CH 3502 4500
... 16. Discuss the mechanism of cleavage of ethers by HI. 17. Explain Williamson’s synthesis of ethers. 18. Discuss Norrish type-I reaction. 19. Discuss the mechanism of Wittig reaction and its uses in organic synthesis. 20. Explain Wolf-Kishner reduction with its mechanism. 21. Give any two methods o ...
... 16. Discuss the mechanism of cleavage of ethers by HI. 17. Explain Williamson’s synthesis of ethers. 18. Discuss Norrish type-I reaction. 19. Discuss the mechanism of Wittig reaction and its uses in organic synthesis. 20. Explain Wolf-Kishner reduction with its mechanism. 21. Give any two methods o ...
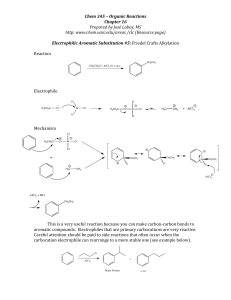
EAS Friedel-Crafts Alkylation
... The Friedel-‐Crafts reaction only requires catalytic amounts of the Lewis acid because it is recycled through the reaction. There is a major disadvantage of the alkylation reaction and that is over-‐ alkyla ...
... The Friedel-‐Crafts reaction only requires catalytic amounts of the Lewis acid because it is recycled through the reaction. There is a major disadvantage of the alkylation reaction and that is over-‐ alkyla ...
Assignment 2 Group A and B
... 9) Which of the following alcohols can be prepared by the reaction of methyl formate with excess Grignard reagent? A) 1-pentanol B) 2-pentanol C) 3-pentanol D) 2-methyl-2-pentanol E) 3-methyl-3-pentanol 10) What reagent(s) would you use to accomplish the following conversion? ...
... 9) Which of the following alcohols can be prepared by the reaction of methyl formate with excess Grignard reagent? A) 1-pentanol B) 2-pentanol C) 3-pentanol D) 2-methyl-2-pentanol E) 3-methyl-3-pentanol 10) What reagent(s) would you use to accomplish the following conversion? ...
Mechanism
... As a preservative to prevent against bacterial and fungal growth. It’s added to many drinks including Diet Coke. Quinone functional group used in the living organisms to perform oxidations with in cell. So your body also does oxidations as we speak. ...
... As a preservative to prevent against bacterial and fungal growth. It’s added to many drinks including Diet Coke. Quinone functional group used in the living organisms to perform oxidations with in cell. So your body also does oxidations as we speak. ...
Study guide for Catalysis 1) Which of the following statements is
... a) changes in [S] are negligible, so [S] can be treated as a constant. b) [ES] can be measured accurately. c) V0 = Vmax. d) changes in Km are negligible, so Km can be treated as a constant. e) varying [S] has no effect on V0. 3) Vmax for an enzyme-catalyzed reaction: a) generally increase when pH in ...
... a) changes in [S] are negligible, so [S] can be treated as a constant. b) [ES] can be measured accurately. c) V0 = Vmax. d) changes in Km are negligible, so Km can be treated as a constant. e) varying [S] has no effect on V0. 3) Vmax for an enzyme-catalyzed reaction: a) generally increase when pH in ...
study note 3 33
... An addition of a molecule to a double or triple bond. The molecule is broken down in the reaction; the two parts of the molecule are added to either side of the double bond, leaving a single bond (or leaving a double bond when the addition is to a triple bond). Halogenation, and hydrogenation are ty ...
... An addition of a molecule to a double or triple bond. The molecule is broken down in the reaction; the two parts of the molecule are added to either side of the double bond, leaving a single bond (or leaving a double bond when the addition is to a triple bond). Halogenation, and hydrogenation are ty ...
$doc.title
... http://www.chem.wisc.edu/areas/clc (Resource page) Reactions of Alcohols #8: Reaction of a 1° Alcohol with Hydrogen Halides ...
... http://www.chem.wisc.edu/areas/clc (Resource page) Reactions of Alcohols #8: Reaction of a 1° Alcohol with Hydrogen Halides ...
Chemistry 201 - Department of Chemistry | Oregon State University
... Which of the following statements is true? (A) a chiral molecule is not superimposable on its mirror image (B) glycine (Gly) is an amino acid which only has 1 chiral carbon (C) all amino acids are chiral (D) a chiral carbon has 3 identical groups bound to it (E) the following molecule is chiral: Cl ...
