4 ORGANIC CHEMISTRY: STRUCTURE AND NOMENCLATURE
... When the unmanned Viking spacecraft carried out experiments designed to search for evidence of life on Mars, the experiments were based on the assumption that living systems contain carbon, and the absence of any evidence for carbon-based life on that planet was presumed to mean that no life existed ...
... When the unmanned Viking spacecraft carried out experiments designed to search for evidence of life on Mars, the experiments were based on the assumption that living systems contain carbon, and the absence of any evidence for carbon-based life on that planet was presumed to mean that no life existed ...
proline2 - Department of Mathematics and Statistics
... curves of C-C-N vs. 2 (Figure 4c) and C-C-C vs. 2 (Figure 4b). For the C-C-C variation, we recalculate the cyclic curves where we fix the C-C-N angle instead of the C-C-C angle, and place the apex of the tetrahedron at the N atom. We thus obtain the curve of C-C-C vs. 2 (Figure 4a ...
... curves of C-C-N vs. 2 (Figure 4c) and C-C-C vs. 2 (Figure 4b). For the C-C-C variation, we recalculate the cyclic curves where we fix the C-C-N angle instead of the C-C-C angle, and place the apex of the tetrahedron at the N atom. We thus obtain the curve of C-C-C vs. 2 (Figure 4a ...
File
... production; HNO3: important industrial chemical, used to form nitrogen-based explosives, strong acid and a very strong oxidizing agent. ...
... production; HNO3: important industrial chemical, used to form nitrogen-based explosives, strong acid and a very strong oxidizing agent. ...
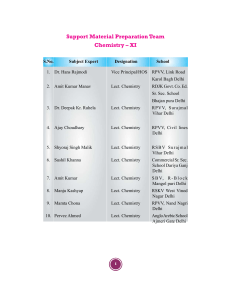
Support Material
... Signi®cance of classi® cation, brief history of the development of periodic table, modern periodic law and the present from of periodic trends in properties of elements : atomic radii, inert gas fadii, lonization enthalpy, electron gain enthalpy, electronegativity, valency. Nomenclature of elements ...
... Signi®cance of classi® cation, brief history of the development of periodic table, modern periodic law and the present from of periodic trends in properties of elements : atomic radii, inert gas fadii, lonization enthalpy, electron gain enthalpy, electronegativity, valency. Nomenclature of elements ...
PDF on arxiv.org - at www.arxiv.org.
... more lofty goals such as designing metalloenzymes. The design process of novel catalysts could be improved if more predictive and accurate models of chemical bonding are created. One recently developed bonding model based on the topology of the electron charge density is the quantum theory of atoms ...
... more lofty goals such as designing metalloenzymes. The design process of novel catalysts could be improved if more predictive and accurate models of chemical bonding are created. One recently developed bonding model based on the topology of the electron charge density is the quantum theory of atoms ...
Chemistry - talcher autonomous college
... application, Solvation energy. (ii) Covalent bond: Lewis structure, Valence Bond theory (Heitler-London approach). Energetics of hybridization, equivalent and non-equivalent hybrid orbitals. Bent’s rule, Resonance and resonance energy, Molecular orbital theory. Molecular orbital diagrams of diatomic ...
... application, Solvation energy. (ii) Covalent bond: Lewis structure, Valence Bond theory (Heitler-London approach). Energetics of hybridization, equivalent and non-equivalent hybrid orbitals. Bent’s rule, Resonance and resonance energy, Molecular orbital theory. Molecular orbital diagrams of diatomic ...
Atomic Polar Tensor Transferabllity and Atomic Charges kr the
... in ref 1. (RtY)represents the center of charge of the h brid orbital (pv),where p and v indicate orbitals of atom A, and R,,YB represents the bonding center of charge since p and v belong to different atoms, A and B, whether chemically bonded or not. These contributions in expression 1 are known, re ...
... in ref 1. (RtY)represents the center of charge of the h brid orbital (pv),where p and v indicate orbitals of atom A, and R,,YB represents the bonding center of charge since p and v belong to different atoms, A and B, whether chemically bonded or not. These contributions in expression 1 are known, re ...
114
... O2reactions. Furthermore, the amphoionic structures were estimated to be significantly higher in energy than the peroxy diradical.7c Since only intermediate minima (and no saddle points) were studied for the formation of peroxirane and dioxetane, the problem of the pathway actually followed by the s ...
... O2reactions. Furthermore, the amphoionic structures were estimated to be significantly higher in energy than the peroxy diradical.7c Since only intermediate minima (and no saddle points) were studied for the formation of peroxirane and dioxetane, the problem of the pathway actually followed by the s ...
chapter 23 the transition elements and their
... Oxides of transition metals become less basic (or more acidic) as oxidation state increases. The oxidation state of chromium in CrO3 is +6 and in CrO is +2, based on the –2 oxidation state of oxygen. The oxide of the higher oxidation state, CrO3, produces a more acidic solution. ...
