Prions Gone Mad - MSOE Center for BioMolecular Modeling
... associated with the [Het-s] prion. Currently, the [Het-s] prion is of interest because it is thought to have a structure similar to that of the human Prp prion. ...
... associated with the [Het-s] prion. Currently, the [Het-s] prion is of interest because it is thought to have a structure similar to that of the human Prp prion. ...
What do Prions, Viruses and Viroids reveal about the formation of
... between proteins and nucleic acids that led to the first cells. In 2002, for the first time an infective virus has been synthesized from pure chemicals (1). The properties of self-assembly would be key in the formation of putative prebiotic DNA, protein constructs. The finding that viruses possibly ...
... between proteins and nucleic acids that led to the first cells. In 2002, for the first time an infective virus has been synthesized from pure chemicals (1). The properties of self-assembly would be key in the formation of putative prebiotic DNA, protein constructs. The finding that viruses possibly ...
Jacob Corn
... Transmissible Spongiform Encephalopathies, or TSEs (Prusiner, 1995). Prion diseases are incurable and ultimately fatal, with death occurring anywhere between 300 days to decades after infection. Surprisingly, prion diseases do not fit into any class of currently known pathogens. The presence of fore ...
... Transmissible Spongiform Encephalopathies, or TSEs (Prusiner, 1995). Prion diseases are incurable and ultimately fatal, with death occurring anywhere between 300 days to decades after infection. Surprisingly, prion diseases do not fit into any class of currently known pathogens. The presence of fore ...
PSI+
... mAbs T1 and T2 were established by immunizing PrP gene ablated mice with recombinant MoPrP of residues 121–231. Both mAbs were cross-reactive with PrP from hamster, sheep, cattle and deer. A linear epitope of mAb T1 was identified at residues 137–143 of MoPrP and buried in PrPC expressed on the cell ...
... mAbs T1 and T2 were established by immunizing PrP gene ablated mice with recombinant MoPrP of residues 121–231. Both mAbs were cross-reactive with PrP from hamster, sheep, cattle and deer. A linear epitope of mAb T1 was identified at residues 137–143 of MoPrP and buried in PrPC expressed on the cell ...
Ellen Sebastian - Fatal Familial Insomnia
... There are two Conformational isoforms of the mammalian prion protein: – PrPc, the normal cellular isoform – PrPSc, the ‘scrapie’ isoform – The conversion of PrPC to PrPsc causes prion diseases ...
... There are two Conformational isoforms of the mammalian prion protein: – PrPc, the normal cellular isoform – PrPSc, the ‘scrapie’ isoform – The conversion of PrPC to PrPsc causes prion diseases ...
Protein Structure
... Provides a “safe folding environment” — Probably binds to polypeptide, to induce the correct folding conformation ...
... Provides a “safe folding environment” — Probably binds to polypeptide, to induce the correct folding conformation ...
Prions—Still a Mystery!
... spongiform diseases. It occurs spontaneously in one out of a million people, 10% of which are inherited mutations in the prion protein gene (PRPN). The disease was first described by Alfons Jakob in 1920 in a paper published in ...
... spongiform diseases. It occurs spontaneously in one out of a million people, 10% of which are inherited mutations in the prion protein gene (PRPN). The disease was first described by Alfons Jakob in 1920 in a paper published in ...
Poster
... ordered crystals required for structural studies. However, a small seven amino acid sequence (GNNQQNY) from Sup35's N-terminus was crystallized by Nelson et al. (2005). We have constructed a 3D physical model of GNNQQNY as part of ...
... ordered crystals required for structural studies. However, a small seven amino acid sequence (GNNQQNY) from Sup35's N-terminus was crystallized by Nelson et al. (2005). We have constructed a 3D physical model of GNNQQNY as part of ...
Prion diseases (transmissible spongiform encephalopathies)
... • Are a unique group of fatal neuro-degenerative disorders occurring in human beings and animals that possess major characteristics: All are transmissible to a variety of mammals, either experimentally or by natural exposure. The infectious agent is composed entirely of protein, without any nuc ...
... • Are a unique group of fatal neuro-degenerative disorders occurring in human beings and animals that possess major characteristics: All are transmissible to a variety of mammals, either experimentally or by natural exposure. The infectious agent is composed entirely of protein, without any nuc ...
Transmissible Spongiform Encephalopathies
... prion protein, a glycoprotein called PrPc formed by the Prnp gene. • TSE-infected cells contain the abnormal form of the protein, called PrPsc. This differs from the normal protein by having beta-sheets instead of alpha-helices ...
... prion protein, a glycoprotein called PrPc formed by the Prnp gene. • TSE-infected cells contain the abnormal form of the protein, called PrPsc. This differs from the normal protein by having beta-sheets instead of alpha-helices ...
Poster
... University of Illinois-Chicago's Laboratory for Molecular Biology are interested in the structure of GNNQQNY because it helps in understanding the structural change of Sup35 from a normal form to the prion form. Furthermore, it can provide insight to how prions fold in human/mammalian ...
