main
... They describe relationships between the fundamental quantities energy, entropy, temperature and heat, which are used to characterize thermodynamic systems. The four laws are very useful to test the validity of new physics. If you propose a new theory in physics and it does not obey the laws of therm ...
... They describe relationships between the fundamental quantities energy, entropy, temperature and heat, which are used to characterize thermodynamic systems. The four laws are very useful to test the validity of new physics. If you propose a new theory in physics and it does not obey the laws of therm ...
Q - Effingham County Schools
... The molecules may also have potential energy in their vibrations and bending. The gas molecules collide with each other and with the walls of their container, transferring energy among each other in the process ...
... The molecules may also have potential energy in their vibrations and bending. The gas molecules collide with each other and with the walls of their container, transferring energy among each other in the process ...
Statistical Mechanics--
... overlooked consequence of these values. If correct, the mixing of gases by diffusion should occur much more rapidly than is observed experimentally. A smell produced in one corner of a room should be noticed instantly in the opposite corner, and it isn’t. (BB thought that this killed C’s theory). Cl ...
... overlooked consequence of these values. If correct, the mixing of gases by diffusion should occur much more rapidly than is observed experimentally. A smell produced in one corner of a room should be noticed instantly in the opposite corner, and it isn’t. (BB thought that this killed C’s theory). Cl ...
chapter 1
... Italy, invented the first thermometer, which used the volume expansion of alcohol when heated. Liquid thermometers use the extension of appropriate liquid placed in a thin capillary tube parallel to a scale, graduated in the selected temperature units. The higher the temperature, the greater is the ...
... Italy, invented the first thermometer, which used the volume expansion of alcohol when heated. Liquid thermometers use the extension of appropriate liquid placed in a thin capillary tube parallel to a scale, graduated in the selected temperature units. The higher the temperature, the greater is the ...
THERMODYNAMICS
... Similar to the distinction of a system and surroundings in thermodynamics, we can distinguish between the entropy of the system and the surroundings. In the example of the freezing water, ∆Ssys is the entropy change of water, and the ∆Ssurr is the entropy change of the surroundings. ...
... Similar to the distinction of a system and surroundings in thermodynamics, we can distinguish between the entropy of the system and the surroundings. In the example of the freezing water, ∆Ssys is the entropy change of water, and the ∆Ssurr is the entropy change of the surroundings. ...
On the definition of a kinetic equilibrium in global gyrokinetic
... we apply a modified algorithm based directly on the constancy of the full distribution function f along the (perturbed) orbits. This scheme has been called direct-δf [4]. Here, it is applied to toroidal configurations and in the Appendix we show that it is equivalent to the standard-δf scheme modifi ...
... we apply a modified algorithm based directly on the constancy of the full distribution function f along the (perturbed) orbits. This scheme has been called direct-δf [4]. Here, it is applied to toroidal configurations and in the Appendix we show that it is equivalent to the standard-δf scheme modifi ...
Outline Introduction Introduction Gibbs Free Energy
... • At all temperatures, the liquid has a higher internal energy U and enthalpy H as compared to the solid. Therefore Gl > Gs at low T. • The liquid phase, however, has a higher entropy S than the solid phase at all T. Therefore Gl decreases more rapidly with T as compared to Gs. • At Tm Gl(T) crosses ...
... • At all temperatures, the liquid has a higher internal energy U and enthalpy H as compared to the solid. Therefore Gl > Gs at low T. • The liquid phase, however, has a higher entropy S than the solid phase at all T. Therefore Gl decreases more rapidly with T as compared to Gs. • At Tm Gl(T) crosses ...
What is equilibrium?
... • The law of chemical equilibrium states that at a given temperature, a chemical system might reach a state in which a particular ratio of reactant and product concentrations has a constant value. ...
... • The law of chemical equilibrium states that at a given temperature, a chemical system might reach a state in which a particular ratio of reactant and product concentrations has a constant value. ...
Relativistic Thermodynamics, a Lagrangian Field Theory for general
... paper begins with a review of the phenomenological approach and presents an alternative. Thermodynamics, formulated as an action principle, is integrated with the Einstein-Hilbert action principle of General Relativity. The source of Einstein’s equation is the energy-momentum tensor of this relativi ...
