CHEM 121. Chapter 15
... A) H atom from the alcohol bonds to the carbonyl carbon atom. B) OH portion from the alcohol bonds to the carbonyl oxygen atom. C) OR portion from the alcohol bonds to the carbonyl carbon atom. D) OH portion from the alcohol bonds to the carbonyl carbon atom. 18. A hemiacetal is a compound in which ...
... A) H atom from the alcohol bonds to the carbonyl carbon atom. B) OH portion from the alcohol bonds to the carbonyl oxygen atom. C) OR portion from the alcohol bonds to the carbonyl carbon atom. D) OH portion from the alcohol bonds to the carbonyl carbon atom. 18. A hemiacetal is a compound in which ...
CHM-373 American Women in Science and Society
... Ultraviolet Spectra of Conjugated Carbonyls • Have characteristic absorption in UV spectrum • Additional conjugate C=C increases max about 30 nm, additional alkyl groups increase about ...
... Ultraviolet Spectra of Conjugated Carbonyls • Have characteristic absorption in UV spectrum • Additional conjugate C=C increases max about 30 nm, additional alkyl groups increase about ...
Organic Chemistry II / CHEM 252 Chapter 16
... • Dissolving aldehydes (or ketones) in water causes formation of an equilibrium between the carbonyl compound and its hydrate – The hydrate is also called a gem-diol (gem i.e. geminal, indicates the presence of two identical substituents on the same carbon) – The equilibrum favors a ketone over its ...
... • Dissolving aldehydes (or ketones) in water causes formation of an equilibrium between the carbonyl compound and its hydrate – The hydrate is also called a gem-diol (gem i.e. geminal, indicates the presence of two identical substituents on the same carbon) – The equilibrum favors a ketone over its ...
Study Guide 2 - Chemistry Teaching Resources
... KHS Jan 2015 - Cheviot Learning Community - based on Challenge Chemistry © R.I.S.E ...
... KHS Jan 2015 - Cheviot Learning Community - based on Challenge Chemistry © R.I.S.E ...
Biological Sciences Workbook
... All living beings (including the cells they are composed of) and non-living objects are made up of chemicals. The ancient Greeks recognised that all matter was composed of minute building blocks and although they were not sure of their existence they used the word atomata to describe them. Today the ...
... All living beings (including the cells they are composed of) and non-living objects are made up of chemicals. The ancient Greeks recognised that all matter was composed of minute building blocks and although they were not sure of their existence they used the word atomata to describe them. Today the ...
Camp 1
... • Termites have such bacteria in their intestines and can use wood as their principal food. • Ruminants (cud-chewing animals) and horses can also © 2006 Thomson Learning, Inc. All rights reserved digest grasses and hay. ...
... • Termites have such bacteria in their intestines and can use wood as their principal food. • Ruminants (cud-chewing animals) and horses can also © 2006 Thomson Learning, Inc. All rights reserved digest grasses and hay. ...
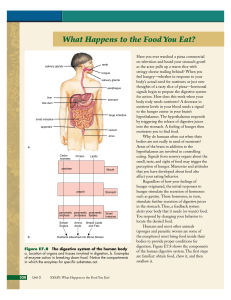
What Happens to the Food You Eat?
... proteins known as enzymes helps make this happen. Enzymes catalyze, or speed up, specific molecular reactions that otherwise would take place very slowly. Enzymes act on molecules called Proteins O substrates. Substrates bind to O specific places on enzymes. OH N O Carbohydrate and protein N OH N mo ...
... proteins known as enzymes helps make this happen. Enzymes catalyze, or speed up, specific molecular reactions that otherwise would take place very slowly. Enzymes act on molecules called Proteins O substrates. Substrates bind to O specific places on enzymes. OH N O Carbohydrate and protein N OH N mo ...
Chapter 25
... Sum of the biochemical reactions that occur in the cells of living organisms Functions: ...
... Sum of the biochemical reactions that occur in the cells of living organisms Functions: ...
Functions of the Rumen
... • Once simple sugars are formed, they are absorbed rapidly by small intestine • Then monosaccharides diffuse into the portal vein which transports them to sites of metabolism ...
... • Once simple sugars are formed, they are absorbed rapidly by small intestine • Then monosaccharides diffuse into the portal vein which transports them to sites of metabolism ...
Chapter 10: Chemistry of Living Systems
... What You Have Learned You have read that most of Earth’s biomass is composed primarily of six elements—carbon, hydrogen, nitrogen, oxygen, phosphorus, and sulfur. All living organisms obtain these elements from their environment. These elements go through natural cycles where they are used and reus ...
... What You Have Learned You have read that most of Earth’s biomass is composed primarily of six elements—carbon, hydrogen, nitrogen, oxygen, phosphorus, and sulfur. All living organisms obtain these elements from their environment. These elements go through natural cycles where they are used and reus ...
