classification of elements and periodicity in properties
... group 1 elements (hydrogen, alkali metals). Elements with two electrons in their outer shells have the configuration of ns2. They belong to group 2 elements (alkaline earth metals). Elements with three electrons (two in ‘s’ orbital and one in ‘p’ orbital) have the configuration of ns2 np2. They belo ...
... group 1 elements (hydrogen, alkali metals). Elements with two electrons in their outer shells have the configuration of ns2. They belong to group 2 elements (alkaline earth metals). Elements with three electrons (two in ‘s’ orbital and one in ‘p’ orbital) have the configuration of ns2 np2. They belo ...
7.2 | Effective Nuclear Charge
... characteristics be listed in the same column forced him to leave blank spaces in his table. For example, both gallium (Ga) and germanium (Ge) were unknown to Mendeleev. He boldly predicted their existence and properties, referring to them as eka-aluminum (“under” aluminum) and eka-silicon (“under” s ...
... characteristics be listed in the same column forced him to leave blank spaces in his table. For example, both gallium (Ga) and germanium (Ge) were unknown to Mendeleev. He boldly predicted their existence and properties, referring to them as eka-aluminum (“under” aluminum) and eka-silicon (“under” s ...
The Periodic Table
... • Photoelectron Spectroscopy (PES) is a technique that is used to gather information about the electrons in an atom. • An atom is bombarded with photons. Some of the photons are absorbed and electrons are emitted. The energy is analyzed. Since we can know the energy of the photons, and we know that ...
... • Photoelectron Spectroscopy (PES) is a technique that is used to gather information about the electrons in an atom. • An atom is bombarded with photons. Some of the photons are absorbed and electrons are emitted. The energy is analyzed. Since we can know the energy of the photons, and we know that ...
94 Lecture Notes 5th Series: Inorganic Chemistry THE MAIN
... practicing chemist. So what chance do you have of understanding inorganic chemistry? Well, believe it or not, you have a very good chance. If we use the periodic table to its full potential then we can predict the chemistry of obscure elements such as antimony (Sb, atomic number 51) or thallium (Tl, ...
... practicing chemist. So what chance do you have of understanding inorganic chemistry? Well, believe it or not, you have a very good chance. If we use the periodic table to its full potential then we can predict the chemistry of obscure elements such as antimony (Sb, atomic number 51) or thallium (Tl, ...
Chapter 7 Electron Configuration and the
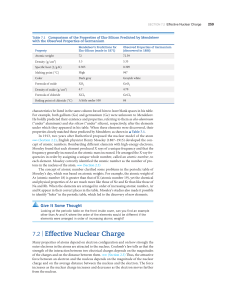
... • Z (nuclear charge) = the number of protons in the nucleus of an atom • Zeff (effective nuclear charge) = the magnitude of positive charge “experienced” by an electron in the atom • Zeff increases from left to right across a period; changes very little down a column Copyright McGraw-Hill 2009 ...
... • Z (nuclear charge) = the number of protons in the nucleus of an atom • Zeff (effective nuclear charge) = the magnitude of positive charge “experienced” by an electron in the atom • Zeff increases from left to right across a period; changes very little down a column Copyright McGraw-Hill 2009 ...
Quantum Thorj and th Atom - Crossroads chemistry 40S
... electron can move only from one allowable orbit to another, and therefore, can emit or absorb only certain amounts of energy. I The four electron transitions that account for visible lines in hydrogen’s atomic emission spectrum are shown in Figure 5-lOb. For example, electrons dropping from the thir ...
... electron can move only from one allowable orbit to another, and therefore, can emit or absorb only certain amounts of energy. I The four electron transitions that account for visible lines in hydrogen’s atomic emission spectrum are shown in Figure 5-lOb. For example, electrons dropping from the thir ...
Primeasia University
... Paulis Exclusion Principle: This principle states that “It is impossible that two electron in a given atom to have all the four quantum numbers identical”. i.e. In an atom two electrons can have maximum three quantum numbers (n, l, m) the same value and the fourth (s) will definitely be having a d ...
