized water (18 MΩ cm) obtained from a Milli-Q apparatus. All glassware was washed using a sulfonitric acid solution (10 g of H2SO4 and 10 g of HNO3 in 100 mL) and rinsed thoroughly with water ...
... Chem. Mater., Vol. 10, No. 6, 1998 1633 All solutions were made up using analytical grade reagents and deionized water (18 MΩ cm) obtained from a Milli-Q apparatus. All glassware was washed using a sulfonitric acid solution (10 g of H2SO4 and 10 g of HNO3 in 100 mL) and rinsed thoroughly with water ...
Concentration of solutions
... • Nature of solute and solvent (polarity, etc.) Solute-solvent interactions - “like dissolves like” • Temperature (for gases an increase in T decreases solubility). For the majority of solids in liquids, increasing T increases their solubility. The degree of solubility can differ greatly and in som ...
... • Nature of solute and solvent (polarity, etc.) Solute-solvent interactions - “like dissolves like” • Temperature (for gases an increase in T decreases solubility). For the majority of solids in liquids, increasing T increases their solubility. The degree of solubility can differ greatly and in som ...
lecture 5 phase equilibria
... temperature at which phase separation occurs. • Above the critical temperature the two components are fully miscible. • On the molecular level, this can be interpreted as the kinetic energy of each molecule over coming molecular interactions that want molecules of one type to come close together. ...
... temperature at which phase separation occurs. • Above the critical temperature the two components are fully miscible. • On the molecular level, this can be interpreted as the kinetic energy of each molecule over coming molecular interactions that want molecules of one type to come close together. ...
Solutions
... have higher boiling points than pure solvents. This is true with solid solutes and heavier liquid solutes. Other liquid solutes may form azeotropes, which are mixtures with lower boiling points than either solute or solvent – example 95% ethanol/water.) Solutes raise the boiling point of liquids ...
... have higher boiling points than pure solvents. This is true with solid solutes and heavier liquid solutes. Other liquid solutes may form azeotropes, which are mixtures with lower boiling points than either solute or solvent – example 95% ethanol/water.) Solutes raise the boiling point of liquids ...
solute
... Solution – homogeneous mixture in which solute & solvent particles are evenly distributed in one another solvent – the dissolving medium; thing that does the dissolving solute –the dissolved particles; thing that is dissolved solutes and solvents can be solids, liquids, or gases ...
... Solution – homogeneous mixture in which solute & solvent particles are evenly distributed in one another solvent – the dissolving medium; thing that does the dissolving solute –the dissolved particles; thing that is dissolved solutes and solvents can be solids, liquids, or gases ...
Title Magnetic Properties of Several Iron Compounds Studied by the
... situation.2 A great fortunate for investigators of solid state, especially of magnetism, is the fact that Fe57 is one of the best Mossbauer nuclei. While the effect originally belonged to nuclear physics, solid state application is now much dominant. Above all, Fe57Mossbauer measurement is regarded ...
... situation.2 A great fortunate for investigators of solid state, especially of magnetism, is the fact that Fe57 is one of the best Mossbauer nuclei. While the effect originally belonged to nuclear physics, solid state application is now much dominant. Above all, Fe57Mossbauer measurement is regarded ...
Cloud droplet activation and surface tension of mixtures of slightly
... major role for the activation of particles consisting of slightly soluble organic compounds (Bilde and Svenningsson, 2004; Hori et al., 2003; Broekhuizen et al., 2004). In the atmosphere particles are most likely to be mixtures of organic and inorganic components. Even if a purely organic particle e ...
... major role for the activation of particles consisting of slightly soluble organic compounds (Bilde and Svenningsson, 2004; Hori et al., 2003; Broekhuizen et al., 2004). In the atmosphere particles are most likely to be mixtures of organic and inorganic components. Even if a purely organic particle e ...
Test3_sp2012with answers
... _BF________H2CO3(aq) = H2O(l) + CO2(g) _CE________CuSO4(aq) + Zn(s) = ZnSO4(aq) + Cu(s) 31. Match each condition with a physical state. Mark ALL correct answers. There can be more than one correct answer. __B______great distance between particles ...
... _BF________H2CO3(aq) = H2O(l) + CO2(g) _CE________CuSO4(aq) + Zn(s) = ZnSO4(aq) + Cu(s) 31. Match each condition with a physical state. Mark ALL correct answers. There can be more than one correct answer. __B______great distance between particles ...