... University of Illinois-Chicago's Laboratory for Molecular Biology are interested in the structure of GNNQQNY because it helps in understanding the structural change of Sup35 from a normal form to the prion form. Furthermore, it can provide insight to how prions fold in human/mammalian ...
Poster
... the prion escapes this targeting, causing these prion misfolds to accumulate in the cell. Prions are not only present in mammalian cells, but also in yeast cells which has made prion research easier. ...
... the prion escapes this targeting, causing these prion misfolds to accumulate in the cell. Prions are not only present in mammalian cells, but also in yeast cells which has made prion research easier. ...
source
... certain degenerative neurological diseases, such as sheep scrapie and bovine spongiform encephalopathy (BSE), and the human dementias Creutzfeldt-Jacob disease (CJD) and Gerstmann-Straussler syndrome (GSS). PrP is encoded in the host genome and is expressed both in normal and infected cells. During ...
... certain degenerative neurological diseases, such as sheep scrapie and bovine spongiform encephalopathy (BSE), and the human dementias Creutzfeldt-Jacob disease (CJD) and Gerstmann-Straussler syndrome (GSS). PrP is encoded in the host genome and is expressed both in normal and infected cells. During ...
ESHRE position statement concerning prion detection in urinary
... gonadotropin, questions the safety of urinary-derived human chorionic gonadotropin. Based on the detection of traces of normal prion protein in licensed urinary derived products, the authors surmise that these widely used gonadotropins might also harbor abnormally folded prion proteins that could re ...
... gonadotropin, questions the safety of urinary-derived human chorionic gonadotropin. Based on the detection of traces of normal prion protein in licensed urinary derived products, the authors surmise that these widely used gonadotropins might also harbor abnormally folded prion proteins that could re ...
prions lecture notes
... - CJD has cerebral involvement so dementia is more common; patient seldom survives a year - GSS is distinct from CJD characterized by cerebellar ataxia and concomitant motor problems, dementia is less common (disease course lasts several years before ultimate death) - fatal insomnias present with an ...
... - CJD has cerebral involvement so dementia is more common; patient seldom survives a year - GSS is distinct from CJD characterized by cerebellar ataxia and concomitant motor problems, dementia is less common (disease course lasts several years before ultimate death) - fatal insomnias present with an ...
The History of Chronic Wasting Disease
... The History of Chronic Wasting Disease Dr. Trent Bollinger, CCWHC One World, One Health Symposium Sept. 29, 2004 ...
... The History of Chronic Wasting Disease Dr. Trent Bollinger, CCWHC One World, One Health Symposium Sept. 29, 2004 ...
New drug offers hope in fight against mad cow disease
... Scientist Uli Herrmann and his research colleagues designed new polythiophenes with optimal ability to immobilize prions, the most promising of which prolonged survival of prion-infected mice and hamsters by more than 80 percent. The mice and hamsters treated with the compound showed fewer prion clu ...
... Scientist Uli Herrmann and his research colleagues designed new polythiophenes with optimal ability to immobilize prions, the most promising of which prolonged survival of prion-infected mice and hamsters by more than 80 percent. The mice and hamsters treated with the compound showed fewer prion clu ...
Acidic pH and Detergents Enhance in Vitro Conversion of Human
... The insoluble, β-sheet form of the prion protein (PrPSc) is the only known component associated with the group of transmissible, fatal neurodegenerative diseases found in humans and other animals. A posttranslational process, changes the conformation of the protein from normal PrPC to PrPSc. ...
... The insoluble, β-sheet form of the prion protein (PrPSc) is the only known component associated with the group of transmissible, fatal neurodegenerative diseases found in humans and other animals. A posttranslational process, changes the conformation of the protein from normal PrPC to PrPSc. ...
Sub-viral Agents
... groups, but was more prolonged in animals immunized before exposure. The increase in the incubation period closely correlated with the anti-prion protein antibody titer. This promising finding suggests that a similar approach may work in humans or other mammalian species at risk for prion disease. ...
... groups, but was more prolonged in animals immunized before exposure. The increase in the incubation period closely correlated with the anti-prion protein antibody titer. This promising finding suggests that a similar approach may work in humans or other mammalian species at risk for prion disease. ...
lecture 12
... deposits are toxic and produce neuronal dysfunction and death prion-related diseases occur when conversion of a normal prion protein, PrP, into an infectious and pathogenic form, PrPSc (Prion Protein Scrapie). Prion diseases: Creutzfeld Jacob disease, Kuru, Gerstmann-Straussler-Scheinker disea ...
... deposits are toxic and produce neuronal dysfunction and death prion-related diseases occur when conversion of a normal prion protein, PrP, into an infectious and pathogenic form, PrPSc (Prion Protein Scrapie). Prion diseases: Creutzfeld Jacob disease, Kuru, Gerstmann-Straussler-Scheinker disea ...
Chapter 50
... FIGURE 50-6: Molecular analysis of PrPSc isoforms. (A) The schematic demonstrates the principle of molecular strain typing of PrPSc isoforms by limited proteolytic digestion and Western blotting. Two distinct aggregates of PrPSc with differing conformations (shown in green or blue) present differen ...
