Medical College of Georgia
... nor generate nucleic acids that can replicate in any living cell (e.g., oligonucleotides or other synthetic nucleic acids that do not contain an origin of replication or contain elements known to interact with either DNA or RNA polymerase), and (2) are not designed to integrate into DNA, and (3) do ...
... nor generate nucleic acids that can replicate in any living cell (e.g., oligonucleotides or other synthetic nucleic acids that do not contain an origin of replication or contain elements known to interact with either DNA or RNA polymerase), and (2) are not designed to integrate into DNA, and (3) do ...
connecticut department of public health
... for repair or maintenance or packaging for transport, in accordance with applicable local, state, or federal regulations. 13. Cultures, tissues, specimens of body fluids, or wastes are placed in a container that prevents leakage during collection, handling, processing, storage, transport, or shippin ...
... for repair or maintenance or packaging for transport, in accordance with applicable local, state, or federal regulations. 13. Cultures, tissues, specimens of body fluids, or wastes are placed in a container that prevents leakage during collection, handling, processing, storage, transport, or shippin ...
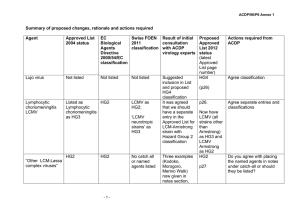
Summary of proposed changes, rationale and actions required Agent Approved List
... Lujo virus (LUJV), a new member of the family Arenaviridae and the first hemorrhagic fever–associated arenavirus from the Old World discovered in three decades, was isolated in South Africa during an outbreak of human disease characterized by nosocomial transmission and an unprecedented high case fa ...
... Lujo virus (LUJV), a new member of the family Arenaviridae and the first hemorrhagic fever–associated arenavirus from the Old World discovered in three decades, was isolated in South Africa during an outbreak of human disease characterized by nosocomial transmission and an unprecedented high case fa ...
Botulism Infection Control
... Thus, C. botulinum spores are hardy, resistant to dessication, heat, UV light, and alcohols, ...
... Thus, C. botulinum spores are hardy, resistant to dessication, heat, UV light, and alcohols, ...
... separated so as to prevent contact between them. (b) A rigid outer packaging of adequate strength for its capacity, mass and intended use. The smallest external dimension shall be not less than 100 mm. Additional requirements: 1 Inner packagings containing infectious substances shall not be consolid ...
Tularemia as a Biological Weapon Medical and Public Health
... infection, some exposures might contaminate the eye, resulting in ocular tularemia; penetrate broken skin, resulting in ulceroglandular or glandular disease; or cause oropharyngeal disease with cervical lymphadenitis. In the aforementioned Swedish outbreak, conjunctivitis was reported in 26% of 140 ...
... infection, some exposures might contaminate the eye, resulting in ocular tularemia; penetrate broken skin, resulting in ulceroglandular or glandular disease; or cause oropharyngeal disease with cervical lymphadenitis. In the aforementioned Swedish outbreak, conjunctivitis was reported in 26% of 140 ...
Transport of Biological Materials
... Biological materials that are hazardous due to an infection risk are allocated to UN class 6.2 infectious substances. Infectious substances are defined as substances which are known or are reasonably expected to contain pathogens which can cause disease in humans or animals. Pathogens are defined as ...
... Biological materials that are hazardous due to an infection risk are allocated to UN class 6.2 infectious substances. Infectious substances are defined as substances which are known or are reasonably expected to contain pathogens which can cause disease in humans or animals. Pathogens are defined as ...
Tularemia as a Biological Weapon
... expected to persist for several weeks and disease relapses to occur during the ensuing weeks or months. It was assumed that vaccinated individuals would be only partially protected against an aerosol exposure. Referring to this model, the Centers for Disease Control and Prevention (CDC) recently exa ...
... expected to persist for several weeks and disease relapses to occur during the ensuing weeks or months. It was assumed that vaccinated individuals would be only partially protected against an aerosol exposure. Referring to this model, the Centers for Disease Control and Prevention (CDC) recently exa ...
Tularemia as a Biological Weapon
... expected to persist for several weeks and disease relapses to occur during the ensuing weeks or months. It was assumed that vaccinated individuals would be only partially protected against an aerosol exposure. Referring to this model, the Centers for Disease Control and Prevention (CDC) recently exa ...
... expected to persist for several weeks and disease relapses to occur during the ensuing weeks or months. It was assumed that vaccinated individuals would be only partially protected against an aerosol exposure. Referring to this model, the Centers for Disease Control and Prevention (CDC) recently exa ...
standard operating procedure for transport of biological specimens
... Different packaging and transport arrangement apply depending on whether materials are infectious substances, biological products, cultures, genetically modified organism (GMO) or exempt substances. The requirements of various regulatory bodies are based on the United Nations Recommendations on the ...