Camp 1
... large numbers of monosaccharide units joined by glycosidic bonds • Starch: a polymer of D-glucose • starch can be separated into amylose and amylopectin • amylose is composed of unbranched chains of up to 4000 D-glucose units joined by -1,4-glycosidic bonds • amylopectin contains chains up to 10,00 ...
... large numbers of monosaccharide units joined by glycosidic bonds • Starch: a polymer of D-glucose • starch can be separated into amylose and amylopectin • amylose is composed of unbranched chains of up to 4000 D-glucose units joined by -1,4-glycosidic bonds • amylopectin contains chains up to 10,00 ...
Additional Science Biology Summary
... 10. Explain the term diffusion and some factors that affect the speed of diffusion Environment and sampling can you??? 1. Suggest some factors that affect the distribution of organisms and explain why?? 2. Explain how to randomly sample (quadrats) and systematically sample (transect)? In the topics ...
... 10. Explain the term diffusion and some factors that affect the speed of diffusion Environment and sampling can you??? 1. Suggest some factors that affect the distribution of organisms and explain why?? 2. Explain how to randomly sample (quadrats) and systematically sample (transect)? In the topics ...
Food and Nutrition
... It picks up the molecules of food and carries them to the cells of your body. Now the apple you ate hours ago is fully digested. It was turned into molecules that travel through your blood. Most of the molecules in an apple are sugars. (An apple tree made the sugars during photosynthesis. It stored ...
... It picks up the molecules of food and carries them to the cells of your body. Now the apple you ate hours ago is fully digested. It was turned into molecules that travel through your blood. Most of the molecules in an apple are sugars. (An apple tree made the sugars during photosynthesis. It stored ...
Review Notes Biology 20
... Metabolism: sum of all chemical reactions that occur within the cell. Catabolism: chemical reactions in which complex molecules are broken down into smaller compounds. Anabolism: chemical reactions where simple molecules are combined together to form more complex compounds. Organic Chemistry: ...
... Metabolism: sum of all chemical reactions that occur within the cell. Catabolism: chemical reactions in which complex molecules are broken down into smaller compounds. Anabolism: chemical reactions where simple molecules are combined together to form more complex compounds. Organic Chemistry: ...
Carbohydrates - Adlai E. Stevenson High School
... 1. Carbohydrates: the starches & sugars found in foods • Body’s chief & preferred source of fuel • Provides 4 calories per gram • Made up of carbon, oxygen, & hydrogen • Examples: potatoes, pasta, bread, fruit,veg. • Recommended: 55%-65% of daily calories come from carbs, mostly complex carbs. ...
... 1. Carbohydrates: the starches & sugars found in foods • Body’s chief & preferred source of fuel • Provides 4 calories per gram • Made up of carbon, oxygen, & hydrogen • Examples: potatoes, pasta, bread, fruit,veg. • Recommended: 55%-65% of daily calories come from carbs, mostly complex carbs. ...
Basic Agricultural Chemistry - Macmillan Education South Africa
... group share similar chemical properties. The Roman numerals listed above each group are the usual number of valence electrons. For example: l Group IA is the alkali metals where the element has one electron in the orbital, for example sodium, potassium and rubidium. Alkali metals form salts and man ...
... group share similar chemical properties. The Roman numerals listed above each group are the usual number of valence electrons. For example: l Group IA is the alkali metals where the element has one electron in the orbital, for example sodium, potassium and rubidium. Alkali metals form salts and man ...
ERP 10 - Haiku Learning
... • This is a complicated system so it cannot be used immediately. It takes time for enough oxygen to become available to meet the demands of the activity and ensure glycogen and fatty acids are completely broken down. • Fatty acid transportation to muscles is low and fatty acids require 15% more oxyg ...
... • This is a complicated system so it cannot be used immediately. It takes time for enough oxygen to become available to meet the demands of the activity and ensure glycogen and fatty acids are completely broken down. • Fatty acid transportation to muscles is low and fatty acids require 15% more oxyg ...
Unit 3 - Amazon Web Services
... is very much like the track. God has allowed circumstances and situations in your life similar to these hurdles. If your life seems peaceful right now, you can count on the fact that there will be some hurdles up ahead. Just like the athletes who train to run the hurdles, God expects us to train in ...
... is very much like the track. God has allowed circumstances and situations in your life similar to these hurdles. If your life seems peaceful right now, you can count on the fact that there will be some hurdles up ahead. Just like the athletes who train to run the hurdles, God expects us to train in ...
here - Polyols EU
... vegetables, for example sorbitol in plums, erythritol in grapes, or xylitol in mushrooms. Starting materials for industrial processes are all from natural sources (corn, wheat, sugar beet, milk, etc.). Depending on the agricultural source, the carbohydrates contained are processed as such, or in the ...