... FIGURE 50-6: Molecular analysis of PrPSc isoforms. (A) The schematic demonstrates the principle of molecular strain typing of PrPSc isoforms by limited proteolytic digestion and Western blotting. Two distinct aggregates of PrPSc with differing conformations (shown in green or blue) present differen ...
CIC bioGUNE researchers embark on work to
... prions, showed a stable propagation of each one of the prion strains, the transgenic mice designed to replicate prions from mule deer propagated an unstable mixture of CWD strains, preventing their separation through their biochemical properties. The significance of this study is due to the fact tha ...
... prions, showed a stable propagation of each one of the prion strains, the transgenic mice designed to replicate prions from mule deer propagated an unstable mixture of CWD strains, preventing their separation through their biochemical properties. The significance of this study is due to the fact tha ...
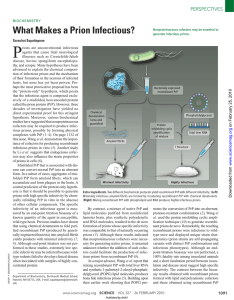
What Makes a Prion Infectious?
... Misfolded PrP that is associated with disease can convert normal PrP into an aberrant form. In a subset of cases, aggregates of misfolded PrP form amyloid fibers, which can accumulate and form plaques in the brain. A central prediction of the protein-only hypothesis is that it should be possible to g ...
... Misfolded PrP that is associated with disease can convert normal PrP into an aberrant form. In a subset of cases, aggregates of misfolded PrP form amyloid fibers, which can accumulate and form plaques in the brain. A central prediction of the protein-only hypothesis is that it should be possible to g ...
prion disease
... Design of such compounds had proven successful in cell-based models but must now be extended to animal models and human clinical trials 2. Vaccine design: The abnormally folded proteins expose a side chain of amino acids which the properly folded protein does not expose. Antibodies specifically code ...
... Design of such compounds had proven successful in cell-based models but must now be extended to animal models and human clinical trials 2. Vaccine design: The abnormally folded proteins expose a side chain of amino acids which the properly folded protein does not expose. Antibodies specifically code ...
Phenotypic diversity in acquired human prion diseases
... spongiform change in grey matter • Associated with conversion of PrPC to PrPSc ...
... spongiform change in grey matter • Associated with conversion of PrPC to PrPSc ...
Prion

A prion (/ˈpriːɒn/) is a protein that can fold in multiple, structurally distinct ways, at least one of which is transmissible to other prion proteins. It is this form of replication that leads to disease that is similar to viral infection. The word prion, coined in 1982 by Stanley B. Prusiner, is short for “proteinaceous infectious particle” derived from the words protein and infection, in reference to a prion's ability to self-propagate and transmit its conformation to other prions. While several yeast proteins have been identified as having prionogenic properties, the first prion protein was discovered in mammals and is referred to as the major prion protein (PrP). This infectious agent causes mammalian transmissible spongiform encephalopathies, including bovine spongiform encephalopathy (BSE, also known as ""mad cow disease"") and scrapie in sheep. In humans, PrP causes Creutzfeldt-Jakob Disease (CJD), variant Creutzfeldt-Jakob Disease (vCJD), Gerstmann–Sträussler–Scheinker syndrome, Fatal Familial Insomnia and kuru.A protein as an infectious agent stands in contrast to all other known infectious agents, like viruses, bacteria, fungi, or parasites—all of which must contain nucleic acids (either DNA, RNA, or both). All known prion diseases in mammals affect the structure of the brain or other neural tissue and all are currently untreatable and universally fatal.Prions are not considered living organisms because they are misfolded protein molecules which may propagate by transmitting a misfolded protein state. If a prion enters a healthy organism, it induces existing, properly folded proteins to convert into the misfolded prion form. In this way, the prion acts as a template to guide the misfolding of more proteins into prion form. In yeast, this refolding is assisted by chaperone proteins such as Hsp104p. These refolded prions can then go on to convert more proteins themselves, leading to a chain reaction resulting in large amounts of the prion form. All known prions induce the formation of an amyloid fold, in which the protein polymerises into an aggregate consisting of tightly packed beta sheets. Amyloid aggregates are fibrils, growing at their ends, and replicate when breakage causes two growing ends to become four growing ends. The incubation period of prion diseases is determined by the exponential growth rate associated with prion replication, which is a balance between the linear growth and the breakage of aggregates. (Note that the propagation of the prion depends on the presence of normally folded protein in which the prion can induce misfolding; animals that do not express the normal form of the prion protein can neither develop nor transmit the disease.)Prion aggregates are extremely stable and accumulate in infected tissue, causing tissue damage and cell death. This structural stability means that prions are resistant to denaturation by chemical and physical agents, making disposal and containment of these particles difficult. Prion structure varies slightly between species, but nonetheless prion replication is subject to occasional epimutation and natural selection just like other forms of replication.