Ionic Liquid Crystals - American Chemical Society
... Ordered smectic phases exhibit long-range bond orientational order of the molecules but short-range positional order within the smectic layers. In the smectic B phase (SmB) there is a 6-fold bondorientational order, which means that the lattice orientation is retained in the layers, but the translat ...
... Ordered smectic phases exhibit long-range bond orientational order of the molecules but short-range positional order within the smectic layers. In the smectic B phase (SmB) there is a 6-fold bondorientational order, which means that the lattice orientation is retained in the layers, but the translat ...
A thermodynamic model for the prediction of phase equilibria and
... the two salts or the equilibrium constants of the above four reactions. Within the last several decades, many researchers have developed different methods to calculate the equilibrium constants (Ruaya, 1988; Mesmer et al., 1988; Anderson et al., 1991) or the chemical potentials of the species (Helges ...
... the two salts or the equilibrium constants of the above four reactions. Within the last several decades, many researchers have developed different methods to calculate the equilibrium constants (Ruaya, 1988; Mesmer et al., 1988; Anderson et al., 1991) or the chemical potentials of the species (Helges ...
atomistic modelling of nanogranular magnetic materials
... the charge on the atom to have a net angular momentum. Any flow of charge causes additional physical effects on the surroundings, usually referred to as a ’magnetic’ effect. In the case of atoms the net angular momentum of the charge cloud causes a magnetic field perpendicular to the rotation of the ...
... the charge on the atom to have a net angular momentum. Any flow of charge causes additional physical effects on the surroundings, usually referred to as a ’magnetic’ effect. In the case of atoms the net angular momentum of the charge cloud causes a magnetic field perpendicular to the rotation of the ...
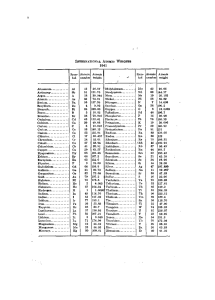
INTEKNATIONAL ATOMIC WEIGHTS Aluminum... Antimony..., Argon
... when it is too long, the instructor will select parts of the "Procedure" which may be omitted. Several of the procedures have been expanded and the directions made more specific without sacrificing the development of self-reUance on the part of the student, which has always been one of the aims of t ...
... when it is too long, the instructor will select parts of the "Procedure" which may be omitted. Several of the procedures have been expanded and the directions made more specific without sacrificing the development of self-reUance on the part of the student, which has always been one of the aims of t ...
Equilibrium Expression (Keq)
... Try to write a dissolution equation for CaCl2(s) CaCl2(s) ↔ Ca+2(aq) + 2Cl-1(aq) ...
... Try to write a dissolution equation for CaCl2(s) CaCl2(s) ↔ Ca+2(aq) + 2Cl-1(aq) ...
PHYSICAL CHEMISTRY IN BRIEF
... This publication is not intended to substitute for any textbooks or books of examples. Yet we believe that it will prove useful during revision lessons leading up to an exam in Physical Chemistry or prior to the final (state) examination, as well as during postgraduate studies. Even experts in Physi ...
... This publication is not intended to substitute for any textbooks or books of examples. Yet we believe that it will prove useful during revision lessons leading up to an exam in Physical Chemistry or prior to the final (state) examination, as well as during postgraduate studies. Even experts in Physi ...
CE 303-121- lecture 17
... for tack coat and surface treatments. Specifications for RC type given in ASTM D2028. – Medium-Curing (MC). Produced by adding a medium diluent of intermediate volatility (generally kerosene) to asphalt cement. These are generally used for prime coat, stockpile patching mixtures, and road-mixing ope ...
... for tack coat and surface treatments. Specifications for RC type given in ASTM D2028. – Medium-Curing (MC). Produced by adding a medium diluent of intermediate volatility (generally kerosene) to asphalt cement. These are generally used for prime coat, stockpile patching mixtures, and road-mixing ope ...
Silica (quartz) Silica, SiO2, is a white or colorless crystalline
... Silica (quartz): Silica, SiO2, is a chemical compound that is composed of one silicon atom and two oxygen atoms. It appears naturally in several crystalline forms, one of which is quartz. Silica Quartz- A colorless, ordorless crystal found in different colors such as white, green, black, purple. It ...
... Silica (quartz): Silica, SiO2, is a chemical compound that is composed of one silicon atom and two oxygen atoms. It appears naturally in several crystalline forms, one of which is quartz. Silica Quartz- A colorless, ordorless crystal found in different colors such as white, green, black, purple. It ...