... Paulis Exclusion Principle: This principle states that “It is impossible that two electron in a given atom to have all the four quantum numbers identical”. i.e. In an atom two electrons can have maximum three quantum numbers (n, l, m) the same value and the fourth (s) will definitely be having a d ...
Periodic Trends Superblock
... The metallic elements are grouped on the left side of the periodic table. Metals are elements that have a high luster when clean and a high electrical conductivity. They are ductile (can be drawn into wire) and malleable (can be beaten into sheets). Most of the elements are metals. They include the ...
... The metallic elements are grouped on the left side of the periodic table. Metals are elements that have a high luster when clean and a high electrical conductivity. They are ductile (can be drawn into wire) and malleable (can be beaten into sheets). Most of the elements are metals. They include the ...
Atoms, Nuclear Decay, Electronic Structure, and Atomic Chemical
... The quantum number m l is called the magnetic quantum number. It describes the orientation of the orbital about an x, y, z coordinate system. Each possible orientation can hold up to 2 electrons maximum. For each orbital, there are 2l + 1 different orientations. The quantum number ml is all integer ...
... The quantum number m l is called the magnetic quantum number. It describes the orientation of the orbital about an x, y, z coordinate system. Each possible orientation can hold up to 2 electrons maximum. For each orbital, there are 2l + 1 different orientations. The quantum number ml is all integer ...
Part 9 - istcp
... Group 2A: The Alkaline Earth Metals Alkaline earth metals are harder and more dense than the alkali metals. The chemistry is dominated by the loss of two s electrons: M M2+ + 2e-. Mg(s) + Cl2(g) MgCl2(s) 2Mg(s) + O2(g) 2MgO(s) Be does not react with water. Mg will only react with steam. Ca onw ...
... Group 2A: The Alkaline Earth Metals Alkaline earth metals are harder and more dense than the alkali metals. The chemistry is dominated by the loss of two s electrons: M M2+ + 2e-. Mg(s) + Cl2(g) MgCl2(s) 2Mg(s) + O2(g) 2MgO(s) Be does not react with water. Mg will only react with steam. Ca onw ...
Chapter 6: The Periodic Table and Periodic Law
... History of the Periodic Table’s Development In the late 1790s, French scientist Antoine Lavoisier compiled a list of elements known at the time. The list contained 23 elements. Many of these elements, such as silver, gold, carbon, and oxygen, were known since prehistoric times. The 1800s brought man ...
... History of the Periodic Table’s Development In the late 1790s, French scientist Antoine Lavoisier compiled a list of elements known at the time. The list contained 23 elements. Many of these elements, such as silver, gold, carbon, and oxygen, were known since prehistoric times. The 1800s brought man ...
Chemical Practice
... It is equally easy for elements with 4 valence electrons to lose or gain 4 electrons so depending on what happens, they can have either a positive or negative charge. 10. To balance out the oxidation #’s for a +1 and -2 bond, there needs to be 2 (+1) for every 1 (-2). To balance out the oxidation #’ ...
... It is equally easy for elements with 4 valence electrons to lose or gain 4 electrons so depending on what happens, they can have either a positive or negative charge. 10. To balance out the oxidation #’s for a +1 and -2 bond, there needs to be 2 (+1) for every 1 (-2). To balance out the oxidation #’ ...
Document
... Humans and their ancestors have used stone tools made from brittle minerals for about 2.5 million years. The shattering of brittle minerals such as flint produced sharp edges useful for cutting and weaponry. Sometimes tools could be repaired by breaking a new cutting edge but the brittleness often m ...
... Humans and their ancestors have used stone tools made from brittle minerals for about 2.5 million years. The shattering of brittle minerals such as flint produced sharp edges useful for cutting and weaponry. Sometimes tools could be repaired by breaking a new cutting edge but the brittleness often m ...
Electrons and Periodic Trends PowerPoint
... f-block: Lanthanides & Actinides • Elements in the period 6 of the f-block are called lanthanides (or rare-earth). • Lanthanides are shiny metals similar in reactivity to alkaline earth metals. • Elements in period 7 of the f-block are called actinides. • Actinides are all radioactive, and many of t ...
