Fundamental Equilibrium Concepts
... The numeric value of Qc for a given reaction varies; it depends on the concentrations of products and reactants present at the time when Qc is determined. When pure reactants are mixed, Qc is initially zero because there are no products present at that point. As the reaction proceeds, the value of Q ...
... The numeric value of Qc for a given reaction varies; it depends on the concentrations of products and reactants present at the time when Qc is determined. When pure reactants are mixed, Qc is initially zero because there are no products present at that point. As the reaction proceeds, the value of Q ...
Go FIGure
... nearly nonpolar, and the molecules are symmetrical enough to cancel much of the weak C ¬ H bond dipoles. The attraction between the polar water molecules and the nonpolar hydrocarbon molecules is not sufficiently strong to allow the formation of a solution. Nonpolar liquids tend to be insoluble in p ...
... nearly nonpolar, and the molecules are symmetrical enough to cancel much of the weak C ¬ H bond dipoles. The attraction between the polar water molecules and the nonpolar hydrocarbon molecules is not sufficiently strong to allow the formation of a solution. Nonpolar liquids tend to be insoluble in p ...
REVIEWS Environmental remediation by photocatalysis R. Vinu AND Giridhar Madras
... a major role (reaction (2)), and the contribution of reactions (8)–(14) for the overall oxidation of the substrate is negligible. Once the active species are generated, the reactants are adsorbed onto the surface of the photocatalyst (reactions (15)–(17)). This is followed by the oxidation of the re ...
... a major role (reaction (2)), and the contribution of reactions (8)–(14) for the overall oxidation of the substrate is negligible. Once the active species are generated, the reactants are adsorbed onto the surface of the photocatalyst (reactions (15)–(17)). This is followed by the oxidation of the re ...
Chapter+12
... Most often we are given a starting mass and want to find out the mass of a product we will get (called theoretical yield) or how much of another reactant we need to completely react with it (no leftover ingredients!) Now we must go from grams to moles, mole ratio, and back to grams of compound we ar ...
... Most often we are given a starting mass and want to find out the mass of a product we will get (called theoretical yield) or how much of another reactant we need to completely react with it (no leftover ingredients!) Now we must go from grams to moles, mole ratio, and back to grams of compound we ar ...
10. Solution Guide to Supplementary Exercises
... COCl2(g) Kc = 22 4HCl(g) + O2(g) Kc = 5.0 x 10–4 C 2HBr(g) H2(g) + Br2(g) Kc = 7.0 x 10–20 D N2(g) + O2(g) 2NO(g) Kc = 1.0 x 10–31 ...
... COCl2(g) Kc = 22 4HCl(g) + O2(g) Kc = 5.0 x 10–4 C 2HBr(g) H2(g) + Br2(g) Kc = 7.0 x 10–20 D N2(g) + O2(g) 2NO(g) Kc = 1.0 x 10–31 ...
CO2 Capture from Flue gas using Amino acid salt
... Ph.D degree at the Technical University of Denmark (DTU). The work was carried out at the Center for Energy Resources Engineering (CERE) in the Department of Chemical and Biochemical Engineering at DTU, under the supervision of Associate Professor Kaj Thomsen and Professor Erling H. Stenby. The Ph.D ...
... Ph.D degree at the Technical University of Denmark (DTU). The work was carried out at the Center for Energy Resources Engineering (CERE) in the Department of Chemical and Biochemical Engineering at DTU, under the supervision of Associate Professor Kaj Thomsen and Professor Erling H. Stenby. The Ph.D ...
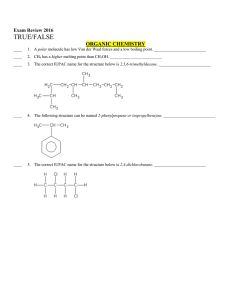
Multiple Choice Exam Review June 2016
... ____ 12. The shape of SO2 is trigonal planar. ____________________ ____ 13. The valence p orbitals in phosphorus, P, are half-filled. ____________________ ____ 14. All of the valence electrons in Fe2+ must have the same spin. _________________________ ____ 15. The shape of boron trifluoride, BF3, is ...
... ____ 12. The shape of SO2 is trigonal planar. ____________________ ____ 13. The valence p orbitals in phosphorus, P, are half-filled. ____________________ ____ 14. All of the valence electrons in Fe2+ must have the same spin. _________________________ ____ 15. The shape of boron trifluoride, BF3, is ...
free sample
... A) A weak acid solution consists of mostly nonionized acid molecules. B) The term "strong electrolyte" means that the substance is extremely reactive. C) A strong acid solution consists of only partially ionized acid molecules. D) The term "weak electrolyte" means that the substance is inert. E) A m ...
... A) A weak acid solution consists of mostly nonionized acid molecules. B) The term "strong electrolyte" means that the substance is extremely reactive. C) A strong acid solution consists of only partially ionized acid molecules. D) The term "weak electrolyte" means that the substance is inert. E) A m ...
