Chapter 7: Structure and Synthesis of Alkenes
... Ú Alkenes are hydrocarbons with C=C in their structure. They are also known as “olefins”. Ú We have already covered the basics of alkenes nomenclature, but here are a few important groups with common names that you can use as well. ...
... Ú Alkenes are hydrocarbons with C=C in their structure. They are also known as “olefins”. Ú We have already covered the basics of alkenes nomenclature, but here are a few important groups with common names that you can use as well. ...
Grade 11: Physical Sciences Outline
... Dehydration of alcohols: Elimination of water from an alcohol Cracking of alkanes: The chemical process in which longer chain hydrocarbon molecules are broken down to shorter more useful molecules. Write down, using structural formulae, equations and reaction conditions for the following substitutio ...
... Dehydration of alcohols: Elimination of water from an alcohol Cracking of alkanes: The chemical process in which longer chain hydrocarbon molecules are broken down to shorter more useful molecules. Write down, using structural formulae, equations and reaction conditions for the following substitutio ...
Topic 10. Organic chemistry
... Crude oil was formed over millions of years ago when the remains of animals and plants were trapped under layers of rock. Crude oil is a complex mixture of many different organic compounds, mainly alkanes. ...
... Crude oil was formed over millions of years ago when the remains of animals and plants were trapped under layers of rock. Crude oil is a complex mixture of many different organic compounds, mainly alkanes. ...
Chapter 10_Organohalides
... reaction pathway with Cl2 or Br2 and light (h) • Not very useful to the lack of control over the reaction and can lead ...
... reaction pathway with Cl2 or Br2 and light (h) • Not very useful to the lack of control over the reaction and can lead ...
Classes and Nomenclature of Halogen Compounds
... C) Formation of organometallic compounds. Most organic chlorides, bromides, and iodides react with certain metals to give organometallic compounds, molecules with carbon-metal bonds. Grignard reagents are obtained by the reaction of alkyl or aryl halides with metallic magnesium in dry ether as t ...
... C) Formation of organometallic compounds. Most organic chlorides, bromides, and iodides react with certain metals to give organometallic compounds, molecules with carbon-metal bonds. Grignard reagents are obtained by the reaction of alkyl or aryl halides with metallic magnesium in dry ether as t ...
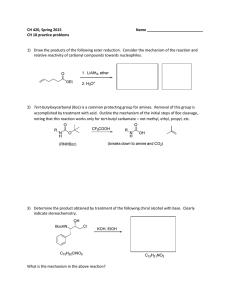
CH 420, Spring 2015 Name ___________________________ CH 18 practice problems
... 3) Determine the product obtained by treatment of the following chiral alcohol with base. Clearly indicate stereochemistry. ...
... 3) Determine the product obtained by treatment of the following chiral alcohol with base. Clearly indicate stereochemistry. ...
Carboxylic Acid
... You know Alkanes and Benzenes, and Alkynes and Alkenes, Amines and Alcohols, Aldehydes and Ketones...... But do you recall, the most famous functional group of all Carboxylic Acid, Has a carbonyl and a hydroxyl It loves to donate protons Then an anion is formed Of all the other acids It’s the most c ...
... You know Alkanes and Benzenes, and Alkynes and Alkenes, Amines and Alcohols, Aldehydes and Ketones...... But do you recall, the most famous functional group of all Carboxylic Acid, Has a carbonyl and a hydroxyl It loves to donate protons Then an anion is formed Of all the other acids It’s the most c ...
Organic Chemistry - EO-204-Distillation
... Organic Molecules that have the same formulas but a differing arrangement of the atoms involved. So……even though they have the same formula ….they have different properties Whether the compounds are Saturated or Unsaturated they are called “Aliphatic Compounds” because they are either straight or br ...
... Organic Molecules that have the same formulas but a differing arrangement of the atoms involved. So……even though they have the same formula ….they have different properties Whether the compounds are Saturated or Unsaturated they are called “Aliphatic Compounds” because they are either straight or br ...
Catalytic Hydrogenation of Alkenes: Relative Stability of
... Secondary and tertiary alcohols dehydrate by an E1 mechanism. The protonated hydroxy forms an alkyloxonium ion providing a good leaving group: water. Loss of water forms a secondary or tertiary carbocation. Deprotonation forms the alkene. Carbocation side reactions (hydrogen shifts, alkyl shifts, e ...
... Secondary and tertiary alcohols dehydrate by an E1 mechanism. The protonated hydroxy forms an alkyloxonium ion providing a good leaving group: water. Loss of water forms a secondary or tertiary carbocation. Deprotonation forms the alkene. Carbocation side reactions (hydrogen shifts, alkyl shifts, e ...
