Fundamentals of Energy Conversion
... called thermodynamic properties, such as pressure, temperature, and volume. Obviously, this approach excludes the possibility of description of the condition of the molecules of the system, a concern that is left to the fields of statistical and quantum mechanics and kinetic theory. Nevertheless, it ...
... called thermodynamic properties, such as pressure, temperature, and volume. Obviously, this approach excludes the possibility of description of the condition of the molecules of the system, a concern that is left to the fields of statistical and quantum mechanics and kinetic theory. Nevertheless, it ...
Temperature
... 4. Thermal Expansion of solids - bimetallic strip The bimetallic strip is made of two metals, one of which expands much more than the other when they are heated. A bi-metallic strip can be used as a sensor because the two metals which make up the strip expand at different rates. When they start to g ...
... 4. Thermal Expansion of solids - bimetallic strip The bimetallic strip is made of two metals, one of which expands much more than the other when they are heated. A bi-metallic strip can be used as a sensor because the two metals which make up the strip expand at different rates. When they start to g ...
Atomic Structure
... 4. Thermal Expansion of solids - bimetallic strip The bimetallic strip is made of two metals, one of which expands much more than the other when they are heated. A bi-metallic strip can be used as a sensor because the two metals which make up the strip expand at different rates. When they start to g ...
... 4. Thermal Expansion of solids - bimetallic strip The bimetallic strip is made of two metals, one of which expands much more than the other when they are heated. A bi-metallic strip can be used as a sensor because the two metals which make up the strip expand at different rates. When they start to g ...
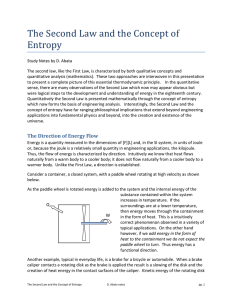
The Second Law and the Concept of Entropy
... The first practical heat engine appeared in the coal regions of Dudley north and slightly east of Cornwall, the southwestern tip of England about 1710 invented and constructed by Thomas Newcomen (1664 –1729) who was an ironmonger (iron and metal merchant) by trade and a Baptist lay preacher by calli ...
... The first practical heat engine appeared in the coal regions of Dudley north and slightly east of Cornwall, the southwestern tip of England about 1710 invented and constructed by Thomas Newcomen (1664 –1729) who was an ironmonger (iron and metal merchant) by trade and a Baptist lay preacher by calli ...
N2(g)
... – the study of transformations of energy – especially as heat and work Used to describe mechanical systems e.g. steam engine all chemical processes (e.g. combustion, dissolving of a solid, expansion of a gas) also involve exchange of heat or work. Terms you’ll use in thermodynamics: Energy – the cap ...
... – the study of transformations of energy – especially as heat and work Used to describe mechanical systems e.g. steam engine all chemical processes (e.g. combustion, dissolving of a solid, expansion of a gas) also involve exchange of heat or work. Terms you’ll use in thermodynamics: Energy – the cap ...
PDF (Chapter 5. Thermodynamics and Equations of State)
... At high temperature, 01T << 1, which is called the Griineisen relation. The thermal energy of a crystal is equal to the sum over all oscillators and, therefore, over all pertinent frequencies. In the Debye theory the sum is replaced by an integral, and it is assumed that all frequencies of vibration ...
... At high temperature, 01T << 1, which is called the Griineisen relation. The thermal energy of a crystal is equal to the sum over all oscillators and, therefore, over all pertinent frequencies. In the Debye theory the sum is replaced by an integral, and it is assumed that all frequencies of vibration ...
PPTX
... Principle: When a fluorescent molecule is excited with plane polarized light, light is emitted in the same polarized plane, provided that the molecule remains stationary throughout the excited state (which has a duration of 4 nanoseconds for fluorescein). If the molecule rotates and tumbles out of t ...
... Principle: When a fluorescent molecule is excited with plane polarized light, light is emitted in the same polarized plane, provided that the molecule remains stationary throughout the excited state (which has a duration of 4 nanoseconds for fluorescein). If the molecule rotates and tumbles out of t ...
- Philsci
... the thermodynamic limit. Basically, the infinite size makes shape irrelevant for SM’s pressure. Similarly the infinite system with realistic4 interactions among its constituents makes SM’s entropy and internal energy truly extensive because the ratio of the summation of bulk interactions to that of ...
... the thermodynamic limit. Basically, the infinite size makes shape irrelevant for SM’s pressure. Similarly the infinite system with realistic4 interactions among its constituents makes SM’s entropy and internal energy truly extensive because the ratio of the summation of bulk interactions to that of ...
Click to open the TEOS-10 teaching aid slides(powerpoint)
... Background to TEOS-10 • The 1980 International Equation of State (EOS-80) has served the community very well for 30 years. • EOS-80 provides separate algorithms for density, sound speed, heat capacity and freezing temperature. • However, EOS-80 does not provide expressions for entropy, internal ene ...
... Background to TEOS-10 • The 1980 International Equation of State (EOS-80) has served the community very well for 30 years. • EOS-80 provides separate algorithms for density, sound speed, heat capacity and freezing temperature. • However, EOS-80 does not provide expressions for entropy, internal ene ...
Thermal radiation
Thermal radiation is electromagnetic radiation generated by the thermal motion of charged particles in matter. An object with a temperature greater than absolute zero emits thermal radiation. When the temperature of the body is greater than absolute zero, interatomic collisions cause the kinetic energy of the atoms or molecules to change. This results in charge-acceleration and/or dipole oscillation which produces electromagnetic radiation, and the wide spectrum of radiation reflects the wide spectrum of energies and accelerations that occur even at a single temperature.Examples of thermal radiation include the visible light and infrared light emitted by an incandescent light bulb, the infrared radiation emitted by animals and detectable with an infrared camera, and the cosmic microwave background radiation. Thermal radiation is different from thermal convection and thermal conduction—a person near a raging bonfire feels radiant heating from the fire, even if the surrounding air is very cold.Sunlight is part of thermal radiation generated by the hot plasma of the Sun. The Earth also emits thermal radiation, but at a much lower intensity and different spectral distribution (infrared rather than visible) because it is cooler. The Earth's absorption of solar radiation, followed by its outgoing thermal radiation are the two most important processes that determine the temperature and climate of the Earth.If a radiation-emitting object meets the physical characteristics of a black body in thermodynamic equilibrium, the radiation is called blackbody radiation. Planck's law describes the spectrum of blackbody radiation, which depends only on the object's temperature. Wien's displacement law determines the most likely frequency of the emitted radiation, and the Stefan–Boltzmann law gives the radiant intensity.Thermal radiation is one of the fundamental mechanisms of heat transfer.