2.3 ThermoChemistry - Chemistry Teaching Resources
... The standard molar enthalpy change of lattice formation is the enthalpy change which occurs when one mole of an ionic crystal is formed from the ions in their gaseous states under standard conditions. ...
... The standard molar enthalpy change of lattice formation is the enthalpy change which occurs when one mole of an ionic crystal is formed from the ions in their gaseous states under standard conditions. ...
Chapter 12
... of the borate anion in borax is shown in Structure 5-XXVII. Borax occurs in large deposits in the Mojave desert of California, which is the major source of boron. o ionic compounds involving simple B3+ cations are formed because the ionization enthalpies for boron are so high that lattice energies o ...
... of the borate anion in borax is shown in Structure 5-XXVII. Borax occurs in large deposits in the Mojave desert of California, which is the major source of boron. o ionic compounds involving simple B3+ cations are formed because the ionization enthalpies for boron are so high that lattice energies o ...
Complexometric Titration
... Molecule or ion with at least 1 pair of unshared electron can form covalent bond with metal ion = ligands The bonding between metal and ligand generally involves formal donation of one or more of the ligand's electron pairs Eg of ligands = ammonia, cyanide ions, halide ions, water (neutral/-ve charg ...
... Molecule or ion with at least 1 pair of unshared electron can form covalent bond with metal ion = ligands The bonding between metal and ligand generally involves formal donation of one or more of the ligand's electron pairs Eg of ligands = ammonia, cyanide ions, halide ions, water (neutral/-ve charg ...
Refraction and Optical Fibres
... Education, Science and Training as a part of the Boosting Innovation in Science, Technology and Mathematics Teaching (BISTMT) Programme.” ...
... Education, Science and Training as a part of the Boosting Innovation in Science, Technology and Mathematics Teaching (BISTMT) Programme.” ...
Title The Separation mechanism of Am(III) from Eu(III) by
... and [M(HMNTA)(H2O)5]3+ complexes. The [M(TMDGA)3] geometry displayed a pseudo tricapped trigonal structure with the ether oxygen of TMDGA as a cap and the C3 rotational axis along the perpendicular direction toward the plane, which included three oxygens of ether for both Δ and Λ conformers. The [M( ...
... and [M(HMNTA)(H2O)5]3+ complexes. The [M(TMDGA)3] geometry displayed a pseudo tricapped trigonal structure with the ether oxygen of TMDGA as a cap and the C3 rotational axis along the perpendicular direction toward the plane, which included three oxygens of ether for both Δ and Λ conformers. The [M( ...
Solubility Workbook
... this class?” If you are determined to be successful the minimum expectation that you should have for yourself is that you do all of these questions by the due dates given by your teacher. There are other things that you should do to prepare for the test at the end of the unit. Remember, what you put ...
... this class?” If you are determined to be successful the minimum expectation that you should have for yourself is that you do all of these questions by the due dates given by your teacher. There are other things that you should do to prepare for the test at the end of the unit. Remember, what you put ...
2015 Unit1 Part3.2 Equilibrium2
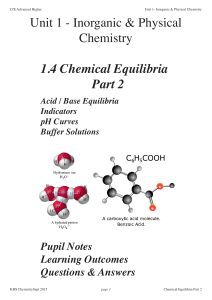
... A buffer solution is one in which the pH of the solution remains approximately constant when small amounts of acid or base are added or the solution is diluted with water. An acidic buffer consists of a solution of a weak acid and one of its salts with a strong alkali. A basic buffer consists of a s ...
... A buffer solution is one in which the pH of the solution remains approximately constant when small amounts of acid or base are added or the solution is diluted with water. An acidic buffer consists of a solution of a weak acid and one of its salts with a strong alkali. A basic buffer consists of a s ...
Theory of Ion Exchange
... (eqn [16]), the rate of exchange increases (t1/2 decreases) as the particle radius decreases, being proportional to 1/r20. For Rlm diffusion (eqn [17]), the rate increases less strongly as r0 is decreased (the proportionality is to 1/r0). In Rlm diffusion the exchange rate can also be increased by i ...
... (eqn [16]), the rate of exchange increases (t1/2 decreases) as the particle radius decreases, being proportional to 1/r20. For Rlm diffusion (eqn [17]), the rate increases less strongly as r0 is decreased (the proportionality is to 1/r0). In Rlm diffusion the exchange rate can also be increased by i ...
Mathematical Models of Ionic Flow Through Open Protein Channels
... densities (C-PNP). While derivations of continuum equations from equilibrium molecular models are common in the statistical physics literature, this is the first derivation of Poisson Nernst Planck type equations from a non equilibrium molecular model. The resulting equations differ in many aspects ...
... densities (C-PNP). While derivations of continuum equations from equilibrium molecular models are common in the statistical physics literature, this is the first derivation of Poisson Nernst Planck type equations from a non equilibrium molecular model. The resulting equations differ in many aspects ...
