Introduction to Inorganic Chemistry
... order to achieve its ends. This means that a good chemist is one who not only has a mastery of chemical theory, but also a good knowledge of chemical facts. With such a knowledge, he can direct a trial and error approach to practical problems in the most promising directions. Inorganic Chemistry Org ...
... order to achieve its ends. This means that a good chemist is one who not only has a mastery of chemical theory, but also a good knowledge of chemical facts. With such a knowledge, he can direct a trial and error approach to practical problems in the most promising directions. Inorganic Chemistry Org ...
Teaching with SCIGRESS - Photochemical Dynamics Group
... provides much of the information upon which these predictions are made. From lectures you have learned to use several models to describe molecules. For example Lewis dot structures delineate connectivity and bonding relationships between atoms of a molecule or ion and can predict bond energies and b ...
... provides much of the information upon which these predictions are made. From lectures you have learned to use several models to describe molecules. For example Lewis dot structures delineate connectivity and bonding relationships between atoms of a molecule or ion and can predict bond energies and b ...
File
... forms from the dissociation of HF, there is a high degree of ordering that takes place as water molecules hydrate this small ion. Entropy is considerably more unfavorable for the formation of hydrated F than for the formation of the other hydrated halides. The result of the more unfavorable ΔS° ter ...
... forms from the dissociation of HF, there is a high degree of ordering that takes place as water molecules hydrate this small ion. Entropy is considerably more unfavorable for the formation of hydrated F than for the formation of the other hydrated halides. The result of the more unfavorable ΔS° ter ...
Chem 11 Review Answers - hrsbstaff.ednet.ns.ca
... 34. A certain atom contains 34 protons, 34 electrons, and 45 neutrons. This atom has a mass number of __. a) 34 b) 45 c) 68 d) 79 35. An example of a compound is a) oxygen b) mercury c) salt d) diamond 36. Carbon is classed as an element rather than as a compound because it a) cannot be chemically d ...
... 34. A certain atom contains 34 protons, 34 electrons, and 45 neutrons. This atom has a mass number of __. a) 34 b) 45 c) 68 d) 79 35. An example of a compound is a) oxygen b) mercury c) salt d) diamond 36. Carbon is classed as an element rather than as a compound because it a) cannot be chemically d ...
Introduction to Inorganic Chemistry
... order to achieve its ends. This means that a good chemist is one who not only has a mastery of chemical theory, but also a good knowledge of chemical facts. With such a knowledge, he can direct a trial and error approach to practical problems in the most promising directions. Inorganic Chemistry Org ...
... order to achieve its ends. This means that a good chemist is one who not only has a mastery of chemical theory, but also a good knowledge of chemical facts. With such a knowledge, he can direct a trial and error approach to practical problems in the most promising directions. Inorganic Chemistry Org ...
2 Atoms and Molecules
... of atoms when the atoms are being characterized or when quantitative calculations are done. In fact, the earliest chemists had no way of determining the actual masses of atoms. For this reason, a system was devised that utilized relative or comparative masses for the atoms. These relative masses are ...
... of atoms when the atoms are being characterized or when quantitative calculations are done. In fact, the earliest chemists had no way of determining the actual masses of atoms. For this reason, a system was devised that utilized relative or comparative masses for the atoms. These relative masses are ...
Week 2
... • Because of its obvious usefullness his periodic table was almost universally adopted, and it remains one of the most valuable tools at the chemist’s use. • The only fundemantal difference between todays table and that of his ...
... • Because of its obvious usefullness his periodic table was almost universally adopted, and it remains one of the most valuable tools at the chemist’s use. • The only fundemantal difference between todays table and that of his ...
Novel Class of Heterometallic Cubane and Boride Clusters
... excess of Te powder yielded [(Cp*Mo)2B4TeH5Cl] (2 and 3), [(Cp*Mo)2B4(μ3-OEt)TeH3Cl] (4), and [(Cp*Mo)4B4H4(μ4BH)3] (5) in moderate to good yields. In parallel with the formation of 2−5 compounds [(Cp*Mo)2B4H4Te2]11 and [(Cp*Mo)2B5H9]12 have also been isolated in good yields. The 11B{1H} NMR spectru ...
