Solubility and Complex-ion Equilibria
... but dissolve in acidic solution. – Qualitative analysis uses this change in solubility of the metal sulfides with pH to separate a mixture of metal ions. – By adjusting the pH in an aqueous solution of H2S, you adjust the sulfide concentration to precipitate the least soluble metal sulfide first. – ...
... but dissolve in acidic solution. – Qualitative analysis uses this change in solubility of the metal sulfides with pH to separate a mixture of metal ions. – By adjusting the pH in an aqueous solution of H2S, you adjust the sulfide concentration to precipitate the least soluble metal sulfide first. – ...
The Determination of the Location of Contact Electrification-Induced Discharge Events
... the contacting metal developed the charge of the mobile counterion (Figure 1a). In previous work,30 we showed that the agitation of poly(styrene) microspheres functionalized with covalently bound, positively charged groups (with mobile negative counterions) developed a positive charge when they char ...
... the contacting metal developed the charge of the mobile counterion (Figure 1a). In previous work,30 we showed that the agitation of poly(styrene) microspheres functionalized with covalently bound, positively charged groups (with mobile negative counterions) developed a positive charge when they char ...
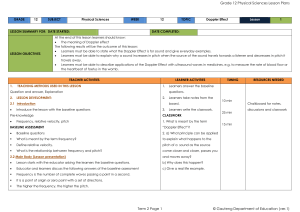
Physical Sciences Grade 12 Term 2
... The siren of a burglar alarm system has a frequency of 960 Hz. During a patrol, a security officer, travelling in his car, hears the siren of the alarm of a house and approaches the house at 2.1 Name the phenomenon that explains the change in the observed frequency. 2.2 Calculate the speed at which ...
... The siren of a burglar alarm system has a frequency of 960 Hz. During a patrol, a security officer, travelling in his car, hears the siren of the alarm of a house and approaches the house at 2.1 Name the phenomenon that explains the change in the observed frequency. 2.2 Calculate the speed at which ...
Chap18 - Bakersfield College
... but dissolve in acidic solution. – Qualitative analysis uses this change in solubility of the metal sulfides with pH to separate a mixture of metal ions. – By adjusting the pH in an aqueous solution of H2S, you adjust the sulfide concentration to precipitate the least soluble metal sulfide first. – ...
... but dissolve in acidic solution. – Qualitative analysis uses this change in solubility of the metal sulfides with pH to separate a mixture of metal ions. – By adjusting the pH in an aqueous solution of H2S, you adjust the sulfide concentration to precipitate the least soluble metal sulfide first. – ...
Unfamiliar Oxidation States and Tkeir Stabilization
... known. Most of the known compounds of dipositive silver and of tripositive copper and silver are complex in nature. T h e preparation of element 85, astatine, by the bombardment of bismuth of mass 209 with 32 m.e.v. alpha particles, and the subsequent observations on its chemical properties, 5 have ...
... known. Most of the known compounds of dipositive silver and of tripositive copper and silver are complex in nature. T h e preparation of element 85, astatine, by the bombardment of bismuth of mass 209 with 32 m.e.v. alpha particles, and the subsequent observations on its chemical properties, 5 have ...
Solubility and Complex-ion Equilibria
... but dissolve in acidic solution. – Qualitative analysis uses this change in solubility of the metal sulfides with pH to separate a mixture of metal ions. – By adjusting the pH in an aqueous solution of H2S, you adjust the sulfide concentration to precipitate the least soluble metal sulfide first. – ...
... but dissolve in acidic solution. – Qualitative analysis uses this change in solubility of the metal sulfides with pH to separate a mixture of metal ions. – By adjusting the pH in an aqueous solution of H2S, you adjust the sulfide concentration to precipitate the least soluble metal sulfide first. – ...
LABORATORY MANUAL CHEMISTRY 121 2013
... which describes the system more fully." Or, "When two elements combine the product has properties which are different from either of the reactants. This indicates that a new compound has been formed." DO NOT repeat your results or data here. There is always a conclusion that can be formed from your ...
... which describes the system more fully." Or, "When two elements combine the product has properties which are different from either of the reactants. This indicates that a new compound has been formed." DO NOT repeat your results or data here. There is always a conclusion that can be formed from your ...
Solubility
... [CrO42-] = 0.01M, [Ag+] = 1.0x10-5 (calculated previously) Ksp = [Ag+][Br-] = (1.0x10-5M)[Br-] = 5.0x10-13 [Br-] = 5.0x10-8M So if we stop the addition of AgNO3 just before Ag2CrO4 starts to precipitate... [Br-] drops from 0.01M to 5.0x10-8M. What % of Br- is left? 5.0x10-8M / 1.0x10-2M *100% ...
