Synthesis of Natural Products and Related Compounds using Enyne
... step, and many bond fissions and bond formations occur during the process. In the total synthesis of natural products using dienyne metathesis, retro-synthetic analyses are completely different from those of the methods reported previously, and the reaction process was generally shortened. Grubbs de ...
... step, and many bond fissions and bond formations occur during the process. In the total synthesis of natural products using dienyne metathesis, retro-synthetic analyses are completely different from those of the methods reported previously, and the reaction process was generally shortened. Grubbs de ...
Kinetics and Mechanisms of the Reactions of Diaryl
... The reactions of methanol, tert-butanol, and THF with a series of simple germylene derivativess dimethyl- (GeMe2), diphenyl- (GePh2), and dimesitylgermylene (GeMes2; Mes ) 2,4,6-trimethylphenyl)s have been studied in hexane solution at 25 °C by laser flash photolysis methods. The results are consist ...
... The reactions of methanol, tert-butanol, and THF with a series of simple germylene derivativess dimethyl- (GeMe2), diphenyl- (GePh2), and dimesitylgermylene (GeMes2; Mes ) 2,4,6-trimethylphenyl)s have been studied in hexane solution at 25 °C by laser flash photolysis methods. The results are consist ...
Full Article - PDF - Brandeis University
... The scope of the reaction was found to be extremely general. Clean kinetic resolutions of extraordinarily high enantioselectivities were attainable with a wide range of UNCAs bearing various substituents and protecting groups (Table 3). Using the same extractive procedure for the isolation of 3a and ...
... The scope of the reaction was found to be extremely general. Clean kinetic resolutions of extraordinarily high enantioselectivities were attainable with a wide range of UNCAs bearing various substituents and protecting groups (Table 3). Using the same extractive procedure for the isolation of 3a and ...
LECTURE 7 REDUCTIVE ELIMINATIONSa
... • In catalysis reactions, a reductive elimination is often the last step in a catalytic cycle, and the resulting LnM n fragment must be able to survive long enough to react with the substrates for the organic reaction and so reenter the catalytic cycle. • Reductive elimination is analogous to the co ...
... • In catalysis reactions, a reductive elimination is often the last step in a catalytic cycle, and the resulting LnM n fragment must be able to survive long enough to react with the substrates for the organic reaction and so reenter the catalytic cycle. • Reductive elimination is analogous to the co ...
New Exp8
... Limitations of E1 Reaction: Acid-Catalyzed Dehydrations Competition can occur with SN1 reaction if reaction conditions are not ‘controlled’ (when protic solvents, non-basic nucleophiles are used). Mixtures of products form with the E1 reaction (also SN1). Unsymmetrical reagents and rearrangements po ...
... Limitations of E1 Reaction: Acid-Catalyzed Dehydrations Competition can occur with SN1 reaction if reaction conditions are not ‘controlled’ (when protic solvents, non-basic nucleophiles are used). Mixtures of products form with the E1 reaction (also SN1). Unsymmetrical reagents and rearrangements po ...
Asymmetric Synthesis: Substrate and Auxiliary Control
... ▪ New synthetic methods mean that some other chiral molecules are now extremely cheap, and are now considered as a new chiral pool. ...
... ▪ New synthetic methods mean that some other chiral molecules are now extremely cheap, and are now considered as a new chiral pool. ...
Cerium(IV) Ammonium Nitrate as a Catalyst in
... A simpler approach to the development of heterogeneous catalysts capable of oxidizing sulfides to sulfoxides consisted of the use of a silica-gel supported CAN-sodium bromate mixture, which catalyzed the oxidation in yields and scope similar to that of the immobilized cerium alkyl phosphonate cataly ...
... A simpler approach to the development of heterogeneous catalysts capable of oxidizing sulfides to sulfoxides consisted of the use of a silica-gel supported CAN-sodium bromate mixture, which catalyzed the oxidation in yields and scope similar to that of the immobilized cerium alkyl phosphonate cataly ...
lecture 11 catalysis_hydrogenation of alkenes
... dihydride leads to labilization of one of the PPh3 ligands (high trans effect of H) to give a site at which the alkene binds. ...
... dihydride leads to labilization of one of the PPh3 ligands (high trans effect of H) to give a site at which the alkene binds. ...
Group Meeting Special Topic: EJ Corey
... • Catalyst produced in 3 steps from (S)-(+)-Phenylglycinol • Diels-Alder experiments performed using Fe(III) halides with 4 and cyclopentadiene ...
... • Catalyst produced in 3 steps from (S)-(+)-Phenylglycinol • Diels-Alder experiments performed using Fe(III) halides with 4 and cyclopentadiene ...