... excess of Te powder yielded [(Cp*Mo)2B4TeH5Cl] (2 and 3), [(Cp*Mo)2B4(μ3-OEt)TeH3Cl] (4), and [(Cp*Mo)4B4H4(μ4BH)3] (5) in moderate to good yields. In parallel with the formation of 2−5 compounds [(Cp*Mo)2B4H4Te2]11 and [(Cp*Mo)2B5H9]12 have also been isolated in good yields. The 11B{1H} NMR spectru ...
Holt Modern Chemistry Workbook
... A molecular compound is any chemical compound whose simplest units are molecules. In other words, a single molecule of any molecular compound is an individual unit that is capable of existing on its own. A molecule may contain two or more atoms of the same element, as in oxygen. Or, a molecule may c ...
... A molecular compound is any chemical compound whose simplest units are molecules. In other words, a single molecule of any molecular compound is an individual unit that is capable of existing on its own. A molecule may contain two or more atoms of the same element, as in oxygen. Or, a molecule may c ...
Chemistry booklet
... (Molecular structure, intra- and inter- molecular bonding ) INTER-ATOMIC BONDING also termed INTRAMOLECULAR bonding ( between atoms joined in a molecule or ion ) Electronegativity, EN = the power of an atom to attract electrons The larger the value, the greater the electron attracting ability. For t ...
... (Molecular structure, intra- and inter- molecular bonding ) INTER-ATOMIC BONDING also termed INTRAMOLECULAR bonding ( between atoms joined in a molecule or ion ) Electronegativity, EN = the power of an atom to attract electrons The larger the value, the greater the electron attracting ability. For t ...
chemistry - My Study materials – Kumar
... KVS RO Bangalore and respected sir Shri M.K. Kulshreshtha, Retd-AC, KVS RO Chandigarh for their blessings, motivation and encouragement in bringing out this project in such an excellent form. I also extend my special thanks to respected sir Shri. P. S. Raju, Principal, KV Gachibowli, respected madam ...
... KVS RO Bangalore and respected sir Shri M.K. Kulshreshtha, Retd-AC, KVS RO Chandigarh for their blessings, motivation and encouragement in bringing out this project in such an excellent form. I also extend my special thanks to respected sir Shri. P. S. Raju, Principal, KV Gachibowli, respected madam ...
The SimSoup Guide - Chris Gordon
... of electrons that can exist in the shell. Atoms ‘prefer’ to have full electron shells, because such configurations have lower energy than other configurations. Electron sharing involves a pair of electrons being ‘partly in’ a shell belonging to one atom, and ‘partly in’ a shell belonging to the othe ...
... of electrons that can exist in the shell. Atoms ‘prefer’ to have full electron shells, because such configurations have lower energy than other configurations. Electron sharing involves a pair of electrons being ‘partly in’ a shell belonging to one atom, and ‘partly in’ a shell belonging to the othe ...
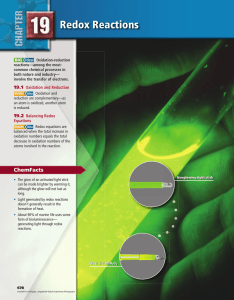
Chapter 19
... involve changes in molecular substances or polyatomic ions in which atoms are covalently bonded to other atoms. For example, the following equation represents the redox reaction used to manufacture ammonia (NH 3). N 2(g) + 3H 2(g) → 2NH 3(g) This process involves neither ions nor any obvious transfe ...
... involve changes in molecular substances or polyatomic ions in which atoms are covalently bonded to other atoms. For example, the following equation represents the redox reaction used to manufacture ammonia (NH 3). N 2(g) + 3H 2(g) → 2NH 3(g) This process involves neither ions nor any obvious transfe ...
Table of Contents Pages Unit 1- Matter and Change 1
... Matter is anything that takes up __________________ and has mass. ______________ is the measure of the amount of matter that an object contains. Virtually all of the matter around us consists of mixtures. A mixture can be defined as something that has _____________________ composition. Soda is a mix ...
... Matter is anything that takes up __________________ and has mass. ______________ is the measure of the amount of matter that an object contains. Virtually all of the matter around us consists of mixtures. A mixture can be defined as something that has _____________________ composition. Soda is a mix ...