... Different packaging and transport arrangement apply depending on whether materials are infectious substances, biological products, cultures, genetically modified organism (GMO) or exempt substances. The requirements of various regulatory bodies are based on the United Nations Recommendations on the ...
GWU Biosafety Manual - George Washington University School of
... GWU has an exposure control plan to comply with the OSHA blood-borne pathogen standard which applies to all on campus whose job requires potential contact with blood or other potentially infectious material. Those who work in laboratories, however, have special considerations due to the non-routine ...
... GWU has an exposure control plan to comply with the OSHA blood-borne pathogen standard which applies to all on campus whose job requires potential contact with blood or other potentially infectious material. Those who work in laboratories, however, have special considerations due to the non-routine ...
anthrax - sfcdcp
... causes illness and death. Of the potential ways that B. anthracis could be used as a biological weapon, an aerosol release is expected to have the most severe medical and public health ...
... causes illness and death. Of the potential ways that B. anthracis could be used as a biological weapon, an aerosol release is expected to have the most severe medical and public health ...
Anthrax JULY 2008 - San Francisco Bay Area Advanced Practice
... produces high levels of two toxins: Edema toxin, which causes massive edema at the site of germination, and lethal toxin, which leads to sepsis. Severity of anthrax disease depends on the route of infection and the presence of complications, with case-fatality ranging from 5% to 95% if untreated.1-3 ...
... produces high levels of two toxins: Edema toxin, which causes massive edema at the site of germination, and lethal toxin, which leads to sepsis. Severity of anthrax disease depends on the route of infection and the presence of complications, with case-fatality ranging from 5% to 95% if untreated.1-3 ...
Bioaerosol - eXtension
... Specifically, endotoxins in the outer membrane of Gram-negative bacteria are chemically stable and heat resistant. Endotoxins are released when the bacterial cells are lysed and will maintain their biological activity (i.e., adverse health effects), even after the bacterial cells are no longer viabl ...
... Specifically, endotoxins in the outer membrane of Gram-negative bacteria are chemically stable and heat resistant. Endotoxins are released when the bacterial cells are lysed and will maintain their biological activity (i.e., adverse health effects), even after the bacterial cells are no longer viabl ...
MCS Industry Day Slides - Biotechnology Industry Organization
... • Congressional Special Interests • University Affiliated Research Centers ...
... • Congressional Special Interests • University Affiliated Research Centers ...
Biological Materials Registration Form
... policies, and other federal, state and local regulations relating to this project. I attest that the information contained in the attached registration is accurate and complete. I accept responsibility for ensuring that all personnel involved in this project will be trained regarding the procedures ...
... policies, and other federal, state and local regulations relating to this project. I attest that the information contained in the attached registration is accurate and complete. I accept responsibility for ensuring that all personnel involved in this project will be trained regarding the procedures ...
biosafety manual - University of North Florida
... Work with known agents should be conducted at the biosafety level recommended unless specific information is available to suggest that virulence, pathogenicity, antibiotic resistance patterns, etc., are significantly altered to require more or allow less stringent practices to be used. As a general ...
... Work with known agents should be conducted at the biosafety level recommended unless specific information is available to suggest that virulence, pathogenicity, antibiotic resistance patterns, etc., are significantly altered to require more or allow less stringent practices to be used. As a general ...
biosafety levels and risk assessment policy
... 1) laboratory personnel have specific training in animal facility procedures and handling pathogenic agents and infected animals; 2) personnel are supervised by individuals with adequate knowledge of potential hazards, microbial agents, animal manipulations and husbandry procedures; 3) access to the ...
... 1) laboratory personnel have specific training in animal facility procedures and handling pathogenic agents and infected animals; 2) personnel are supervised by individuals with adequate knowledge of potential hazards, microbial agents, animal manipulations and husbandry procedures; 3) access to the ...
Institutional Biosafety Committee (IBC) Protocol Submittal Form
... physically handle biological materials, biohazardous agents or recombinant DNA molecules and are conceivably at risk from research procedures involving the use of these biological materials. Approval of the proposed experiment is given only for the identified personnel listed below. The Biosafety Of ...
... physically handle biological materials, biohazardous agents or recombinant DNA molecules and are conceivably at risk from research procedures involving the use of these biological materials. Approval of the proposed experiment is given only for the identified personnel listed below. The Biosafety Of ...