Herbert Ipser, Olga P. Semenova, Regina
... does not melt congruently and we were not able to find suitable single crystals. However, we succeeded to prepare the pure ternary compound in order to perform X-ray powder diffraction using a Guinier image plate technique. The entire diffraction spectrum was refined by Full profile Rietveld method ...
... does not melt congruently and we were not able to find suitable single crystals. However, we succeeded to prepare the pure ternary compound in order to perform X-ray powder diffraction using a Guinier image plate technique. The entire diffraction spectrum was refined by Full profile Rietveld method ...
1.09 MB / 64 pages
... dissolved in water, does not conduct an electric current, so does not contain ions. The identity of the ions in solution A cannot be determined from these data. Problem 2.20. (a) Barium chloride, BaCl2(s), dissolves in water to give barium cations, Ba2+(aq), and chloride anions, Cl–(aq). (b) Potassi ...
... dissolved in water, does not conduct an electric current, so does not contain ions. The identity of the ions in solution A cannot be determined from these data. Problem 2.20. (a) Barium chloride, BaCl2(s), dissolves in water to give barium cations, Ba2+(aq), and chloride anions, Cl–(aq). (b) Potassi ...
The Inhibition of Carbon Steel Corrosion in Hydrochloric Acid
... MXads refer to the halide ion and ion – pair respectively in the adsorbed state. This ion – pair interaction increases the surface coverage thereby reducing metal dissolution. The order of decreasing inhibition efficiency of the investigated compounds on addition of a specific concentration of the u ...
... MXads refer to the halide ion and ion – pair respectively in the adsorbed state. This ion – pair interaction increases the surface coverage thereby reducing metal dissolution. The order of decreasing inhibition efficiency of the investigated compounds on addition of a specific concentration of the u ...
Solutions and solubility
... decreased If, for example, one compares the effect of equivalent amount of lithium chloride, sodium chloride, potassium chloride, rubidium chloride and cesium chloride (all of which belong to the family of alkali metals and are of the same valence type), it is observed that, lithium chloride decreas ...
... decreased If, for example, one compares the effect of equivalent amount of lithium chloride, sodium chloride, potassium chloride, rubidium chloride and cesium chloride (all of which belong to the family of alkali metals and are of the same valence type), it is observed that, lithium chloride decreas ...
Cold interactions between an Yb ion and a Li atom
... coupling V so (R) which has the same form and a similar effect as the spin-spin coupling V ss (R) [45,46]. The bound rovibrational levels are calculated by diagonalizing the nuclear Hamiltonian represented on a Fourier grid with adaptive step size [47–49]. The total scattering wave function is const ...
... coupling V so (R) which has the same form and a similar effect as the spin-spin coupling V ss (R) [45,46]. The bound rovibrational levels are calculated by diagonalizing the nuclear Hamiltonian represented on a Fourier grid with adaptive step size [47–49]. The total scattering wave function is const ...
Answer Key
... Brass (mixture of Zn and Cu); solution of solid Zn dissolved in solid Cu F. Milk; colloid of intermediate size, solid fat particles dispersed in liquid water. G. Paint; suspension of large solid TiO2 particles dispersed in liquid water or oil H. Italian dressing; not a homogeneous mixture at all, re ...
... Brass (mixture of Zn and Cu); solution of solid Zn dissolved in solid Cu F. Milk; colloid of intermediate size, solid fat particles dispersed in liquid water. G. Paint; suspension of large solid TiO2 particles dispersed in liquid water or oil H. Italian dressing; not a homogeneous mixture at all, re ...
Solutions
... where all particles exist as individual molecules or ions. • Mixtures in chemistry are combinations of different substances where each substance retains its chemical properties. ...
... where all particles exist as individual molecules or ions. • Mixtures in chemistry are combinations of different substances where each substance retains its chemical properties. ...
SUPERCRITICAL AQUEOUS SOLUTIONS OF SODIUM CHLORIDE
... preferentially formed at lower densities. Also, a materials science application is presented where the rapid quenching of hydrothermally formed sodium chloride clusters leads to a variety of nanostructures, ii ...
... preferentially formed at lower densities. Also, a materials science application is presented where the rapid quenching of hydrothermally formed sodium chloride clusters leads to a variety of nanostructures, ii ...
Full-Text PDF
... Potassium dichromate is a readily available and inexpensive reagent. The catalytic effects of several Lewis acids upon the activity of this oxidant were thoroughly studied. For this purpose the oxidation of benzhydrol to benzophenone was investigated in the presence of AlCl3, FeCl3, BiCl3, NiCl2, Ce ...
... Potassium dichromate is a readily available and inexpensive reagent. The catalytic effects of several Lewis acids upon the activity of this oxidant were thoroughly studied. For this purpose the oxidation of benzhydrol to benzophenone was investigated in the presence of AlCl3, FeCl3, BiCl3, NiCl2, Ce ...
