CRYOGENICS
... Absolute zero is a temperature marked by a 0 entropy configuration. It is the coldest temperature theoretically possible and cannot be reached by artificial or natural means. Temperature is an entropically defined quantity that effectively determines the number of thermodynamically accessible states ...
... Absolute zero is a temperature marked by a 0 entropy configuration. It is the coldest temperature theoretically possible and cannot be reached by artificial or natural means. Temperature is an entropically defined quantity that effectively determines the number of thermodynamically accessible states ...
Document
... The critical current density and the critical magnetic field The supercurrent density has a limit: JC When the superconductor is applied with a magnetic field, a supercurrent is generated so as to maintain the perfect diamagnetism. If the current density needed to screen the field exceeds JC, the s ...
... The critical current density and the critical magnetic field The supercurrent density has a limit: JC When the superconductor is applied with a magnetic field, a supercurrent is generated so as to maintain the perfect diamagnetism. If the current density needed to screen the field exceeds JC, the s ...
state of matter - Mayfield City Schools
... energy. In general, the warmer an object, the more kinetic energy its atoms and molecules possess. - Temperature, the degree of “hotness” or “coldness” of an object, is proportional to the average (NOT total) kinetic energy of the atoms or molecules making it up. ...
... energy. In general, the warmer an object, the more kinetic energy its atoms and molecules possess. - Temperature, the degree of “hotness” or “coldness” of an object, is proportional to the average (NOT total) kinetic energy of the atoms or molecules making it up. ...
BCJ0205-15 Thermal phenomena (3-1-4)
... Week 1: Thermal equilibrium. Temperature and the zeroth law of thermodynamics. Scales and measures of temperature. Thermal expansion. Week 2: Heat, internal energy, heat capacity and specific heat. Latent heat. Week 3: Mechanisms for heat transfer. Week 4: Heat and work in thermodynamic processes. P ...
... Week 1: Thermal equilibrium. Temperature and the zeroth law of thermodynamics. Scales and measures of temperature. Thermal expansion. Week 2: Heat, internal energy, heat capacity and specific heat. Latent heat. Week 3: Mechanisms for heat transfer. Week 4: Heat and work in thermodynamic processes. P ...
CHE 301 Problem set #3
... CHE 301 Problem set #8 1. An experimental arrangement for measuring the thermal conductivity of solid materials involves the use of two long rods that are equivalent in every respect, except that one is fabricated from a standard material of known thermal conductivity kA while the other is fabricate ...
... CHE 301 Problem set #8 1. An experimental arrangement for measuring the thermal conductivity of solid materials involves the use of two long rods that are equivalent in every respect, except that one is fabricated from a standard material of known thermal conductivity kA while the other is fabricate ...
Thermodynamics - StrikerPhysics
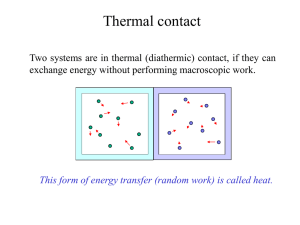
... • We use macroscopic means for analysis of these systems of many particles - involving quantities such as pressure, volume and temperature. ...
... • We use macroscopic means for analysis of these systems of many particles - involving quantities such as pressure, volume and temperature. ...
system
... Microscopic state of the system depends on the masses, velocities, positions, and all modes of motion of all of the constituent particles In the absence of detailed knowledge, thermodynamics considers the properties of the system which, when determined, define the macroscopic state of the system Sti ...
... Microscopic state of the system depends on the masses, velocities, positions, and all modes of motion of all of the constituent particles In the absence of detailed knowledge, thermodynamics considers the properties of the system which, when determined, define the macroscopic state of the system Sti ...
B Day Classes-I am sorry I am not here to give you the
... a liquid. The point this occurs is called Melting Point. Once melted, there are no forces holding the particles in the solid state. As the energy continues to increase, the motion increases and therefore the temperature increases. Some particles begin to break free from other particles. This point i ...
... a liquid. The point this occurs is called Melting Point. Once melted, there are no forces holding the particles in the solid state. As the energy continues to increase, the motion increases and therefore the temperature increases. Some particles begin to break free from other particles. This point i ...
Atomic Structure
... 1. Consider the human body as a system and apply the first law of thermodynamics to it. We know that over any given period of sufficient length (say one day), there will be a net heat flow from the body (i.e. Q is negative) and the body will do some external work on its surroundings (i.e. W is posit ...
... 1. Consider the human body as a system and apply the first law of thermodynamics to it. We know that over any given period of sufficient length (say one day), there will be a net heat flow from the body (i.e. Q is negative) and the body will do some external work on its surroundings (i.e. W is posit ...
Kelvin scale
... abbreviation) each month. This list, which began January 1999 with the letter a, is alphabetical but uses successive letters for each month’s entry. (We will return to the letter a after twenty-six months.) Word of the Month for November 1999 kelvin (K) The SI (Système International or International ...
... abbreviation) each month. This list, which began January 1999 with the letter a, is alphabetical but uses successive letters for each month’s entry. (We will return to the letter a after twenty-six months.) Word of the Month for November 1999 kelvin (K) The SI (Système International or International ...