74 CHAPTER-IV "LEAD (IV) ACETATE OXIDATIONS"
... low yields and mixture of products this reaction proved to be of little synthetic value. So Bacha and coworkers modified this reaction by using catalytic amounts of Cu (II) acetate which greatly enhanced the rate of decarboxylation and yields of alkenes. 38 This effect of Cu (IT) acetate is attribut ...
... low yields and mixture of products this reaction proved to be of little synthetic value. So Bacha and coworkers modified this reaction by using catalytic amounts of Cu (II) acetate which greatly enhanced the rate of decarboxylation and yields of alkenes. 38 This effect of Cu (IT) acetate is attribut ...
... not be explained by the other proposed mechanisms.4 The mechanism has also experimental support by Grubbs, T.J. Katz, Schrock and others and is now generally accepted as the mechanism for metathesis. Chauvin`s mechanism, with a metal alkylidene initiating metathesis, suggested that one could just sy ...
Studies toward the Stereoselective Synthesis of the
... In the dissertation the retrosynhetic analysis of this 3,7-dimethylundecane-1,5,6-triol building block identifies a simple precursor, ethyl 2-heptenoate, as the starting material for the proposed synthetic route toward this target. The Sharpless asymmetric epoxidation reaction plays a pivotal role i ...
... In the dissertation the retrosynhetic analysis of this 3,7-dimethylundecane-1,5,6-triol building block identifies a simple precursor, ethyl 2-heptenoate, as the starting material for the proposed synthetic route toward this target. The Sharpless asymmetric epoxidation reaction plays a pivotal role i ...
06_10_13.html
... The more stable the carbocation, the faster it is formed, and the faster the reaction rate. ...
... The more stable the carbocation, the faster it is formed, and the faster the reaction rate. ...
A Mild and Convenient Conversion of Ketones to the Corresponding
... to incorporate deuterium regiospecifically by using deuterium oxide as the source of deuterium.’O The purpose of this study was to investigate reducing agents which are as versatile as catecholborane but which can be more readily prepared. We report that bis(benzoyloxy)borane, I,l1J2effectively redu ...
... to incorporate deuterium regiospecifically by using deuterium oxide as the source of deuterium.’O The purpose of this study was to investigate reducing agents which are as versatile as catecholborane but which can be more readily prepared. We report that bis(benzoyloxy)borane, I,l1J2effectively redu ...
View/Open
... The rings of cycloalkenes containing five carbon atoms or fewer exist only in the cis form (Fig. 7.3). The introduction of a trans double bond into rings this small would, if it were possible, introduce greater strain than the bonds of the ring atoms could accommodate. ...
... The rings of cycloalkenes containing five carbon atoms or fewer exist only in the cis form (Fig. 7.3). The introduction of a trans double bond into rings this small would, if it were possible, introduce greater strain than the bonds of the ring atoms could accommodate. ...
Learning Guide for Chapter 16
... Learning Guide for Chapter 16 - Ethers and Epoxides I. Introduction to ethers, sulfides, and epoxides ...
... Learning Guide for Chapter 16 - Ethers and Epoxides I. Introduction to ethers, sulfides, and epoxides ...
Development of Multi-Component Reactions using Catalytically Generated Allyl Metal Reagents
... The term domino reactions was first defined by Tietze2 as “a process involving two or more bond-forming transformations (usually C-C bonds) which take place under the same reaction conditions without adding additional reagents and catalysts, and in which the subsequent reactions result as a conseque ...
... The term domino reactions was first defined by Tietze2 as “a process involving two or more bond-forming transformations (usually C-C bonds) which take place under the same reaction conditions without adding additional reagents and catalysts, and in which the subsequent reactions result as a conseque ...
Photoremovable Protecting Groups
... When the literature is insufficient for providing a comprehensive treatment of applications of a photoprotecting group, then only a brief discussion is provided. An exhaustive list of applications for any of the chromophores is not included; these may be found by consulting other reviews or the orig ...
... When the literature is insufficient for providing a comprehensive treatment of applications of a photoprotecting group, then only a brief discussion is provided. An exhaustive list of applications for any of the chromophores is not included; these may be found by consulting other reviews or the orig ...
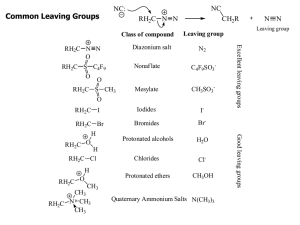
Common Leaving Groups
... Whenever substitution reactions are possible, we must also consider whether or not elimination reactions might occur under the same reaction conditions. In elimination reactions, a “neutral” molecule is „eliminated‟ from the substrate to form a π bond. The π bond is formed between the two carbon ato ...