Class-XII, Summer assignment
... 2. PH3 has lower boiling point than NH3. Why? Ans: Unlike NH3, PH3 molecules are not associated through hydrogen bonding in liquid state. That is why the boiling point of PH3 is lower than NH3. 3. Why are pentahalides more covalent than trihalides ? Ans: Higher the positive oxidation state of centra ...
... 2. PH3 has lower boiling point than NH3. Why? Ans: Unlike NH3, PH3 molecules are not associated through hydrogen bonding in liquid state. That is why the boiling point of PH3 is lower than NH3. 3. Why are pentahalides more covalent than trihalides ? Ans: Higher the positive oxidation state of centra ...
Table of Contents
... ___________________ ___________________ and dust free air (mixture of nitrogen, oxygen, argon, carbon dioxide, water vapor and other gases) are examples of homogeneous mixtures. Brass (solid mixture of copper and ______________) is also a homogeneous mixture. Brass is a(n) _________________, which i ...
... ___________________ ___________________ and dust free air (mixture of nitrogen, oxygen, argon, carbon dioxide, water vapor and other gases) are examples of homogeneous mixtures. Brass (solid mixture of copper and ______________) is also a homogeneous mixture. Brass is a(n) _________________, which i ...
Has the Periodic Table Been Successfully Axiomatized?
... In fact, at no point in the evolution of Mendeleev’s periodic tables, over half of which were published,8 did Mendeleev ever adopt such a view (van Spronsen, 1969). If one considers the group of alkali metals, for example, and one asks the question of how many elements occur before the repetition of ...
... In fact, at no point in the evolution of Mendeleev’s periodic tables, over half of which were published,8 did Mendeleev ever adopt such a view (van Spronsen, 1969). If one considers the group of alkali metals, for example, and one asks the question of how many elements occur before the repetition of ...
101
... not represent an ionic charge, because the elements are not present as ions. However, to assign oxidation numbers to the elements in a covalent molecule or polyatomic ion, you can pretend the bonds are ionic. ...
... not represent an ionic charge, because the elements are not present as ions. However, to assign oxidation numbers to the elements in a covalent molecule or polyatomic ion, you can pretend the bonds are ionic. ...
Subject Area Standard Area Organizing Category Grade Level
... CHEM.B.2.2.1: Utilize mathematical relationships to predict changes in the number of particles, the temperature, the pressure, and the volume in a gaseous system (i.e., Boyle’s law, Charles’s law, Dalton’s law of partial pressures, the combined gas law, and the ideal gas law). ...
... CHEM.B.2.2.1: Utilize mathematical relationships to predict changes in the number of particles, the temperature, the pressure, and the volume in a gaseous system (i.e., Boyle’s law, Charles’s law, Dalton’s law of partial pressures, the combined gas law, and the ideal gas law). ...
COURSE STRUCTURE
... (4) Magnetic spin quantum number (ms) It can take the values of +½ or ½ and determines the orientation of spin. Pauli’s Exclusion Principle : ‘‘No two electrons in an atom can have the same set of four quantum numbers.’’ Two electrons can have same values for n, l and ml provided their spins are op ...
... (4) Magnetic spin quantum number (ms) It can take the values of +½ or ½ and determines the orientation of spin. Pauli’s Exclusion Principle : ‘‘No two electrons in an atom can have the same set of four quantum numbers.’’ Two electrons can have same values for n, l and ml provided their spins are op ...
23. Oxidation and Reduction
... 1. The oxidation number of an atom of a free element is zero. Elements are free if they are not combined with other elements. If atoms of an element are combined with themselves, they are still considered to be free. For example, a free atom of Ag has an oxidation number of zero. In addition, atoms ...
... 1. The oxidation number of an atom of a free element is zero. Elements are free if they are not combined with other elements. If atoms of an element are combined with themselves, they are still considered to be free. For example, a free atom of Ag has an oxidation number of zero. In addition, atoms ...
AP Chemistry Curriculum Map - Belle Vernon Area School District
... properties of atoms allow for the prediction of physical and chemical properties. Eligible Content CHEM.A.2.3.1 – Explain how the periodicity of chemical properties led to the arrangement of elements on the periodic table. Compare and/or predict the properties (e.g., electron affinity, ionizatio ...