... vegetables, for example sorbitol in plums, erythritol in grapes, or xylitol in mushrooms. Starting materials for industrial processes are all from natural sources (corn, wheat, sugar beet, milk, etc.). Depending on the agricultural source, the carbohydrates contained are processed as such, or in the ...
Complete the following
... 1- circulation is the process by which energy is extracted from bonds of the food (produced by plants and consumed by others 2- Carbohydrates are forms of storing energy (and transport energy between cells and between living organisms) 3- Protein is a carbohydrate that is used commonly by the majori ...
... 1- circulation is the process by which energy is extracted from bonds of the food (produced by plants and consumed by others 2- Carbohydrates are forms of storing energy (and transport energy between cells and between living organisms) 3- Protein is a carbohydrate that is used commonly by the majori ...
Nutrients
... What is the function of vitamin C? Vitamin C is needed for the normal structure and function of body tissues, e.g. collagen. Vitamin C also helps the body to absorb the iron from non meat sources such as vegetables. It also assists the healing process. ...
... What is the function of vitamin C? Vitamin C is needed for the normal structure and function of body tissues, e.g. collagen. Vitamin C also helps the body to absorb the iron from non meat sources such as vegetables. It also assists the healing process. ...
Carbohydrates - Livonia Public Schools
... 1. Carbohydrates: the starches & sugars found in foods • Body’s chief & preferred source of fuel • Provides 4 calories per gram • Made up of carbon, oxygen, & hydrogen • Examples: potatoes, pasta, bread, fruit,veg. • Recommended: 55%-65% of daily calories come from carbs, mostly complex carbs. ...
... 1. Carbohydrates: the starches & sugars found in foods • Body’s chief & preferred source of fuel • Provides 4 calories per gram • Made up of carbon, oxygen, & hydrogen • Examples: potatoes, pasta, bread, fruit,veg. • Recommended: 55%-65% of daily calories come from carbs, mostly complex carbs. ...
Topical Questions and Answers
... Foods high in saturated fat include butter, cream, cheese and the fat from meat as well as coconut oil and palm oil. The link between saturated fat consumption, blood cholesterol levels and heart disease is well established, with evidence building over the past 60 years. Recent comments, highlighted ...
... Foods high in saturated fat include butter, cream, cheese and the fat from meat as well as coconut oil and palm oil. The link between saturated fat consumption, blood cholesterol levels and heart disease is well established, with evidence building over the past 60 years. Recent comments, highlighted ...
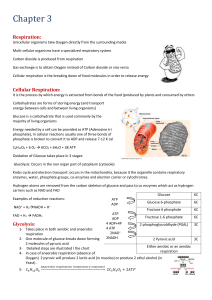
Cellular respiration
... ADP = adenosine diphosphate H2O + ATP ADP + P + energy • This is the energy used by the body to carry out the functions of life ...
... ADP = adenosine diphosphate H2O + ATP ADP + P + energy • This is the energy used by the body to carry out the functions of life ...
Carbohydrate
A carbohydrate is a biological molecule consisting of carbon (C), hydrogen (H) and oxygen (O) atoms, usually with a hydrogen:oxygen atom ratio of 2:1 (as in water); in other words, with the empirical formula Cm(H2O)n (where m could be different from n). Some exceptions exist; for example, deoxyribose, a sugar component of DNA, has the empirical formula C5H10O4. Carbohydrates are technically hydrates of carbon; structurally it is more accurate to view them as polyhydroxy aldehydes and ketones.The term is most common in biochemistry, where it is a synonym of saccharide, a group that includes sugars, starch, and cellulose. The saccharides are divided into four chemical groups: monosaccharides, disaccharides, oligosaccharides, and polysaccharides. In general, the monosaccharides and disaccharides, which are smaller (lower molecular weight) carbohydrates, are commonly referred to as sugars. The word saccharide comes from the Greek word σάκχαρον (sákkharon), meaning ""sugar."" While the scientific nomenclature of carbohydrates is complex, the names of the monosaccharides and disaccharides very often end in the suffix -ose. For example, grape sugar is the monosaccharide glucose, cane sugar is the disaccharide sucrose and milk sugar is the disaccharide lactose (see illustration).Carbohydrates perform numerous roles in living organisms. Polysaccharides serve for the storage of energy (e.g., starch and glycogen) and as structural components (e.g., cellulose in plants and chitin in arthropods). The 5-carbon monosaccharide ribose is an important component of coenzymes (e.g., ATP, FAD and NAD) and the backbone of the genetic molecule known as RNA. The related deoxyribose is a component of DNA. Saccharides and their derivatives include many other important biomolecules that play key roles in the immune system, fertilization, preventing pathogenesis, blood clotting, and development.In food science and in many informal contexts, the term carbohydrate often means any food that is particularly rich in the complex carbohydrate starch (such as cereals, bread and pasta) or simple carbohydrates, such as sugar (found in candy, jams, and desserts).