... Whenever substitution reactions are possible, we must also consider whether or not elimination reactions might occur under the same reaction conditions. In elimination reactions, a “neutral” molecule is „eliminated‟ from the substrate to form a π bond. The π bond is formed between the two carbon ato ...
RELATIONSHIP BETWEEN Sn1 and E1 REACTIONS
... H Why is the trisubstituted alkene more stable than the other two? The relative stabilities of alkenes is measured by their heat of hydrogenation (much like the relative stabilities of cycloalkanes is measured by their heats of combustion, see ch. 3). The relationship is inverse: the higher the heat ...
... H Why is the trisubstituted alkene more stable than the other two? The relative stabilities of alkenes is measured by their heat of hydrogenation (much like the relative stabilities of cycloalkanes is measured by their heats of combustion, see ch. 3). The relationship is inverse: the higher the heat ...
Solvent and Temperature Effects on the Reduction and Amination
... To further examine the product distributions resulting from iPr-LAB, 1 M in THF, prepared from 1 equiv of nBuLi (Scheme 3), a series of reactions of iPr-LAB were performed, varying halides, temperatures, and solvents. The LAB reagent was prepared in THF and varying amounts of dioxane were added to c ...
... To further examine the product distributions resulting from iPr-LAB, 1 M in THF, prepared from 1 equiv of nBuLi (Scheme 3), a series of reactions of iPr-LAB were performed, varying halides, temperatures, and solvents. The LAB reagent was prepared in THF and varying amounts of dioxane were added to c ...
the suzuki-miyaura reaction and boron reagents – mechanism
... The mechanism and the role of the base in the transmetalation step has been of much debate. The work of Amatore and Jutand, Hartwig, and Schmidt provide compelling evidence for the catalytic transit through oxo-palladium pathway B. However, this only applies with some certainty to the coupling of ar ...
... The mechanism and the role of the base in the transmetalation step has been of much debate. The work of Amatore and Jutand, Hartwig, and Schmidt provide compelling evidence for the catalytic transit through oxo-palladium pathway B. However, this only applies with some certainty to the coupling of ar ...
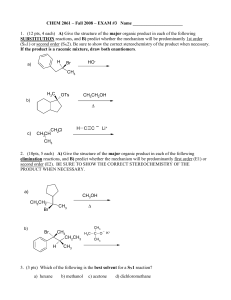
Exam #3
... (12 pts) Bromoetherification, the addition of the elements Br and OR to a double bond, is a common method for constructing rings containing oxygen atoms. Draw a stepwise mechanism for the following INTRAMOLECULAR bromoetherification reaction. Hint: the mechanism is analogous to that of bromohydrin f ...
... (12 pts) Bromoetherification, the addition of the elements Br and OR to a double bond, is a common method for constructing rings containing oxygen atoms. Draw a stepwise mechanism for the following INTRAMOLECULAR bromoetherification reaction. Hint: the mechanism is analogous to that of bromohydrin f ...
Catalytic Nucleophilic Fluorination of Secondary and Tertiary
... to the pharmaceutical[1] and agrochemical industries,[2] positron-emission tomography,[3] and materials science.[4] Catalytic fluorination has been the focus of many investigations,[5] in which either electrophilic or nucleophilic fluorine sources have been used.[6] Despite these significant advance ...
... to the pharmaceutical[1] and agrochemical industries,[2] positron-emission tomography,[3] and materials science.[4] Catalytic fluorination has been the focus of many investigations,[5] in which either electrophilic or nucleophilic fluorine sources have been used.[6] Despite these significant advance ...
Biomimetic Organic Synthesis. 2 Volume Set Brochure
... Biomimetic organic synthesis transposes the efficiency of nature's chemistry into the laboratory. Natural products (also known as secondary metabolites) are among the best examples of how nature can assemble atoms into highly complex structures. Therefore, biomimetic total syntheses of natural produ ...
... Biomimetic organic synthesis transposes the efficiency of nature's chemistry into the laboratory. Natural products (also known as secondary metabolites) are among the best examples of how nature can assemble atoms into highly complex structures. Therefore, biomimetic total syntheses of natural produ ...
Vinylcyclopropane rearrangement

The vinylcyclopropane rearrangement or vinylcyclopropane-cyclopentene rearrangement is a ring expansion reaction, converting a vinyl-substituted cyclopropane ring into a cyclopentene ring.Intense experimental as well as computational investigations have revealed that mechanistically, the vinylcyclopropane rearrangement can be thought of as either a diradical-mediated two-step and/or orbital-symmetry-controlled pericyclic process. The amount by which each of the two mechanisms is operative is highly dependent on the substrate.Due to its ability to form cyclopentene rings the vinylcyclopropane rearrangement has served several times as a key reaction in complex natural product synthesis.