Presentation 3
... Mg2+(aq) + SO42-(aq) = MgSO4(aq) Given that –COO- tends to form water bridges to soil minerals via divalent cations adsorbed onto mineral surfaces, is the MgSO4 complex inner- or outer-sphere? ...
... Mg2+(aq) + SO42-(aq) = MgSO4(aq) Given that –COO- tends to form water bridges to soil minerals via divalent cations adsorbed onto mineral surfaces, is the MgSO4 complex inner- or outer-sphere? ...
The Degradation of Cellulose with Ferric and Cupric Ions in a Low
... • Iron ions act as a catalyst for the cleavage of cellulose 1-4-β-glucosidic bond, whereas copper ions catalyst the oxidation on the anhydroglucose ring. The Lewis mechanism (Fig. 5), until now only postulated, is therefore experimentally verified. • Measurements of average "cold" and "hot" degree o ...
... • Iron ions act as a catalyst for the cleavage of cellulose 1-4-β-glucosidic bond, whereas copper ions catalyst the oxidation on the anhydroglucose ring. The Lewis mechanism (Fig. 5), until now only postulated, is therefore experimentally verified. • Measurements of average "cold" and "hot" degree o ...
Grossmont College Chemistry 120 Laboratory Manual 6th Edition
... safety goggles. Contact lenses should not be worn in the laboratory, even under safety goggles. By themselves they offer no protection from splashes and they are considered unsafe even under safety goggles because various fumes for instance, hydrogen chloride gas-may accumulate under the lens and ca ...
... safety goggles. Contact lenses should not be worn in the laboratory, even under safety goggles. By themselves they offer no protection from splashes and they are considered unsafe even under safety goggles because various fumes for instance, hydrogen chloride gas-may accumulate under the lens and ca ...
Part II - American Chemical Society
... omit formulas for all ions or molecules that do not take part in a reaction. Write structural formulas for all organic substances. You need not balance the equations. All reactions occur in aqueous solution unless otherwise indicated. a. Excess carbon dioxide is bubbled through a suspension of calci ...
... omit formulas for all ions or molecules that do not take part in a reaction. Write structural formulas for all organic substances. You need not balance the equations. All reactions occur in aqueous solution unless otherwise indicated. a. Excess carbon dioxide is bubbled through a suspension of calci ...
History, Introduction, and Kinetics of Ion Exchange Materials
... are exchanged for different ions of similar charge in solution. The exchanger must have an open network structure, either organic or inorganic, which carries the ions and which allows ions to pass through it. An ion exchanger is a water-insoluble substance which can exchange some of its ions for sim ...
... are exchanged for different ions of similar charge in solution. The exchanger must have an open network structure, either organic or inorganic, which carries the ions and which allows ions to pass through it. An ion exchanger is a water-insoluble substance which can exchange some of its ions for sim ...
Correlated/non-correlated ion dynamics of charge
... The title salt, 3-butyl-1-methyl-1H-imidazolium hexafluorophosphate, [C4mim][PF6] (see Fig. 1), has to date received much attention in the arena of molecular dynamics (MD), usually employing static high field approaches that probe fast motions.9–11 Our interest in this particular salt, which possess ...
... The title salt, 3-butyl-1-methyl-1H-imidazolium hexafluorophosphate, [C4mim][PF6] (see Fig. 1), has to date received much attention in the arena of molecular dynamics (MD), usually employing static high field approaches that probe fast motions.9–11 Our interest in this particular salt, which possess ...
Part II - American Chemical Society
... c. Two reactions, A and B, have rate constants that are equal at 25˚C but the rate constant for reaction A is much greater than that for reaction B at 35˚C. d. The rates of reactions catalyzed by complex molecules, such as enzymes, increase with an increase in temperature up to a certain point above ...
... c. Two reactions, A and B, have rate constants that are equal at 25˚C but the rate constant for reaction A is much greater than that for reaction B at 35˚C. d. The rates of reactions catalyzed by complex molecules, such as enzymes, increase with an increase in temperature up to a certain point above ...
Ionic compound

In chemistry, an ionic compound is a chemical compound in which ions are held together in a structure by electrostatic forces termed ionic bonds. The positively charged ions are called cations and the negatively charged ions are called anions. These can be simple ions such as the sodium (Na+) and chloride (Cl−) in sodium chloride, or polyatomic species such as the carbonate ion (CO32−) in calcium carbonate. Individual ions within an ionic compound usually have multiple nearest neighbours, so are not considered to be part of molecules, but instead part of a continuous three-dimensional network, usually in a crystalline structure.Ionic compounds typically have high melting and boiling points, and are hard and brittle. As solids they are almost always electrically insulating, but when melted or dissolved they become highly conductive, because the ions are mobilized.Ionic compounds without the acidic hydrogen ion (H+), or the basic ions hydroxide (OH−) or oxide (O2−), are also known as salts and can be formed by acid-base reactions. Ionic compounds containing hydrogen ions are classified as acids and compounds containing hydroxide or oxide ions are classified as bases.