... f-block: Lanthanides & Actinides • Elements in the period 6 of the f-block are called lanthanides (or rare-earth). • Lanthanides are shiny metals similar in reactivity to alkaline earth metals. • Elements in period 7 of the f-block are called actinides. • Actinides are all radioactive, and many of t ...
Unit_2_Electrons_and_Periodic_Trends
... f-block: Lanthanides & Actinides • Elements in the period 6 of the f-block are called lanthanides (or rare-earth). • Lanthanides are shiny metals similar in reactivity to alkaline earth metals. • Elements in period 7 of the f-block are called actinides. • Actinides are all radioactive, and many of t ...
... f-block: Lanthanides & Actinides • Elements in the period 6 of the f-block are called lanthanides (or rare-earth). • Lanthanides are shiny metals similar in reactivity to alkaline earth metals. • Elements in period 7 of the f-block are called actinides. • Actinides are all radioactive, and many of t ...
Introduction to Atomic Structure - California K
... table) are most stable when they contain eight electrons in their valence shell. He suggested that atoms with fewer than eight valence electrons bond together to share electrons and complete their valence shells. Remember that noble gases (or inert gases) are generally not reactive and exist as stab ...
... table) are most stable when they contain eight electrons in their valence shell. He suggested that atoms with fewer than eight valence electrons bond together to share electrons and complete their valence shells. Remember that noble gases (or inert gases) are generally not reactive and exist as stab ...
Unit 1 Module 3 The Periodic Table
... existence to the chemist. Pharmaceuticals, hair products, soap and cosmetics have all been made in chemical reactions using methods devised by research chemists. These products have then been tested in the laboratory by quality control chemists before being put on sale. Wherever you look, you can se ...
... existence to the chemist. Pharmaceuticals, hair products, soap and cosmetics have all been made in chemical reactions using methods devised by research chemists. These products have then been tested in the laboratory by quality control chemists before being put on sale. Wherever you look, you can se ...
1.3 Explaining Periodic Trends
... electronegativity. Describe their relationship with each other in the periodic table. ...
... electronegativity. Describe their relationship with each other in the periodic table. ...
Worked solutions
... 4 C Germanium, it has properties of both a metal (e.g. forms alloys with other metals) and a non-metal (e.g. brittle). Calcium, manganese and magnesium are all metals. 5 B Graphite, as it has a network of delocalized electrons throughout its structure. 6 C Elements are arranged in the Perio ...
... 4 C Germanium, it has properties of both a metal (e.g. forms alloys with other metals) and a non-metal (e.g. brittle). Calcium, manganese and magnesium are all metals. 5 B Graphite, as it has a network of delocalized electrons throughout its structure. 6 C Elements are arranged in the Perio ...
Chemistry
... missing at the top. It's not organized alphabetically so elements can be found easily. But to a chemist, the periodic table is a very powerful tool. The periodic table is organized by properties, both chemical and physical. Those properties relate to the electronic structure of the atoms of each ele ...
... missing at the top. It's not organized alphabetically so elements can be found easily. But to a chemist, the periodic table is a very powerful tool. The periodic table is organized by properties, both chemical and physical. Those properties relate to the electronic structure of the atoms of each ele ...
Electrons and Periodicity
... Where are the nonmetals located on the periodic table? Where are the metalloids listed on the periodic table? Classify each of the following elements as metals, nonmetals, or metalloids. ...
... Where are the nonmetals located on the periodic table? Where are the metalloids listed on the periodic table? Classify each of the following elements as metals, nonmetals, or metalloids. ...
Usefulness of the periodic table in studying the chemistry of elements:
... The Group 18 elements are normally called the noble gases and they include helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe) and radon (Rn). They are called noble gases because they are chemically not very reactive. They are also called rare gases as they are found only in very small quan ...
... The Group 18 elements are normally called the noble gases and they include helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe) and radon (Rn). They are called noble gases because they are chemically not very reactive. They are also called rare gases as they are found only in very small quan ...
Periods and Blocks of the Periodic Table
... • Group 2 metals are less reactive than the alkali metals, but are still too reactive to be found in nature in pure form. Chapter menu ...
... • Group 2 metals are less reactive than the alkali metals, but are still too reactive to be found in nature in pure form. Chapter menu ...