... Which of the following statements is true? (A) a chiral molecule is not superimposable on its mirror image (B) glycine (Gly) is an amino acid which only has 1 chiral carbon (C) all amino acids are chiral (D) a chiral carbon has 3 identical groups bound to it (E) the following molecule is chiral: Cl ...
Name
... 1. Two compounds with the same formula but different structures are called what? (1) Isomers 2. Organic compounds (1) A) always contain nitrogen. B) are synthesized by only animal cells. C) always contain carbon. D) can only be synthesized in a laboratory. E) always contain oxygen. ...
... 1. Two compounds with the same formula but different structures are called what? (1) Isomers 2. Organic compounds (1) A) always contain nitrogen. B) are synthesized by only animal cells. C) always contain carbon. D) can only be synthesized in a laboratory. E) always contain oxygen. ...
Problem Set for "The Origin of Chirality" by Anne
... Problem Set for "The Origin of Chirality" by Anne‐Catherine Bédard 1. In 2008, Breslow reported that α‐methyl amino acids found on meteorites to be a plausible source of biological homochirality. These enantioenriched amino acids can transfer their chirality through copper catalysis ...
... Problem Set for "The Origin of Chirality" by Anne‐Catherine Bédard 1. In 2008, Breslow reported that α‐methyl amino acids found on meteorites to be a plausible source of biological homochirality. These enantioenriched amino acids can transfer their chirality through copper catalysis ...
CHEM 201 Name Quiz 10 (Ch 17) ID Q1. Which of the following
... Q3. Which of the following reactions would not normally yield an alcohol? a) Oxymercuration/ demercuraction of ...
... Q3. Which of the following reactions would not normally yield an alcohol? a) Oxymercuration/ demercuraction of ...
Enantiospecific skeleton expanding cross
... cost organic synthesis. In just one example, a lactic acid derivative is converted to 2-methylhexanoic acid with excellent yield. This compound is an important building block in the production of the potential high-potency sweetener NC-00637. Ultimately, this new reaction presents an extremely attra ...
... cost organic synthesis. In just one example, a lactic acid derivative is converted to 2-methylhexanoic acid with excellent yield. This compound is an important building block in the production of the potential high-potency sweetener NC-00637. Ultimately, this new reaction presents an extremely attra ...
AMINO ACIDS Ethan Secor, John N. Gitua (Mentor)
... The titanium complex was prepared in purified tetrahydrofuran using titanium(IV) isopropoxide and butyl lithium at low temperatures maintained by mixing acetone and dry ice. The imine was added to this mixture. The reaction mixture was gradually warmed to room temperature. Carbon dioxide was introdu ...
... The titanium complex was prepared in purified tetrahydrofuran using titanium(IV) isopropoxide and butyl lithium at low temperatures maintained by mixing acetone and dry ice. The imine was added to this mixture. The reaction mixture was gradually warmed to room temperature. Carbon dioxide was introdu ...
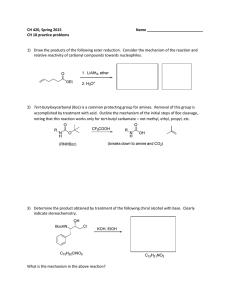
CH 420, Spring 2015 Name ___________________________ CH 18 practice problems
... 7) Rank the following compounds according to their relative acidity: cyclohexanol, phenol, pmethoxyphenol, p-nitrophenol. ...
... 7) Rank the following compounds according to their relative acidity: cyclohexanol, phenol, pmethoxyphenol, p-nitrophenol. ...
Petasis reaction

The Petasis reaction (alternatively called the Petasis borono–Mannich (PBM) reaction) is the chemical reaction of an amine, aldehyde, and vinyl- or aryl-boronic acid to form substituted amines.Reported in 1993 by Nicos Petasis as a practical method towards the synthesis of a geometrically pure antifungal agent, naftifine, the Petasis reaction can be described as a variation of the Mannich reaction. Rather than generating an enolate to form the substituted amine product, in the Petasis reaction, the vinyl group of the organoboronic acid serves as the nucleophile. In comparison to other methods of generating allyl amines, the Petasis reaction tolerates a multifunctional scaffold, with a variety of amines and organoboronic acids as potential starting materials. Additionally, the reaction does not require anhydrous or inert conditions. As a mild, selective synthesis, the Petasis reaction is useful in generating α-amino acids, and is utilized in combinatorial chemistry and drug discovery.