GCSE Chemistry Sample Assessment Materials
... A level of response mark scheme is used. Before applying the mark scheme please read through the whole answer from start to finish. Firstly, decide which level descriptor matches best with the candidate’s response: remember that you should be considering the overall quality of the response. Then dec ...
... A level of response mark scheme is used. Before applying the mark scheme please read through the whole answer from start to finish. Firstly, decide which level descriptor matches best with the candidate’s response: remember that you should be considering the overall quality of the response. Then dec ...
Massachusetts Tests for Educator Licensure (MTEL )
... Correct Response: C. Keeping the sash lowered is necessary in order to contain fumes produced by the reaction within the exhaust hood. A is incorrect because using Bunsen burners within the fume exhaust hood does not present a safety hazard and is a necessary part of performing the experiment. B is ...
... Correct Response: C. Keeping the sash lowered is necessary in order to contain fumes produced by the reaction within the exhaust hood. A is incorrect because using Bunsen burners within the fume exhaust hood does not present a safety hazard and is a necessary part of performing the experiment. B is ...
hydrogen storage
... sample. The amount of adsorbed hydrogen from the gas phase at 77 K and electrochemically at RT is 1.5 × 10-3 mass%·m-2 g. Together with the maximum specific surface area of carbon (1315 m2 g-1), the maximum measured absorption capacity of the nanostructured material is 2 mass%. The experimental resu ...
... sample. The amount of adsorbed hydrogen from the gas phase at 77 K and electrochemically at RT is 1.5 × 10-3 mass%·m-2 g. Together with the maximum specific surface area of carbon (1315 m2 g-1), the maximum measured absorption capacity of the nanostructured material is 2 mass%. The experimental resu ...
Department of Chemistry - Catalog
... including dropping a course with a grade of “W” or taking an equivalent course at another institution, will be required to change his or her major. CHE 1103 or CHE 1143 ...
... including dropping a course with a grade of “W” or taking an equivalent course at another institution, will be required to change his or her major. CHE 1103 or CHE 1143 ...
Chapter 14 (Kinetics) – Slides and Practice
... Measuring Reaction Rate • To measure the reaction rate you need to be able to measure the concentration of at least one component in the mixture at many points in time • There are two ways of approaching this problem 1. for reactions that are complete in less than 1 hour, it is best to use continuo ...
... Measuring Reaction Rate • To measure the reaction rate you need to be able to measure the concentration of at least one component in the mixture at many points in time • There are two ways of approaching this problem 1. for reactions that are complete in less than 1 hour, it is best to use continuo ...
Document
... Measuring Reaction Rate • To measure the reaction rate you need to be able to measure the concentration of at least one component in the mixture at many points in time • There are two ways of approaching this problem 1. for reactions that are complete in less than 1 hour, it is best to use continuo ...
... Measuring Reaction Rate • To measure the reaction rate you need to be able to measure the concentration of at least one component in the mixture at many points in time • There are two ways of approaching this problem 1. for reactions that are complete in less than 1 hour, it is best to use continuo ...
Part 2-ICHO-26-30
... organically bound nitrogen to ammonium ion. Concentrated sodium hydroxide is then added, and the ammonia formed is distilled into hydrochloric acid of known volume and concentration. The excess hydrochloric acid is then back-titrated with a standard solution of sodium hydroxide, to determine nitroge ...
... organically bound nitrogen to ammonium ion. Concentrated sodium hydroxide is then added, and the ammonia formed is distilled into hydrochloric acid of known volume and concentration. The excess hydrochloric acid is then back-titrated with a standard solution of sodium hydroxide, to determine nitroge ...
Chemistry and Biochemistry
... beginning of each semester (fall, spring, summer). For consideration for a graduate assistantship, all application materials should be received at least five months prior to the desired starting date. Students whose undergraduate degree is not equivalent to the American Chemical Society certified Ba ...
... beginning of each semester (fall, spring, summer). For consideration for a graduate assistantship, all application materials should be received at least five months prior to the desired starting date. Students whose undergraduate degree is not equivalent to the American Chemical Society certified Ba ...
unit-4-notes-1_enthalpy-and-entropy
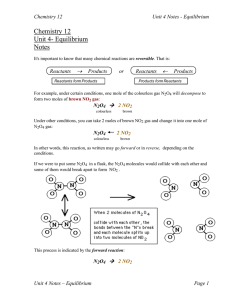
... and no NO2. The forward reaction rate was high at the start, but the reverse reaction rate eventually "caught up", the rates became equal and equilibrium was established. Can you guess what would happen if we had started with pure NO2 instead (no N2O4 )? The reverse rate would start out high and the ...
... and no NO2. The forward reaction rate was high at the start, but the reverse reaction rate eventually "caught up", the rates became equal and equilibrium was established. Can you guess what would happen if we had started with pure NO2 instead (no N2O4 )? The reverse rate would start out high and the ...
Chemistry MCQS 12 class
... (CaSO4.2H2O, 2CaSO4.H2O, (CaSO4)2H2O) 18. The atoms of the elements belonging to the same period of the Periodic table have __________. (Same number of protons, same number of neutrons, same number of valence shells) 19. Sodium thiosulphate is used in photography because of its __________. ...
... (CaSO4.2H2O, 2CaSO4.H2O, (CaSO4)2H2O) 18. The atoms of the elements belonging to the same period of the Periodic table have __________. (Same number of protons, same number of neutrons, same number of valence shells) 19. Sodium thiosulphate is used in photography because of its __________. ...