Topic Guide
... Dehydrohalogenation (twice)-mechanism Know C--C bond rxn; use of R-C=C Na as a base-mechanism halogenations; addn of HX (sia)2BH; H2O2; OH and HgSO4; H2SO4 keto-enol tautomerism & how it is used Complete & partial; stereochemistry Know how to do simple two step synthesis-how to use the reactions in ...
... Dehydrohalogenation (twice)-mechanism Know C--C bond rxn; use of R-C=C Na as a base-mechanism halogenations; addn of HX (sia)2BH; H2O2; OH and HgSO4; H2SO4 keto-enol tautomerism & how it is used Complete & partial; stereochemistry Know how to do simple two step synthesis-how to use the reactions in ...
Functional Groups - SISIBChemistry2012
... Functional Groups - The reactive portion of a molecule that undergoes predictable reactions. We have already looked at alkenes, alkynes and haloalkanes. - Functional groups are attached to the carbon chain, which is represented by R, which means the rest of the carbons. - Solubility in water depends ...
... Functional Groups - The reactive portion of a molecule that undergoes predictable reactions. We have already looked at alkenes, alkynes and haloalkanes. - Functional groups are attached to the carbon chain, which is represented by R, which means the rest of the carbons. - Solubility in water depends ...
lec-2- 211(ES +Add)
... on the addition of hydrogen halides to alkenes. •The rule states that : "when an unsymmetrical alkene reacts with a hydrogen halide to give an alkyl halide, the hydrogen adds to the carbon of the alkene that has the greater number of hydrogen substituents, and the halogen to the carbon of the alkene ...
... on the addition of hydrogen halides to alkenes. •The rule states that : "when an unsymmetrical alkene reacts with a hydrogen halide to give an alkyl halide, the hydrogen adds to the carbon of the alkene that has the greater number of hydrogen substituents, and the halogen to the carbon of the alkene ...
Table
... With halogens or hydrogen halides: Halogenation With Nitric Acid Complete combustion reactions Addition Reactions With H2: Hydrogenation With halogens or hydrogen halides: Halogenation With water: Hydration Substitution Reactions With X2: Halogenation Ø-X With HNO3: Nitration Ø-NO2 With RX: Alkylati ...
... With halogens or hydrogen halides: Halogenation With Nitric Acid Complete combustion reactions Addition Reactions With H2: Hydrogenation With halogens or hydrogen halides: Halogenation With water: Hydration Substitution Reactions With X2: Halogenation Ø-X With HNO3: Nitration Ø-NO2 With RX: Alkylati ...
Chap Thirteen: Alcohols
... ii. Reduction of Esters and Carboxylic Acids with LiAlH4 only LEARNING OUTCOMES: Understand the formation of alcohols through use of organometallics in carboncarbon bond forming reactions with electrophiles Predict the stereochemistry and optical activity of a product from an understanding of it ...
... ii. Reduction of Esters and Carboxylic Acids with LiAlH4 only LEARNING OUTCOMES: Understand the formation of alcohols through use of organometallics in carboncarbon bond forming reactions with electrophiles Predict the stereochemistry and optical activity of a product from an understanding of it ...
Topic 10 IB Chemistry Definitions
... hydroxyl group). Can be oxidized to aldehydes (by loss of hydrogens) and then to carboxylic acids (by gain of oxygen). Ethanol can be oxidized to ethanal by the orange Cr2O72- ion, which itself becomes reduced to the green Cr3+ ion. ...
... hydroxyl group). Can be oxidized to aldehydes (by loss of hydrogens) and then to carboxylic acids (by gain of oxygen). Ethanol can be oxidized to ethanal by the orange Cr2O72- ion, which itself becomes reduced to the green Cr3+ ion. ...
TYPES OF REACTIONS IN ORGANIC CHEMISTRY
... ~ Ethane is produced in small amounts, can only be explained by CH3 + CH3 ~ The presence of tetramethyl - lead greatly speeds up the reaction as it is a source of methyl free radicals. ...
... ~ Ethane is produced in small amounts, can only be explained by CH3 + CH3 ~ The presence of tetramethyl - lead greatly speeds up the reaction as it is a source of methyl free radicals. ...
organometallic reagents
... Watch out for pitfalls in planning syntheses. Try to minimize the total number of transformations required to convert the initial starting material into the desired product. A seven-step synthesis with a yield of 85% at each step gives an overall efficiency of conversion of 32%. A four-step synthes ...