Liquid Intrusion and Alternative Methods for the Characterisation of
... The main objective of this document is to review critically the various liquid intrusion techniques commonly used to assess the pore size of materials containing macropores (i.e., pores of width >50 nm), especially those in the approximate range 50 nm to 500 μm. Further objectives are to examine the ...
... The main objective of this document is to review critically the various liquid intrusion techniques commonly used to assess the pore size of materials containing macropores (i.e., pores of width >50 nm), especially those in the approximate range 50 nm to 500 μm. Further objectives are to examine the ...
Structure and packing of phosphatidylcholines in lamellar and
... area of the bilayer.11-23 Note that this equation considers macroscopic properties. It is desirable, however, to introduce the microscopic average properties as obtained from 2H NMR. As it is only for the “plateau” values that we can utilize a ”cell shape” corresponding to a rectangular parallelepip ...
... area of the bilayer.11-23 Note that this equation considers macroscopic properties. It is desirable, however, to introduce the microscopic average properties as obtained from 2H NMR. As it is only for the “plateau” values that we can utilize a ”cell shape” corresponding to a rectangular parallelepip ...
Perspectives on the Physical Chemistry of
... known about the structure and composition of colloidal semiconductor nanocrystal surfaces. Independent of the large number of surface atoms, semiconductor nanocrystals with the same interior bonding geometry as a known bulk phase often exhibit strong variations in their optical and electrical proper ...
... known about the structure and composition of colloidal semiconductor nanocrystal surfaces. Independent of the large number of surface atoms, semiconductor nanocrystals with the same interior bonding geometry as a known bulk phase often exhibit strong variations in their optical and electrical proper ...
Thermodynamics and Phase Diagrams
... where kB is Boltzmann’s constant and t is the multiplicity of the system. Somewhat loosely, t is the number of possible equivalent microstates in a macrostate, that is the number of quantum states for the system which are accessible under the applicable conditions of energy, volume, etc. For example ...
... where kB is Boltzmann’s constant and t is the multiplicity of the system. Somewhat loosely, t is the number of possible equivalent microstates in a macrostate, that is the number of quantum states for the system which are accessible under the applicable conditions of energy, volume, etc. For example ...
Redox reactions in deep eutectic solvents
... ideas for new experimental devices in to real objects. ...
... ideas for new experimental devices in to real objects. ...
Beverley John C. Beverley IE 500/PHI 598: Ontological Engineering
... As indicated above, Thermodynamics is the study of energy, but more specifically, it is the study of energy inhering in a Thermodynamic System. Such systems will be formally defined below in the Classes section, but the intuition underlying the concept is easily grasped: they are arbitrary regions o ...
... As indicated above, Thermodynamics is the study of energy, but more specifically, it is the study of energy inhering in a Thermodynamic System. Such systems will be formally defined below in the Classes section, but the intuition underlying the concept is easily grasped: they are arbitrary regions o ...
as PDF
... The soft-combustion technique offers several advantages over conventional high temperature and other low temperature methods. Materials prepared via the solid-state route contain two-phase mixtures due to the inhomogeneity caused by physical mixing of the raw materials. The particle morphology is of ...
... The soft-combustion technique offers several advantages over conventional high temperature and other low temperature methods. Materials prepared via the solid-state route contain two-phase mixtures due to the inhomogeneity caused by physical mixing of the raw materials. The particle morphology is of ...
Glass transition
The glass–liquid transition or glass transition for short is the reversible transition in amorphous materials (or in amorphous regions within semicrystalline materials) from a hard and relatively brittle state into a molten or rubber-like state. An amorphous solid that exhibits a glass transition is called a glass. Supercooling a viscous liquid into the glass state is called vitrification, from the Latin vitreum, ""glass"" via French vitrifier.Despite the massive change in the physical properties of a material through its glass transition, the transition is not itself a phase transition of any kind; rather it is a laboratory phenomenon extending over a range of temperature and defined by one of several conventions. Such conventions include a constant cooling rate (20 K/min) and a viscosity threshold of 1012 Pa·s, among others. Upon cooling or heating through this glass-transition range, the material also exhibits a smooth step in the thermal-expansion coefficient and in the specific heat, with the location of these effects again being dependent on the history of the material. However, the question of whether some phase transition underlies the glass transition is a matter of continuing research.The glass-transition temperature Tg is always lower than the melting temperature, Tm, of the crystalline state of the material, if one exists.