... [CrO42-] = 0.01M, [Ag+] = 1.0x10-5 (calculated previously) Ksp = [Ag+][Br-] = (1.0x10-5M)[Br-] = 5.0x10-13 [Br-] = 5.0x10-8M So if we stop the addition of AgNO3 just before Ag2CrO4 starts to precipitate... [Br-] drops from 0.01M to 5.0x10-8M. What % of Br- is left? 5.0x10-8M / 1.0x10-2M *100% ...
Document
... • A battery creates a fixed potential difference using chemical processes. • All batteries use chemical reactions to create internal charge separation. ...
... • A battery creates a fixed potential difference using chemical processes. • All batteries use chemical reactions to create internal charge separation. ...
Ch 21 - Keene ISD
... • A battery creates a fixed potential difference using chemical processes. • All batteries use chemical reactions to create internal charge separation. • In biology, chemicals produce a potential difference of about 70 mV between the inside and the outside of a cell, with inside the cell more negati ...
... • A battery creates a fixed potential difference using chemical processes. • All batteries use chemical reactions to create internal charge separation. • In biology, chemicals produce a potential difference of about 70 mV between the inside and the outside of a cell, with inside the cell more negati ...
Soluble - HCC Learning Web
... How does the solubility of silver phosphate in water compare to that of silver phosphate in an acidic solution (made by adding nitric acid to the solution)? Explain. The silver phosphate is more soluble in an acidic solution. Copyright © Cengage Learning. All rights reserved ...
... How does the solubility of silver phosphate in water compare to that of silver phosphate in an acidic solution (made by adding nitric acid to the solution)? Explain. The silver phosphate is more soluble in an acidic solution. Copyright © Cengage Learning. All rights reserved ...
III. Polyelectrolyte Phenomena (Dautzenberg et al., Polyelectrolytes
... c and that electrostatic interactions between chains influence significantly at unexpectedly low c, in a c regime where chain interactions are conventionally thought to be relative weak. Competing, nonconfiguration-based models for polyelectrolyte viscosity also have appeared, motivated largely by ...
... c and that electrostatic interactions between chains influence significantly at unexpectedly low c, in a c regime where chain interactions are conventionally thought to be relative weak. Competing, nonconfiguration-based models for polyelectrolyte viscosity also have appeared, motivated largely by ...
Complete Solution Manual
... a. Cathode: The electrode at which reduction occurs. b. Anode: The electrode at which oxidation occurs. c. Oxidation half-reaction: The half-reaction in which electrons are products. In a galvanic cell, the oxidation half-reaction always occurs at the anode. d. Reduction half-reaction: The half-reac ...
... a. Cathode: The electrode at which reduction occurs. b. Anode: The electrode at which oxidation occurs. c. Oxidation half-reaction: The half-reaction in which electrons are products. In a galvanic cell, the oxidation half-reaction always occurs at the anode. d. Reduction half-reaction: The half-reac ...
Chapter 15 - Cengage Learning
... 15. In this problem we are asked to calculate how much of a concentrated stock solution, which is 12 M HCl, is needed to prepare a dilute HCl solution. We will need to know how many moles of HCl are present in 2.5 L of 1.0 M HCl, that is, in the dilute solution. Then we need to find a volume of the ...
... 15. In this problem we are asked to calculate how much of a concentrated stock solution, which is 12 M HCl, is needed to prepare a dilute HCl solution. We will need to know how many moles of HCl are present in 2.5 L of 1.0 M HCl, that is, in the dilute solution. Then we need to find a volume of the ...
Complete Solution Manual
... a. Cathode: The electrode at which reduction occurs. b. Anode: The electrode at which oxidation occurs. c. Oxidation half-reaction: The half-reaction in which electrons are products. In a galvanic cell, the oxidation half-reaction always occurs at the anode. d. Reduction half-reaction: The half-reac ...
... a. Cathode: The electrode at which reduction occurs. b. Anode: The electrode at which oxidation occurs. c. Oxidation half-reaction: The half-reaction in which electrons are products. In a galvanic cell, the oxidation half-reaction always occurs at the anode. d. Reduction half-reaction: The half-reac ...
History of electrochemistry
Electrochemistry, a branch of chemistry, went through several changes during its evolution from early principles related to magnets in the early 16th and 17th centuries, to complex theories involving conductivity, electric charge and mathematical methods. The term electrochemistry was used to describe electrical phenomena in the late 19th and 20th centuries. In recent decades, electrochemistry has become an area of current research, including research in batteries and fuel cells, preventing corrosion of metals, the use of electrochemical cells to remove refractory organics and similar contaminants in wastewater electrocoagulation and improving techniques in refining chemicals with electrolysis and electrophoresis.