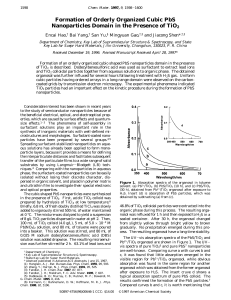
Formation of Orderly Organized Cubic PbS Nanoparticles Domain in
... surfactant solutions play an important role in the synthesis of inorganic materials with well-defined microstructures and morphologies. Surfactant-coated nanoparticles have been prepared by several groups.4,5 Spreading surfactant-stabilized nanoparticles on aqueous solutions has already been applied ...
... surfactant solutions play an important role in the synthesis of inorganic materials with well-defined microstructures and morphologies. Surfactant-coated nanoparticles have been prepared by several groups.4,5 Spreading surfactant-stabilized nanoparticles on aqueous solutions has already been applied ...
Effect of Sulfuric Acid Manufacturing Process on
... • Inline defect detection can also miss the defects on the wafer in similar situations where defects are smaller than the detection limit. • Any particle excursion in any chemical can potentially degrade the inline defects. • Sulfuric acid is usually produced using either glass or Teflon-lined steel ...
... • Inline defect detection can also miss the defects on the wafer in similar situations where defects are smaller than the detection limit. • Any particle excursion in any chemical can potentially degrade the inline defects. • Sulfuric acid is usually produced using either glass or Teflon-lined steel ...
Nanoparticle Suspension Preparation Using Ultrasonic Vibration
... (XRD) to confirm the lattice structure of particles. The nanoparticles produced from the process are anatase type TiO2 . Among the heat source variables in the electric arc discharge, the magnitude of the pulse duration is found to have an influence on the submerged arc temperature and the crater radi ...
... (XRD) to confirm the lattice structure of particles. The nanoparticles produced from the process are anatase type TiO2 . Among the heat source variables in the electric arc discharge, the magnitude of the pulse duration is found to have an influence on the submerged arc temperature and the crater radi ...
Thermodynamics of Crystal-Melt Phase Change
... by transforming it from solid to liquid, or vice versa, is equal to the changes in the system’s enthalpy function. Equation (2.8) also demonstrates an extremely useful property of the enthalpy function that allows one to sketch an enthalpy-temperature phase diagram for a pure substance, held at fixe ...
... by transforming it from solid to liquid, or vice versa, is equal to the changes in the system’s enthalpy function. Equation (2.8) also demonstrates an extremely useful property of the enthalpy function that allows one to sketch an enthalpy-temperature phase diagram for a pure substance, held at fixe ...
IOSR Journal of Applied Physics (IOSR-JAP)
... diamond indenter, in the form of a right pyramid with a square base and an angle of 136º. The loads varying from 10g-50g were applied over a fixed interval of time. The full load is normally applied for 10 to 15 seconds. The two diagonals of the indentation left in the surface of the material after ...
... diamond indenter, in the form of a right pyramid with a square base and an angle of 136º. The loads varying from 10g-50g were applied over a fixed interval of time. The full load is normally applied for 10 to 15 seconds. The two diagonals of the indentation left in the surface of the material after ...
The Physical Properties And Physical Changes of Substances
... suitable solvent. The solvent is allowed to slowly evaporate causing some of the desired solid to come out of solution as crystals; however, not all the solvent is allowed to evaporate. The crystals that are formed can then be separated by HAND SEPARATION or FILTRATION. ...
... suitable solvent. The solvent is allowed to slowly evaporate causing some of the desired solid to come out of solution as crystals; however, not all the solvent is allowed to evaporate. The crystals that are formed can then be separated by HAND SEPARATION or FILTRATION. ...
B. The Physical Properties of Matter
... suitable solvent. The solvent is allowed to slowly evaporate causing some of the desired solid to come out of solution as crystals; however, not all the solvent is allowed to evaporate. The crystals that are formed can then be separated by HAND SEPARATION or FILTRATION. ...
... suitable solvent. The solvent is allowed to slowly evaporate causing some of the desired solid to come out of solution as crystals; however, not all the solvent is allowed to evaporate. The crystals that are formed can then be separated by HAND SEPARATION or FILTRATION. ...