Set 1 Answers
... 8. What defines a state function? Give an example. A state function is independent of path. In other words, it is a property of the system that is inherent and does not depend on how the system arrived at that state. For example, the internal energy inherent in a molecule (i.e. sum potential energy ...
... 8. What defines a state function? Give an example. A state function is independent of path. In other words, it is a property of the system that is inherent and does not depend on how the system arrived at that state. For example, the internal energy inherent in a molecule (i.e. sum potential energy ...
Entropy generation minimization of one and two
... pressure drop of the hydrogen. Control volumes CVref,1..N and CVhyd,1..N exchange only heat i.e no exchange of work or mass. The heat exchange rate relation is derived by applying a first law energy balance to CVtot . This is seen in (3). The needed U A value is determined using (4) which is derived ...
... pressure drop of the hydrogen. Control volumes CVref,1..N and CVhyd,1..N exchange only heat i.e no exchange of work or mass. The heat exchange rate relation is derived by applying a first law energy balance to CVtot . This is seen in (3). The needed U A value is determined using (4) which is derived ...
Download Pdf Article
... reproduced by all five equations of state. - equations SRK and PR can be used with the same temperature function with three parameters like the GEOS3C equation (eqs. 3-5). This fact leads to better values for vapor pressures but the values of saturated volumes can not be improved without a translati ...
... reproduced by all five equations of state. - equations SRK and PR can be used with the same temperature function with three parameters like the GEOS3C equation (eqs. 3-5). This fact leads to better values for vapor pressures but the values of saturated volumes can not be improved without a translati ...
File - Association of Chemical Engineering Students
... Points A and B on the P V diagram of Fig. 5.5 represent two equilibrium states of a particular fluid, and paths ACB and ADB show two arbitrary reversible processes connecting these points. Integration of Eq. (5.11) for each path gives: and ...
... Points A and B on the P V diagram of Fig. 5.5 represent two equilibrium states of a particular fluid, and paths ACB and ADB show two arbitrary reversible processes connecting these points. Integration of Eq. (5.11) for each path gives: and ...
Thermodynamics Notes
... We observe that energy flows spontaneously from one block (A) to the other (B). We say that block A has a higher temperature than block B. In fact, we say that the energy flow occurs because the blocks have different temperatures. We further observe that after the lapse of some time, called the rel ...
... We observe that energy flows spontaneously from one block (A) to the other (B). We say that block A has a higher temperature than block B. In fact, we say that the energy flow occurs because the blocks have different temperatures. We further observe that after the lapse of some time, called the rel ...
Chemical Thermodynamics
... The Molecular Interpretation of Entropy On a molecular level, we will see that an entropy increase is associated with an increase in the number of accessible microstates. Entropy change can also be thought of as a measure of a system's randomness at a stated temperature. The third law of thermodynam ...
... The Molecular Interpretation of Entropy On a molecular level, we will see that an entropy increase is associated with an increase in the number of accessible microstates. Entropy change can also be thought of as a measure of a system's randomness at a stated temperature. The third law of thermodynam ...
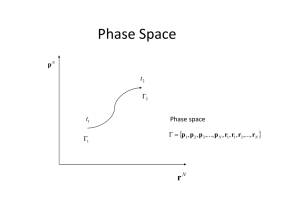
Phase Space Phase Space
... initial conditions is not considered; thus, p(t), q(t) are not sought. Instead, the motion of a whole set of phase points, representing the collection of possible states of the given system. Such a set of phase points is called a phase space ensemble. If each point in the phase space is considered a ...
... initial conditions is not considered; thus, p(t), q(t) are not sought. Instead, the motion of a whole set of phase points, representing the collection of possible states of the given system. Such a set of phase points is called a phase space ensemble. If each point in the phase space is considered a ...
PDF File - Tulane University
... Work, W, is defined as force x distance. Since Pressure, P, is defined as Force/surface area, Force = P x surface area, and thus W = P x surface area x distance = P x V, where V is volume. If the work is done at constant pressure, then W = PdV. Substitution of this relationship into (1) yields: dE = ...
... Work, W, is defined as force x distance. Since Pressure, P, is defined as Force/surface area, Force = P x surface area, and thus W = P x surface area x distance = P x V, where V is volume. If the work is done at constant pressure, then W = PdV. Substitution of this relationship into (1) yields: dE = ...
Fundamentals of Chemical Engineering Thermodynamics
... plants and refrigeration/liquefaction systems. This is the part of the course that most directly relates to processes discussed in capstone design and justifies the “Chemical Engineering” in the title of the book. It is one of the longer chapters, with several examples and end-of-chapter problems. Th ...