... properties of atoms allow for the prediction of physical and chemical properties. Eligible Content CHEM.A.2.3.1 – Explain how the periodicity of chemical properties led to the arrangement of elements on the periodic table. Compare and/or predict the properties (e.g., electron affinity, ionizatio ...
enjoy chemistry
... Ans:Halogens are coloured. This is due to absorption of radiations in visible region which results in the excitation of outer electrons to higher energy level. By absorbing different quanta of radiation, they display different colours. For example, F2, has yellow, Cl2 , greenish yellow, Br2, red and ...
... Ans:Halogens are coloured. This is due to absorption of radiations in visible region which results in the excitation of outer electrons to higher energy level. By absorbing different quanta of radiation, they display different colours. For example, F2, has yellow, Cl2 , greenish yellow, Br2, red and ...
PDF on arxiv.org - at www.arxiv.org.
... predicting chemical properties. While many twentieth century bonding models provide useful information for a variety of chemical systems, these models are sometimes less insightful for more lofty goals such as designing metalloenzymes. The design process of novel catalysts could be improved if more ...
... predicting chemical properties. While many twentieth century bonding models provide useful information for a variety of chemical systems, these models are sometimes less insightful for more lofty goals such as designing metalloenzymes. The design process of novel catalysts could be improved if more ...
chemistry writing team
... For a given value of ‘l’, ml has a total of (2l + 1) values ranging from −l to +l including ‘0’. It determines the orientation of orbital. (4) Magnetic spin quantum number (ms) It can take the values of +½ or −½ and determines the orientation of spin. Pauli’s Exclusion Principle : ‘‘No two electrons ...
... For a given value of ‘l’, ml has a total of (2l + 1) values ranging from −l to +l including ‘0’. It determines the orientation of orbital. (4) Magnetic spin quantum number (ms) It can take the values of +½ or −½ and determines the orientation of spin. Pauli’s Exclusion Principle : ‘‘No two electrons ...
Electronegativity

Electronegativity, symbol χ, is a chemical property that describes the tendency of an atom or a functional group to attract electrons (or electron density) towards itself. An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity number, the more an element or compound attracts electrons towards it. The term ""electronegativity"" was introduced by Jöns Jacob Berzelius in 1811,though the concept was known even before that and was studied by many chemists including Avogadro.In spite of its long history, an accurate scale of electronegativity had to wait till 1932, when Linus Pauling proposed an electronegativity scale, which depends on bond energies, as a development of valence bond theory. It has been shown to correlate with a number of other chemical properties. Electronegativity cannot be directly measured and must be calculated from other atomic or molecular properties. Several methods of calculation have been proposed, and although there may be small differences in the numerical values of the electronegativity, all methods show the same periodic trends between elements. The most commonly used method of calculation is that originally proposed by Linus Pauling. This gives a dimensionless quantity, commonly referred to as the Pauling scale, on a relative scale running from around 0.7 to 3.98 (hydrogen = 2.20). When other methods of calculation are used, it is conventional (although not obligatory) to quote the results on a scale that covers the same range of numerical values: this is known as an electronegativity in Pauling units. As it is usually calculated, electronegativity is not a property of an atom alone, but rather a property of an atom in a molecule. Properties of a free atom include ionization energy and electron affinity. It is to be expected that the electronegativity of an element will vary with its chemical environment, but it is usually considered to be a transferable property, that is to say that similar values will be valid in a variety of situations.On the most basic level, electronegativity is determined by factors like the nuclear charge (the more protons an atom has, the more ""pull"" it will have on electrons) and the number/location of other electrons present in the atomic shells (the more electrons an atom has, the farther from the nucleus the valence electrons will be, and as a result the less positive charge they will experience—both because of their increased distance from the nucleus, and because the other electrons in the lower energy core orbitals will act to shield the valence electrons from the positively charged nucleus).The opposite of electronegativity is electropositivity: a measure of an element's ability to donate electrons.Caesium is the least electronegative element in the periodic table (=0.79), while fluorine is most electronegative (=3.98). (Francium and caesium were originally assigned both assigned 0.7; caesium's value was later refined to 0.79, but no experimental data allows a similar refinement for francium. However, francium's ionization energy is known to be slightly higher than caesium's, in accordance with the relativistic stabilization of the 7s orbital, and this in turn implies that caesium is in fact more electronegative than francium.)