... Oxides of transition metals become less basic (or more acidic) as oxidation state increases. The oxidation state of chromium in CrO3 is +6 and in CrO is +2, based on the –2 oxidation state of oxygen. The oxide of the higher oxidation state, CrO3, produces a more acidic solution. ...
Fluorinated Butatrienes - diss.fu-berlin.de
... wurde ein Enin-Isomer entdeckt, das erstaunlicherweise stabiler als sein Butatrien Isomer ist, obwohl es an der Dreifachbindung fluoriert ist. Eben jene Fluorierung an der Dreifachbindung ist eigentlich notwendig um die Energie fluorierter But-1-en-3-ine relativ ...
... wurde ein Enin-Isomer entdeckt, das erstaunlicherweise stabiler als sein Butatrien Isomer ist, obwohl es an der Dreifachbindung fluoriert ist. Eben jene Fluorierung an der Dreifachbindung ist eigentlich notwendig um die Energie fluorierter But-1-en-3-ine relativ ...
chemistry - Textbooks Online
... In using the term mole for ionic substances, we mean the number of formula units of the substance. For example, a mole of sodium carbonate, Na2CO3 is a quantity containing 6.023 x 1023 Na2CO3 units. But each formula unit of Na2CO3 contains 2 x 6.023 x 1023 Na+ ions and one CO32ions and 1 x 6.023 x 1 ...
... In using the term mole for ionic substances, we mean the number of formula units of the substance. For example, a mole of sodium carbonate, Na2CO3 is a quantity containing 6.023 x 1023 Na2CO3 units. But each formula unit of Na2CO3 contains 2 x 6.023 x 1023 Na+ ions and one CO32ions and 1 x 6.023 x 1 ...
CHAPTER 23 THE TRANSITION ELEMENTS AND THEIR
... chromium in CrO3 is +6 and in CrO is +2, based on the -2 oxidation state of oxygen. The oxide of the higher oxidation state, CrO3, produces a more acidic solution. ...
... chromium in CrO3 is +6 and in CrO is +2, based on the -2 oxidation state of oxygen. The oxide of the higher oxidation state, CrO3, produces a more acidic solution. ...
Advanced Chemistry
... Solution The value 4.0 has two significant figures, whereas 4.00 has three. This difference implies that the 4.0 has more uncertainty. A mass reported as 4.0 g indicates that the uncertainty is in the first decimal place. Thus, the mass might be anything between 3.9 and 4.1 g, which we can represent ...
... Solution The value 4.0 has two significant figures, whereas 4.00 has three. This difference implies that the 4.0 has more uncertainty. A mass reported as 4.0 g indicates that the uncertainty is in the first decimal place. Thus, the mass might be anything between 3.9 and 4.1 g, which we can represent ...
Teaching with CAChe - Photochemical Dynamics Group
... relationships between atoms of a molecule or ion and can predict bond energies and bond lengths. However, Lewis dot structures do not predict shape. The most basic theory that predicts geometry is the valence-shell electron-pair repulsion (VSEPR) model. The VSEPR model is based on O the premise that ...
... relationships between atoms of a molecule or ion and can predict bond energies and bond lengths. However, Lewis dot structures do not predict shape. The most basic theory that predicts geometry is the valence-shell electron-pair repulsion (VSEPR) model. The VSEPR model is based on O the premise that ...
Organic Molecules
... In organic chemistry, a functional group is a speci c group of atoms within molecules, that are responsible for the characteristic chemical reactions of those molecules. The same functional group will undergo the same or similar chemical reaction(s) regardless of the size of the molecule it is a par ...
... In organic chemistry, a functional group is a speci c group of atoms within molecules, that are responsible for the characteristic chemical reactions of those molecules. The same functional group will undergo the same or similar chemical reaction(s) regardless of the size of the molecule it is a par ...
STUDY GUIDE
... cotton, and wood are all composed of polymers made by living organisms. Polymers are large molecules—natural or synthetic—made up of many monomers linked together. Homopolymers are polymers made of only a single type of monomer. Copolymers are polymers made of two or more types of monomers. Addition ...
... cotton, and wood are all composed of polymers made by living organisms. Polymers are large molecules—natural or synthetic—made up of many monomers linked together. Homopolymers are polymers made of only a single type of monomer. Copolymers are polymers made of two or more types of monomers. Addition ...
