Surface Tension Measurements
... Surface Tension Measurements In this laboratory exercise we will measure the Surface Tension of Methanol as a function of temperature. This will allow us to determine the Surface Energy (ES) of this liquid. Also, the Surface Tension of aqueous solutions of Ethanol and n-Butanol will be measured as a ...
... Surface Tension Measurements In this laboratory exercise we will measure the Surface Tension of Methanol as a function of temperature. This will allow us to determine the Surface Energy (ES) of this liquid. Also, the Surface Tension of aqueous solutions of Ethanol and n-Butanol will be measured as a ...
Structure and Properties of the Shikimate Pathway Type I D 3
... alignment). The structure was refined to R (Rfree) = 0.18 (0.23) using data to 2.60 Å from an orthorhombic crystal, space group I222, with parameters a = 87.231, b = 110.024, c = 144.350 Å, = = = 90o and Z (Z') = 16(2). The structure is a (/)8 barrel, which crystallographically appears as a ...
... alignment). The structure was refined to R (Rfree) = 0.18 (0.23) using data to 2.60 Å from an orthorhombic crystal, space group I222, with parameters a = 87.231, b = 110.024, c = 144.350 Å, = = = 90o and Z (Z') = 16(2). The structure is a (/)8 barrel, which crystallographically appears as a ...
Physical concept of the surface tension of the liquid until some time
... coefficient n must be equal to unity. The processing of the reference data of the thermophysical properties of most substances has confirmed the theoretical formulas with an accuracy of 15%. Some substances with asymmetric form possess a factor of the molecule efficiency different from the unit. Fur ...
... coefficient n must be equal to unity. The processing of the reference data of the thermophysical properties of most substances has confirmed the theoretical formulas with an accuracy of 15%. Some substances with asymmetric form possess a factor of the molecule efficiency different from the unit. Fur ...
Modelling Mass Transfer in Nitrification Processes Using
... strongly related to the lattice structure. • Most selective oxidation reactions kinetics can be described in terms of the “REDOX” mechanism. • Until recently, few studies focussed on the catalyst surface dynamics in relation to the “REDOX” properties. The secret is ...
... strongly related to the lattice structure. • Most selective oxidation reactions kinetics can be described in terms of the “REDOX” mechanism. • Until recently, few studies focussed on the catalyst surface dynamics in relation to the “REDOX” properties. The secret is ...
Total
... (A) cannot be used to solve one-electron atoms (B) can be solved exactly only for one-electron atoms (C) is not used for large molecules ...
... (A) cannot be used to solve one-electron atoms (B) can be solved exactly only for one-electron atoms (C) is not used for large molecules ...
Nonlinear laser lithography to control surface properties of stainless
... the operating temperature in order to induce different wettability behaviour. Some of the above methods are expensive, complicated and time consuming; others involve chemical treatments and weak coatings that are generally evanescent or fragile. Use of methods that only change the surface morphology ...
... the operating temperature in order to induce different wettability behaviour. Some of the above methods are expensive, complicated and time consuming; others involve chemical treatments and weak coatings that are generally evanescent or fragile. Use of methods that only change the surface morphology ...
Coefficient of Sliding Friction
... with which it is in contact, Newton’s 1st Law requires application of a force with a component parallel to the direction of motion which is equal in magnitude but opposite in direction to the force of friction, which opposes the motion. ...
... with which it is in contact, Newton’s 1st Law requires application of a force with a component parallel to the direction of motion which is equal in magnitude but opposite in direction to the force of friction, which opposes the motion. ...
Surface and colloidal chemistry
... • Water does not wet waxed/oily surfaces because • cohesive forces within the water drops >> the adhesive forces between the ...
... • Water does not wet waxed/oily surfaces because • cohesive forces within the water drops >> the adhesive forces between the ...
Dimples due to dislocations at the superfluid/solid interface of
... nation is that spiral growth takes place along dislocation lines, penetrating the crystal. These imperfections are expected to form definite traces on the surface, viz., dimples whose observation using high-resolution interferometric techniques should be possible. Recently, it has been shown that su ...
... nation is that spiral growth takes place along dislocation lines, penetrating the crystal. These imperfections are expected to form definite traces on the surface, viz., dimples whose observation using high-resolution interferometric techniques should be possible. Recently, it has been shown that su ...
EXPERIMENT NO: 2
... type I, addition of solute leads to an increase in surface tension, but the increase is generally not large. Such behavior is exhibited by strong electrolytes, sucrose, and aminobenzoic acid in water. On the other hand, with non-electrolytes or weak electrolytes in water, the behavior most often en ...
