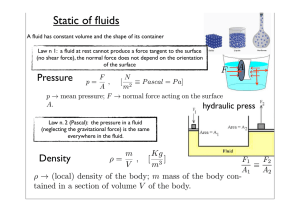
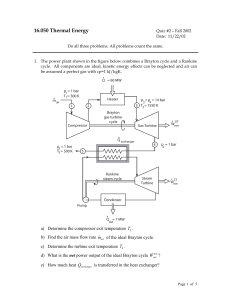
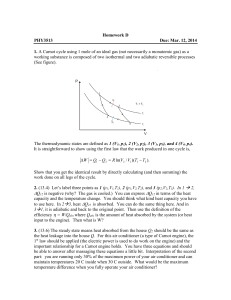
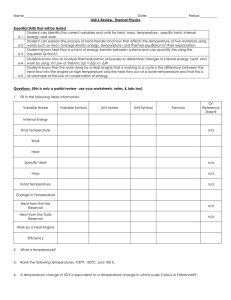
FE Review Common Pitfalls in Thermodynamics
... This result is correct only when the process is constant pressure, and P = P1 = P2. 8. Temperature—When the value of temperature is substituted into an equation such as the ideal equation of state, the temperature must be in absolute units. A temperature change in the ordinary units, celsius or fahr ...
... This result is correct only when the process is constant pressure, and P = P1 = P2. 8. Temperature—When the value of temperature is substituted into an equation such as the ideal equation of state, the temperature must be in absolute units. A temperature change in the ordinary units, celsius or fahr ...
The Laws of Thermodynamics
... dS is independent of the path. Some text books refer to dS therefore as being an exact differential. In general the change in entropy between any two arbitrary states depends only on those states and not on the path between them. We can write an infinitesimal version of the first law informally as d ...
... dS is independent of the path. Some text books refer to dS therefore as being an exact differential. In general the change in entropy between any two arbitrary states depends only on those states and not on the path between them. We can write an infinitesimal version of the first law informally as d ...
T - Massey University
... The two forms of energy that influence this internal energy are heat, either transferred to the system from a source at higher temperature or lost to a sink at lower temperature and work, which can increase the internal energy if work is done on the system by its surroundings, or decrease the intern ...
... The two forms of energy that influence this internal energy are heat, either transferred to the system from a source at higher temperature or lost to a sink at lower temperature and work, which can increase the internal energy if work is done on the system by its surroundings, or decrease the intern ...
Chemistry and the material world
... the adiabatic path and w for the non-adiabatic path. q = wad – w Finally, from the first law of thermodynamics also follows that the internal energy of an isolated system cannot change. Because for an isolated system there is w = 0 and q = 0 and with ΔU = q + w it follows that ΔU = 0. The state of a ...
... the adiabatic path and w for the non-adiabatic path. q = wad – w Finally, from the first law of thermodynamics also follows that the internal energy of an isolated system cannot change. Because for an isolated system there is w = 0 and q = 0 and with ΔU = q + w it follows that ΔU = 0. The state of a ...
v = Y
... reservoir when heat is discarded into it (TC). ◦ Any finite temperature drop would result in an irreversible processes. ◦ Every process that involves heat transfer must be isothermal. ◦ Any process in which the the working substance is between TH and TC, there must be no heat transfer into the hot o ...
... reservoir when heat is discarded into it (TC). ◦ Any finite temperature drop would result in an irreversible processes. ◦ Every process that involves heat transfer must be isothermal. ◦ Any process in which the the working substance is between TH and TC, there must be no heat transfer into the hot o ...
U / ∂V
... object. By defining the calorie, as a heat unit, as the amount of heat required to raise the temperature of one gram of water from 14.5° c to 15.5° c it was found that: 1calorie = 4.184 joule This constant, 4.184, is known as the mechanical equivalent of heat ...
... object. By defining the calorie, as a heat unit, as the amount of heat required to raise the temperature of one gram of water from 14.5° c to 15.5° c it was found that: 1calorie = 4.184 joule This constant, 4.184, is known as the mechanical equivalent of heat ...
lecture21
... restriction on direction. A process will not occur unless it satisfies both the first and second laws of thermodynamics. Second law not only identifies the direction of process, it also asserts that energy has quality as well as quantity. Thermal Reservoir A thermal reservoir is a large system (very ...
... restriction on direction. A process will not occur unless it satisfies both the first and second laws of thermodynamics. Second law not only identifies the direction of process, it also asserts that energy has quality as well as quantity. Thermal Reservoir A thermal reservoir is a large system (very ...
Objectives Recognize that a system can absorb or release energy
... o If the internal energy for the roller coaster (the system) and the energy dissipated to the surrounding air (the environment) are taken into account, then ...
... o If the internal energy for the roller coaster (the system) and the energy dissipated to the surrounding air (the environment) are taken into account, then ...
Thermodynamics - SeyedAhmad.com
... • The internal energy U of a system is the total of all kinds of energy possessed by the particles that make up the system. Usually the internal energy consists of the sum of the potential and kinetic energies of the working gas molecules. ...
... • The internal energy U of a system is the total of all kinds of energy possessed by the particles that make up the system. Usually the internal energy consists of the sum of the potential and kinetic energies of the working gas molecules. ...
First Law of Thermodynamics
... of a system’s thermal energy can be converted to other forms of energy. Thermodynamic variables describe the state of the thermal system and the equation of state relates these variables. If we draw diagrams of p − V or p − T or T − V , the changes in the state of the system can be represented as cu ...
... of a system’s thermal energy can be converted to other forms of energy. Thermodynamic variables describe the state of the thermal system and the equation of state relates these variables. If we draw diagrams of p − V or p − T or T − V , the changes in the state of the system can be represented as cu ...
Heat

In physics, heat is energy in a process of transfer between a system and its surroundings, other than as work or with the transfer of matter. When there is a suitable physical pathway, heat flows from a hotter body to a colder one. The pathway can be direct, as in conduction and radiation, or indirect, as in convective circulation.Because it refers to a process of transfer between two systems, the system of interest, and its surroundings considered as a system, heat is not a state or property of a single system. If heat transfer is slow and continuous, so that the temperature of the system of interest remains well defined, it can sometimes be described by a process function.Kinetic theory explains heat as a macroscopic manifestation of the motions and interactions of microscopic constituents such as molecules and photons.In calorimetry, sensible heat is defined with respect to a specific chosen state variable of the system, such as pressure or volume. Sensible heat transferred into or out of the system under study causes change of temperature while leaving the chosen state variable unchanged. Heat transfer that occurs with the system at constant temperature and that does change that particular state variable is called latent heat with respect to that variable. For infinitesimal changes, the total incremental heat transfer is then the sum of the latent and sensible heat increments. This is a basic paradigm for thermodynamics, and was important in the historical development of the subject.The quantity of energy transferred as heat is a scalar expressed in an energy unit such as the joule (J) (SI), with a sign that is customarily positive when a transfer adds to the energy of a system. It can be measured by calorimetry, or determined by calculations based on other quantities, relying on the first law of thermodynamics.