Packaging and Shipping - Minnesota Department of Health
... Summary of Regulations: The regulations divide infectious substances into two categories – A (infectious), and B (diagnostic). When clinical specimens are being shipped for the diagnosis and treatment of a patient and are not known to contain a Category A organism, they may be treated as diagnostic ...
... Summary of Regulations: The regulations divide infectious substances into two categories – A (infectious), and B (diagnostic). When clinical specimens are being shipped for the diagnosis and treatment of a patient and are not known to contain a Category A organism, they may be treated as diagnostic ...
Laboratory Biosafety Level Criteria
... research, or production facilities in which work is done with indigenous or exotic agents which may cause serious or potentially lethal disease as a result of exposure by the inhalation route. Laboratory personnel have specific training in handling pathogenic and potentially lethal agents, and are s ...
... research, or production facilities in which work is done with indigenous or exotic agents which may cause serious or potentially lethal disease as a result of exposure by the inhalation route. Laboratory personnel have specific training in handling pathogenic and potentially lethal agents, and are s ...
Biosafety Manual - Texas Tech University Departments
... consist entirely of DNA segments from a single nonchromosomal or viral DNA source, though one or more of the segments may be a synthetic equivalent. consist entirely of DNA from a prokaryotic host including its indigenous plasmids or viruses when propagated only in that host (or a closely related st ...
... consist entirely of DNA segments from a single nonchromosomal or viral DNA source, though one or more of the segments may be a synthetic equivalent. consist entirely of DNA from a prokaryotic host including its indigenous plasmids or viruses when propagated only in that host (or a closely related st ...
UH-Biological Safety Manual - University of Houston
... Biological agents are those pathogenic bacteria, viruses, fungi, and parasites that can be transmitted to a person or animal, directly or indirectly, and are capable of causing disease in the new host. Biological agents classified according to risk are listed in section III and Appendix B. If the ag ...
... Biological agents are those pathogenic bacteria, viruses, fungi, and parasites that can be transmitted to a person or animal, directly or indirectly, and are capable of causing disease in the new host. Biological agents classified according to risk are listed in section III and Appendix B. If the ag ...
Amendments - Office of the Vice Provost
... Life sciences research that, based on current understanding, can be reasonably anticipated to provide knowledge, information, products, or technologies that could be directly misapplied to pose a significant threat with broad potential consequences to public health and safety, agricultural crops and ...
... Life sciences research that, based on current understanding, can be reasonably anticipated to provide knowledge, information, products, or technologies that could be directly misapplied to pose a significant threat with broad potential consequences to public health and safety, agricultural crops and ...
Biological Agents and Factors Affecting Decontamination
... R egulatory Hurdle − EPA does not allow real or implied claims for any infectious agent that is not stated on the product label. − Very few commercial products have been tested against the candidate biological agents. ...
... R egulatory Hurdle − EPA does not allow real or implied claims for any infectious agent that is not stated on the product label. − Very few commercial products have been tested against the candidate biological agents. ...
Biological warfare

Biological warfare (BW)—also known as germ warfare—is the use of biological toxins or infectious agents such as bacteria, viruses, and fungi with the intent to kill or incapacitate humans, animals or plants as an act of war. Biological weapons (often termed ""bio-weapons"", ""biological threat agents"", or ""bio-agents"") are living organisms or replicating entities (viruses, which are not universally considered ""alive"") that reproduce or replicate within their host victims. Entomological (insect) warfare is also considered a type of biological weapon. This type of warfare is distinct from nuclear warfare and chemical warfare, which together with biological warfare make up NBC, the military acronym for nuclear, biological, and chemical warfare using weapons of mass destruction (WMDs). None of these are conventional weapons, which are primarily effective due to their explosive, kinetic, or incendiary potential.Biological weapons may be employed in various ways to gain a strategic or tactical advantage over the enemy, either by threats or by actual deployments. Like some of the chemical weapons, biological weapons may also be useful as area denial weapons. These agents may be lethal or non-lethal, and may be targeted against a single individual, a group of people, or even an entire population. They may be developed, acquired, stockpiled or deployed by nation states or by non-national groups. In the latter case, or if a nation-state uses it clandestinely, it may also be considered bioterrorism.There is an overlap between biological warfare and chemical warfare, as the use of toxins produced by living organisms is considered under the provisions of both the Biological Weapons Convention and the Chemical Weapons Convention. Toxins and psychochemical weapons are often referred to as midspectrum agents. Unlike bioweapons, these midspectrum agents do not reproduce in their host and are typically characterized by shorter incubation periods.