... paper begins with a review of the phenomenological approach and presents an alternative. Thermodynamics, formulated as an action principle, is integrated with the Einstein-Hilbert action principle of General Relativity. The source of Einstein’s equation is the energy-momentum tensor of this relativi ...
Physics 1 Module 2: Thermodynamics
... • An ideal gas is defined as one in which all collisions between atoms or molecules are perfectly elastic, and in which there are no intermolecular attractive forces. One can visualize it as a collection of perfectly hard spheres which collide but which otherwise do not interact with each other. In ...
... • An ideal gas is defined as one in which all collisions between atoms or molecules are perfectly elastic, and in which there are no intermolecular attractive forces. One can visualize it as a collection of perfectly hard spheres which collide but which otherwise do not interact with each other. In ...
The Concentration Dependence of the
... For biochemical processes that occur at constant temperature and pressure, the directionality of a reaction is determined solely by the concentrations of the reactants and products that are present. Equation 2 contains explicit concentration terms and is therefore obviously concentrationdependent, w ...
... For biochemical processes that occur at constant temperature and pressure, the directionality of a reaction is determined solely by the concentrations of the reactants and products that are present. Equation 2 contains explicit concentration terms and is therefore obviously concentrationdependent, w ...
Chapter 17 - Groupfusion.net
... • The driving force for a spontaneous process is an increase in the entropy of the universe • Order (lower entropy) to disorder (higher entropy) • Thermodynamic function that describes the number of arrangements (positions and/or energy levels) available to a system in a given state. Associated with ...
... • The driving force for a spontaneous process is an increase in the entropy of the universe • Order (lower entropy) to disorder (higher entropy) • Thermodynamic function that describes the number of arrangements (positions and/or energy levels) available to a system in a given state. Associated with ...
Chapter 2
... By now, you may have noticed that several of the forms of energy we have defined overlap. For instance, on a microscopic level, thermal energy is really a combination of kinetic energy and some forms of potential energy. Chemical potential energy is often a significant portion of non-thermal interna ...
... By now, you may have noticed that several of the forms of energy we have defined overlap. For instance, on a microscopic level, thermal energy is really a combination of kinetic energy and some forms of potential energy. Chemical potential energy is often a significant portion of non-thermal interna ...
thermodynamics
... 29. When does gas absorb heat and does work during isothermal process? 30. When does gas release heat and work is done by the surrounding? 31. Mention the condition for work done by the gas in an adiabatic process. 32. Mention the condition for work done on the gas in an adiabatic process. 33. What ...
... 29. When does gas absorb heat and does work during isothermal process? 30. When does gas release heat and work is done by the surrounding? 31. Mention the condition for work done by the gas in an adiabatic process. 32. Mention the condition for work done on the gas in an adiabatic process. 33. What ...
Explaining organisational change - ANU Press
... of labour; a hierarchy of offices; a set of general rules that govern performance; a separation of personal from official property and rights; selection of personnel on the basis of technical qualifications; and employment viewed as a career by participants (Scott, 1998). From an engineering viewpoi ...
... of labour; a hierarchy of offices; a set of general rules that govern performance; a separation of personal from official property and rights; selection of personnel on the basis of technical qualifications; and employment viewed as a career by participants (Scott, 1998). From an engineering viewpoi ...
Chapter 2
... The surroundings are the rest of the universe that can in any significant way affect or be affected by the system. For example, if an ice cube is placed in a glass of water, we might take the ice to be the system and the water to be the surroundings. In this example we would usually ignore the inter ...
... The surroundings are the rest of the universe that can in any significant way affect or be affected by the system. For example, if an ice cube is placed in a glass of water, we might take the ice to be the system and the water to be the surroundings. In this example we would usually ignore the inter ...
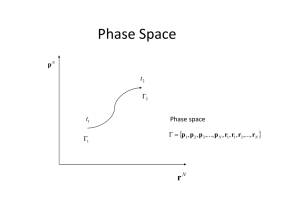
Phase Space Phase Space
... Within the statistical description, the motion of one single system with given initial conditions is not considered; thus, p(t), q(t) are not sought. Instead, the motion of a whole set of phase points, representing the collection of possible states of the given system. Such a set of phase points is ...
... Within the statistical description, the motion of one single system with given initial conditions is not considered; thus, p(t), q(t) are not sought. Instead, the motion of a whole set of phase points, representing the collection of possible states of the given system. Such a set of phase points is ...