- Chemistry
... The standard molar enthalpy of formation of liquid methanol, CH3OH(l), is the standard enthalpy change of the following reaction: ...
... The standard molar enthalpy of formation of liquid methanol, CH3OH(l), is the standard enthalpy change of the following reaction: ...
Removal of hydrogen fluoride from gas streams
... Figure 2.1: Interaction between CaF2 and HF ......................................... 32 Figure 2.2:Thermogravimetric curve for CaF2 (BDH) after exposure to 10% HF/N2 at room temperature............................................ 43 Figure 2.3:Thermogravimetric curve for CaF2 (Saarchem) after expos ...
... Figure 2.1: Interaction between CaF2 and HF ......................................... 32 Figure 2.2:Thermogravimetric curve for CaF2 (BDH) after exposure to 10% HF/N2 at room temperature............................................ 43 Figure 2.3:Thermogravimetric curve for CaF2 (Saarchem) after expos ...
Workshop materials for Class XII
... If you have any difficulty with this write down a list of your personal charecteristics. Some of these will hopefully be strengths. ...
... If you have any difficulty with this write down a list of your personal charecteristics. Some of these will hopefully be strengths. ...
Teacher Edition Calculations
... Ionic compounds do not exist as molecules. Ionic substances exist as 3 -D crystalline structures not as molecules, which are small discrete units held together by covalent bonds between nonmetal atoms. The term formula weight, being the sum o f the atomic weights of the atoms represented in the simp ...
... Ionic compounds do not exist as molecules. Ionic substances exist as 3 -D crystalline structures not as molecules, which are small discrete units held together by covalent bonds between nonmetal atoms. The term formula weight, being the sum o f the atomic weights of the atoms represented in the simp ...
synthesis and properties of v3+ analogues of jarosite-group
... all three mechanisms play a role in maintaining the charge balance in the precipitates of the V3+ analogues of jarosite-type compounds. In Table 3, we present the compositional data for the V3+ analogues of ammoniojarosite, dorallcharite and hydronium jarosite. The V3+ analogue of dorallcharite was ...
... all three mechanisms play a role in maintaining the charge balance in the precipitates of the V3+ analogues of jarosite-type compounds. In Table 3, we present the compositional data for the V3+ analogues of ammoniojarosite, dorallcharite and hydronium jarosite. The V3+ analogue of dorallcharite was ...
1 Ag PO 7.5 10 1.79 10 418.57 mol x gL x M g
... charged species but the assumption does not work well when working with charges greater than +1 or −1. Ion paring becomes a significant factor when ions charges are large and when concentrations are high. ...
... charged species but the assumption does not work well when working with charges greater than +1 or −1. Ion paring becomes a significant factor when ions charges are large and when concentrations are high. ...
Synthetic Organic Chemistry - Name
... The halogen metal exchange is useful for converting aryl and alkenyl halides to the corresponding lithium compounds by the use of of butyl lithium.Driving force of this reaction is the greater stability of sp2 carbanion than the sp3 carbanion. ...
... The halogen metal exchange is useful for converting aryl and alkenyl halides to the corresponding lithium compounds by the use of of butyl lithium.Driving force of this reaction is the greater stability of sp2 carbanion than the sp3 carbanion. ...
From Kinetics to Equilibrium
... • There are spaces between the particles of matter. The speed and spacing determine the physical state of matter. (See Figure 12.3.) • Adding heat increases the speed of the moving particles, thus increasing their kinetic energy, as well as the space they occupy. The kinetic molecular theory (KMT) e ...
... • There are spaces between the particles of matter. The speed and spacing determine the physical state of matter. (See Figure 12.3.) • Adding heat increases the speed of the moving particles, thus increasing their kinetic energy, as well as the space they occupy. The kinetic molecular theory (KMT) e ...
Chapter 4 - Chemistry
... (c) is a weak electrolyte. A small amount of the compound dissociates into ions in water. When NaCl dissolves in water it dissociates into Na and Cl ions. When the ions are hydrated, the water molecules will be oriented so that the negative end of the water dipole interacts with the positive sodiu ...
... (c) is a weak electrolyte. A small amount of the compound dissociates into ions in water. When NaCl dissolves in water it dissociates into Na and Cl ions. When the ions are hydrated, the water molecules will be oriented so that the negative end of the water dipole interacts with the positive sodiu ...
COMPETITION PTOBLEMS 1
... This publication contains the competition problems from the first twenty International Chemistry Olympiads (ICHO) organized in the years 1968 – 1988. It has been published by the ICHO International Information Centre in Bratislava (Slovakia) on the occasion of the 40th anniversary of this internatio ...
... This publication contains the competition problems from the first twenty International Chemistry Olympiads (ICHO) organized in the years 1968 – 1988. It has been published by the ICHO International Information Centre in Bratislava (Slovakia) on the occasion of the 40th anniversary of this internatio ...