Supplementary Notes on Volumetric Analysis
... (c) Draw an arrow on the diagram clearly to show the transition corresponding to the ionization energy of atomic hydrogen in its most stable state. ...
... (c) Draw an arrow on the diagram clearly to show the transition corresponding to the ionization energy of atomic hydrogen in its most stable state. ...
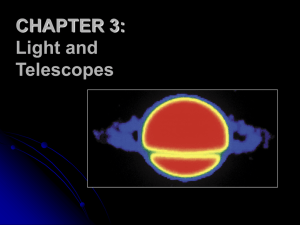
Light and the Electromagnetic Spectrum
... Violet light, at the other end of the visible range, has nearly double the frequency—7.5 × 1014 Hz—and (since the speed of light is the same in either case) just over half the wavelength— 4.0 × 107 m (400nm). ...
... Violet light, at the other end of the visible range, has nearly double the frequency—7.5 × 1014 Hz—and (since the speed of light is the same in either case) just over half the wavelength— 4.0 × 107 m (400nm). ...
A Student Introduction to Solar Energy
... The working principle of solar cells is based on the photovoltaic effect, i.e. the generation of a potential difference at the junction of two different materials in response to electromagnetic radiation. The photovoltaic effect is closely related to the photoelectric effect, where electrons are emi ...
... The working principle of solar cells is based on the photovoltaic effect, i.e. the generation of a potential difference at the junction of two different materials in response to electromagnetic radiation. The photovoltaic effect is closely related to the photoelectric effect, where electrons are emi ...
Unit 8 Heat Study Guide A change of state is a ___ Process by
... 20. Heat is the __________ of __________ between objects of __________ temperatures. 21. If two different substances have the same temperature, then ____ a. They have the same total kinetic energy b. They have the same state of matter c. Their specific heat is the same d. None of the above 22. In wh ...
... 20. Heat is the __________ of __________ between objects of __________ temperatures. 21. If two different substances have the same temperature, then ____ a. They have the same total kinetic energy b. They have the same state of matter c. Their specific heat is the same d. None of the above 22. In wh ...
Unit 3 - Periodic Trends and Spectroscopy Test Review
... 3. Use knowledge of the properties of light, the electromagnetic spectrum, and the structure of the Bohr model of the atom to analyze the relationships between energetic transitions of electrons, wavelength, frequency, and energy. 4. If given c=λν, E=hν, c = 3.00x108 m/s, and h = 6.626x10-34 Js, be ...
... 3. Use knowledge of the properties of light, the electromagnetic spectrum, and the structure of the Bohr model of the atom to analyze the relationships between energetic transitions of electrons, wavelength, frequency, and energy. 4. If given c=λν, E=hν, c = 3.00x108 m/s, and h = 6.626x10-34 Js, be ...