... plants and refrigeration/liquefaction systems. This is the part of the course that most directly relates to processes discussed in capstone design and justifies the “Chemical Engineering” in the title of the book. It is one of the longer chapters, with several examples and end-of-chapter problems. Th ...
Equilibrium Statistical Mechanics
... We begin with a fixed, adiabatic, impermeable piston with both sides in equilibrium and remove one or more constraints and ask what happens and how can we describe it. The following postulates are equivalent to the ususal statement of the second law of thermodynamics. I ...
... We begin with a fixed, adiabatic, impermeable piston with both sides in equilibrium and remove one or more constraints and ask what happens and how can we describe it. The following postulates are equivalent to the ususal statement of the second law of thermodynamics. I ...
chapter 4 general relationships between state variables of
... turned out to give a very simple relationship between .I and .W for the case of constant volume processes. However, this form of the equation is not as useful when we want to consider internal energy changes for constant pressure processes, or if we simply want to calculate the change in internal en ...
... turned out to give a very simple relationship between .I and .W for the case of constant volume processes. However, this form of the equation is not as useful when we want to consider internal energy changes for constant pressure processes, or if we simply want to calculate the change in internal en ...
Physical Chemistry of Semiconductor Materials and Processes Brochure
... electrical conductivity of ionic solids. Today, solid state technologies forms the background of the society in which we live. The aim of this book is threefold: first, to present the background physical chemistry on which the technology of solid state devices is based; secondly, to describe specifi ...
... electrical conductivity of ionic solids. Today, solid state technologies forms the background of the society in which we live. The aim of this book is threefold: first, to present the background physical chemistry on which the technology of solid state devices is based; secondly, to describe specifi ...
2. THERMODYNAMICS and ENSEMBLES (Part A) Introduction
... dice initially, as well as their velocities at the instant they are thrown, it is possible to predict the outcome by solving the equation of motion. However, such detailed knowledge is not available and then we start talking about the probability of outcome of a particular event. The theoretical aim ...
... dice initially, as well as their velocities at the instant they are thrown, it is possible to predict the outcome by solving the equation of motion. However, such detailed knowledge is not available and then we start talking about the probability of outcome of a particular event. The theoretical aim ...
Chapter 2 Buoyancy and Coriolis forces
... In this chapter we address several topics that we need to understand before starting on our study of geophysical uid dynamics. ...
... In this chapter we address several topics that we need to understand before starting on our study of geophysical uid dynamics. ...
Review of Chemical Thermodynamics 7.51 September 1999 ∆G
... energy of this process when the reaction proceeds without changing the concentrations of reactants and products. For the reaction, A+B ⇔ AB, imagine that we have a solution containing 1 M [A], 1 M [B], and 1 M [AB]. Now allow 1 molecule of A to bind 1 molecule of B to form a new AB complex. Clearly ...
... energy of this process when the reaction proceeds without changing the concentrations of reactants and products. For the reaction, A+B ⇔ AB, imagine that we have a solution containing 1 M [A], 1 M [B], and 1 M [AB]. Now allow 1 molecule of A to bind 1 molecule of B to form a new AB complex. Clearly ...
Introduction to Physical Biochemistry
... details of the mathematical manipulations. The mathematics used is not simply ...
... details of the mathematical manipulations. The mathematics used is not simply ...
Review of fundamental principles ? Thermodynamics : Part II
... Also, from Eq. (5.1b), T ds=dh - vdP , hence for a constant pressure process (dP = 0), therefore, for a constant pressure process Tds = dh, which means that for an isobaric process the area under the curve is equal to change in enthalpy on T-s diagram. Properties at Saturation The properties of refr ...
... Also, from Eq. (5.1b), T ds=dh - vdP , hence for a constant pressure process (dP = 0), therefore, for a constant pressure process Tds = dh, which means that for an isobaric process the area under the curve is equal to change in enthalpy on T-s diagram. Properties at Saturation The properties of refr ...
Entropy in thermodynamics and information theory
There are close parallels between the mathematical expressions for the thermodynamic entropy, usually denoted by S, of a physical system in the statistical thermodynamics established by Ludwig Boltzmann and J. Willard Gibbs in the 1870s, and the information-theoretic entropy, usually expressed as H, of Claude Shannon and Ralph Hartley developed in the 1940s. Shannon, although not initially aware of this similarity, commented on it upon publicizing information theory in A Mathematical Theory of Communication.This article explores what links there are between the two concepts, and how far they can be regarded as connected.