P-BLOCK ELEMENTS
... (iii) Ionization Enthalpy: The ionization enthalpy value decreases down the group. But, decrease is less than that expected, this is because, the d and f electrons which have very poor screening effect is unable to compensate the increase in nuclear charge. The ionisation enthalpy decreases from B t ...
... (iii) Ionization Enthalpy: The ionization enthalpy value decreases down the group. But, decrease is less than that expected, this is because, the d and f electrons which have very poor screening effect is unable to compensate the increase in nuclear charge. The ionisation enthalpy decreases from B t ...
SED122 - National Open University of Nigeria
... we use everyday, directly or indirectly are products of chemical research and examples of useful products of chemical reactions are limitless. What then is matter? Matter is anything that has mass and occupies space. Matter is classified into solid, liquid and gas. ...
... we use everyday, directly or indirectly are products of chemical research and examples of useful products of chemical reactions are limitless. What then is matter? Matter is anything that has mass and occupies space. Matter is classified into solid, liquid and gas. ...
CHAPTER 19 TRANSITION METALS AND COORDINATION
... Chromium ([Ar]4s03d5) and copper ([Ar]4s13d10) have electron configurations that are different from that predicted from the periodic table. Other exceptions to the predicted filling order are transition metal ions. These all lose the s electrons before they lose the d electrons. In neutral atoms, th ...
... Chromium ([Ar]4s03d5) and copper ([Ar]4s13d10) have electron configurations that are different from that predicted from the periodic table. Other exceptions to the predicted filling order are transition metal ions. These all lose the s electrons before they lose the d electrons. In neutral atoms, th ...
KHOA: HÓA HỌC - CCS - Trường Đại học Sư phạm Hà Nội
... element is a substance comprised of a single type of atom. The elements are the building blocks of our nature. An element is either discovered in nature or synthesized in the laboratory in pure form that cannot be separated into simpler substances by chemical methods. Currently, there are about 118 ...
... element is a substance comprised of a single type of atom. The elements are the building blocks of our nature. An element is either discovered in nature or synthesized in the laboratory in pure form that cannot be separated into simpler substances by chemical methods. Currently, there are about 118 ...
chemistry writing team
... for n, l and ml provided their spins are opposite (ms is different). An orbital can have at the most two electrons if they have opporite spins. Hund’s Rule of maximum Multiplicity : ‘‘The electrons start pairing only when all the degenerate orbitals of a subshell are singly occupied with parrallel s ...
... for n, l and ml provided their spins are opposite (ms is different). An orbital can have at the most two electrons if they have opporite spins. Hund’s Rule of maximum Multiplicity : ‘‘The electrons start pairing only when all the degenerate orbitals of a subshell are singly occupied with parrallel s ...
Chemical Bonding
... These formations are made of crystals of calcium carbonate, CaCO3(s), also known as limestone. Calcium carbonate, as its name and formula suggest, is a compound made up of three different elements. In addition to its crystalline structure, calcium carbonate has high melting and boiling points and di ...
... These formations are made of crystals of calcium carbonate, CaCO3(s), also known as limestone. Calcium carbonate, as its name and formula suggest, is a compound made up of three different elements. In addition to its crystalline structure, calcium carbonate has high melting and boiling points and di ...
Bent's rule

Bent's rule describes and explains the relationship between the isovalent hybridization of central atoms in molecules and the electronegativities of substituents. The rule was stated by Henry Bent as follows: ""Atomic s character concentrates in orbitals directed toward electropositive substituents"".The chemical structure of a molecule is intimately related to its properties and reactivity. Valence bond theory proposes that molecular structures are due to covalent bonds between the atoms and that each bond consists of two overlapping and typically hybridised atomic orbitals. Traditionally, p-block elements in molecules are assumed to hybridise strictly as spn, where n is either 1, 2, or 3. In addition, the hybrid orbitals are all assumed to be equivalent (i.e. the n+1 spn orbitals have the same p character). Results from this approach are usually good, but they can be improved upon by allowing hybridised orbitals with noninteger and unequal p character. Bent's rule provides a qualitative estimate as to how these hybridised orbitals should be constructed. Bent's rule is that in a molecule, a central atom bonded to multiple groups will hybridise so that orbitals with more s character are directed towards electropositive groups, while orbitals with more p character will be directed towards groups that are more electronegative. By removing the assumption that all hybrid orbitals are equivalent spn orbitals, better predictions and explanations of properties such as molecular geometry and bond strength can be obtained.Bent's rule can be generalized to d-block elements as well. The hybridisation of a metal center is arranged so that orbitals with more s character are directed towards ligands that form bonds with more covalent character. Equivalently, orbitals with more d character are directed towards groups that form bonds of greater ionic character.