... type I, addition of solute leads to an increase in surface tension, but the increase is generally not large. Such behavior is exhibited by strong electrolytes, sucrose, and aminobenzoic acid in water. On the other hand, with non-electrolytes or weak electrolytes in water, the behavior most often en ...
Introduction to Nanoscience Study Guide
... hydrogens and do not require any more satisfaction. The contact angle of drops on this surface tend to be greater than 90° (e.g. the water is more spherical in shape). Hydrophilic surfaces, on the other hand, do have enough energy to break the surface tension of water. For example, a metal like gold ...
... hydrogens and do not require any more satisfaction. The contact angle of drops on this surface tend to be greater than 90° (e.g. the water is more spherical in shape). Hydrophilic surfaces, on the other hand, do have enough energy to break the surface tension of water. For example, a metal like gold ...
Hw01.pdf
... Use the Excel function RAND()to create 1000 simulated (pseudo) random numbers R, substitute in the above equation and assume that the computed values of z represent asperity heights measured with a profilometer at distances of 1 micrometer along the surface. a) Plot the surface profile thus obtained ...
... Use the Excel function RAND()to create 1000 simulated (pseudo) random numbers R, substitute in the above equation and assume that the computed values of z represent asperity heights measured with a profilometer at distances of 1 micrometer along the surface. a) Plot the surface profile thus obtained ...
Fundamentals of Adhesion
... of attraction between unlike materials. The strength of attraction is determined by the surface energy of the material. The higher the surface energy, the greater the molecular attraction. The lower the surface energy, the weaker the attractive forces. ...
... of attraction between unlike materials. The strength of attraction is determined by the surface energy of the material. The higher the surface energy, the greater the molecular attraction. The lower the surface energy, the weaker the attractive forces. ...
08. Physical-chemical essence of surface phenomenon
... • The phenomenon of attracting and retaining the molecules of а substance on the surface of а liquid or а solid resulting into a higher concentration of the molecules on the surface is called adsorption. • The substance thus adsorbed on the surface is called the adsorbate and the substance on which ...
... • The phenomenon of attracting and retaining the molecules of а substance on the surface of а liquid or а solid resulting into a higher concentration of the molecules on the surface is called adsorption. • The substance thus adsorbed on the surface is called the adsorbate and the substance on which ...
04. Physical-chemical essence of surface phenomenon
... • The phenomenon of attracting and retaining the molecules of а substance on the surface of а liquid or а solid resulting into a higher concentration of the molecules on the surface is called adsorption. • The substance thus adsorbed on the surface is called the adsorbate and the substance on which ...
... • The phenomenon of attracting and retaining the molecules of а substance on the surface of а liquid or а solid resulting into a higher concentration of the molecules on the surface is called adsorption. • The substance thus adsorbed on the surface is called the adsorbate and the substance on which ...
Functionalizing spin-textured surfaces with tailored organic bonds
... Recent developments in molecular spintronics indicate that the deposition of aromatic organic molecules on the strongly reactive surfaces of ferromagnetic metals leads to a change in the local magnetic properties of the atoms hybridized with the molecule, such as exchange interaction, magnetic momen ...
... Recent developments in molecular spintronics indicate that the deposition of aromatic organic molecules on the strongly reactive surfaces of ferromagnetic metals leads to a change in the local magnetic properties of the atoms hybridized with the molecule, such as exchange interaction, magnetic momen ...
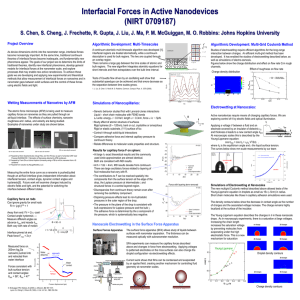
Interfacial Forces in Active Nanodevices
... and retracted from water interface Forces consistent with bulk surface tension and contact angles: θadv = 58°, θrec = 47° ...
... and retracted from water interface Forces consistent with bulk surface tension and contact angles: θadv = 58°, θrec = 47° ...
Wetting

Wetting is the ability of a liquid to maintain contact with a solid surface, resulting from intermolecular interactions when the two are brought together. The degree of wetting (wettability) is determined by a force balance between adhesive and cohesive forces. Wetting deals with the three phases of materials: gas, liquid, and solid. It is now a center of attention in nanotechnology and nanoscience studies due to the advent of many nanomaterials in the past two decades (e.g. graphene, carbon nanotube).Wetting is important in the bonding or adherence of two materials. Wetting and the surface forces that control wetting are also responsible for other related effects, including so-called capillary effects. Regardless of the amount of wetting, the shape of a liquid drop on a rigid surface is roughly a truncated sphere.There are two types of wetting: non-reactive wetting and active wetting.