... Watch out for pitfalls in planning syntheses. Try to minimize the total number of transformations required to convert the initial starting material into the desired product. A seven-step synthesis with a yield of 85% at each step gives an overall efficiency of conversion of 32%. A four-step synthes ...
Alkenes
... The 2s electron and two of the 2p electrons combine to form three sp2 hybrid orbitals, leaving a spare porbital on each of the carbon atoms ...
... The 2s electron and two of the 2p electrons combine to form three sp2 hybrid orbitals, leaving a spare porbital on each of the carbon atoms ...
Hydrocarbons
... components are added. There are a number of different types of addition reactions: Hydrogenation – the addition of hydrogen (with a Pt catalyst) Halogenation – the addition of a halogen (e.g. Cl2) Hydrohalogenation – the addition of a hydrogen halide (e.g. HCl) Hydration – the addition of water Oxid ...
... components are added. There are a number of different types of addition reactions: Hydrogenation – the addition of hydrogen (with a Pt catalyst) Halogenation – the addition of a halogen (e.g. Cl2) Hydrohalogenation – the addition of a hydrogen halide (e.g. HCl) Hydration – the addition of water Oxid ...
Physical Properties of Hydrocarbons
... • bigger the molecule greater the intermolecular force • ... more viscous as the intermolecular attractive forces between molecules increases. • ... higher melting point as more vibrational kinetic energy is needed to overcome the intermolecular attractive forces holding the molecules together to fo ...
... • bigger the molecule greater the intermolecular force • ... more viscous as the intermolecular attractive forces between molecules increases. • ... higher melting point as more vibrational kinetic energy is needed to overcome the intermolecular attractive forces holding the molecules together to fo ...
Chapter 8_part 1
... The Rate Determining step Step 1 is the rate determined step Formation of carbocation ...
... The Rate Determining step Step 1 is the rate determined step Formation of carbocation ...
Alkanes
... What is the general formula for an alkene? CnH2n What are cis/trans structural isomers? How do you determine whether a molecule is cis or trans? Cis/trans isomers have different rotations about their double bonds. Cis=biggest chains on same side; trans=biggest chains on opposite sides What does the ...
... What is the general formula for an alkene? CnH2n What are cis/trans structural isomers? How do you determine whether a molecule is cis or trans? Cis/trans isomers have different rotations about their double bonds. Cis=biggest chains on same side; trans=biggest chains on opposite sides What does the ...
Haloalkane

The haloalkanes (also known, as halogenoalkanes or alkyl halides) are a group of chemical compounds derived from alkanes containing one or more halogens. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely used commercially and, consequently, are known under many chemical and commercial names. They are used as flame retardants, fire extinguishants, refrigerants, propellants, solvents, and pharmaceuticals. Subsequent to the widespread use in commerce, many halocarbons have also been shown to be serious pollutants and toxins. For example, the chlorofluorocarbons have been shown to lead to ozone depletion. Methyl bromide is a controversial fumigant. Only haloalkanes which contain chlorine, bromine, and iodine are a threat to the ozone layer, but fluorinated volatile haloalkanes in theory may have activity as greenhouse gases. Methyl iodide, a naturally occurring substance, however, does not have ozone-depleting properties and the United States Environmental Protection Agency has designated the compound a non-ozone layer depleter. For more information, see Halomethane. Haloalkane or alkyl halides are the compounds which have the general formula ″RX″ where R is an alkyl or substituted alkyl group and X is a halogen (F, Cl, Br, I).Haloalkanes have been known for centuries. Chloroethane was produced synthetically in the 15th century. The systematic synthesis of such compounds developed in the 19th century in step with the development of organic chemistry and the understanding of the structure of alkanes. Methods were developed for the selective formation of C-halogen bonds. Especially versatile methods included the addition of halogens to alkenes, hydrohalogenation of alkenes, and the conversion of alcohols to alkyl halides. These methods are so reliable and so easily implemented that haloalkanes became cheaply available for use in industrial chemistry because the halide could be further replaced by other functional groups.While most haloalkanes are human-produced, non-artificial-source haloalkanes do occur on Earth, mostly through enzyme-mediated synthesis by bacteria, fungi, and especially sea macroalgae (seaweeds). More than 1600 halogenated organics have been identified, with bromoalkanes being the most common haloalkanes. Brominated organics in biology range from biologically produced methyl bromide to non-alkane aromatics and unsaturates (indoles, terpenes, acetogenins, and phenols). Halogenated alkanes in land plants are more rare, but do occur, as for example the fluoroacetate produced as a toxin by at least 40 species of known plants. Specific dehalogenase enzymes in bacteria which remove halogens from haloalkanes, are also known.