On Exotic Orders in Stongly Correlated Systems
... many skills beyond those necessary to complete the task at hand. Always ready with unforeseen questions, he has repeatedly challenged me to understand things more deeply, and to recognize honestly the boundaries of my own understanding. But above all, he has been a constant reminder of what drew me ...
... many skills beyond those necessary to complete the task at hand. Always ready with unforeseen questions, he has repeatedly challenged me to understand things more deeply, and to recognize honestly the boundaries of my own understanding. But above all, he has been a constant reminder of what drew me ...
Theoretical study of solitonic excitations in Bose
... (Summary of the Ph.D. thesis) On the basis of quantum mechanics and statistical physics it is verifiable theoretically that a macroscopical part of system of bosonic particles fills the same quantum state at low temperature. This effect was predicted in 1924, but the experimental observation has bee ...
... (Summary of the Ph.D. thesis) On the basis of quantum mechanics and statistical physics it is verifiable theoretically that a macroscopical part of system of bosonic particles fills the same quantum state at low temperature. This effect was predicted in 1924, but the experimental observation has bee ...
20.2 Oxidation Numbers
... roads in the winter, why is it important to wash the salt off your car? Many parts of your car’s body are made of steel, an iron alloy. The salt can cause the metal to corrode, or rust, faster than it would otherwise because when mixed with water, the salt creates a conductive solution that allows e ...
... roads in the winter, why is it important to wash the salt off your car? Many parts of your car’s body are made of steel, an iron alloy. The salt can cause the metal to corrode, or rust, faster than it would otherwise because when mixed with water, the salt creates a conductive solution that allows e ...
Review Packet Answers - Bremerton School District
... (A complete explanation based on Le Chatelier's principle is also acceptable.) c) two points The mass of NH4HS increases. A decrease in volume causes the pressure of each gas to increase. To maintain the value of the pressure equilibrium constant, Kp, the pressure of each of the gases must decrease. ...
... (A complete explanation based on Le Chatelier's principle is also acceptable.) c) two points The mass of NH4HS increases. A decrease in volume causes the pressure of each gas to increase. To maintain the value of the pressure equilibrium constant, Kp, the pressure of each of the gases must decrease. ...
Figure 2 - Universität Düsseldorf
... in magnetic confinement nuclear fusion devices. However, through reactions with the hydrogen based fusion plasma, carbon forms amorphous hydrogenated carbon co-deposits (a-C:H) in the vacuum vessels. If tritium is used to fuel the reactor, this co-deposition can quickly lead to an inacceptable high ...
... in magnetic confinement nuclear fusion devices. However, through reactions with the hydrogen based fusion plasma, carbon forms amorphous hydrogenated carbon co-deposits (a-C:H) in the vacuum vessels. If tritium is used to fuel the reactor, this co-deposition can quickly lead to an inacceptable high ...
Technical description of GLYDE-II 1 Supplemental Information
... archetypes to specify the parts of larger entities. For example, a free_atom (“C”, “H”, “N”, “O”, etc.) can be referenced (used as an archetype) to specify a bound_atom within a molecule, such as a monosaccharide. This monosaccharide, in turn, can be referenced (used as an archetype) to specify a gl ...
... archetypes to specify the parts of larger entities. For example, a free_atom (“C”, “H”, “N”, “O”, etc.) can be referenced (used as an archetype) to specify a bound_atom within a molecule, such as a monosaccharide. This monosaccharide, in turn, can be referenced (used as an archetype) to specify a gl ...
Sample pages 2 PDF
... In other words, particle size distribution narrows, independently on the initial distribution while all particles grow and no additional nucleation appears. This selfregulating mechanism of size distribution is often called “focusing effect” [29]. On the whole, particle growth is controlled by compe ...
... In other words, particle size distribution narrows, independently on the initial distribution while all particles grow and no additional nucleation appears. This selfregulating mechanism of size distribution is often called “focusing effect” [29]. On the whole, particle growth is controlled by compe ...
Exact Results for Thermodynamics of the Hydrogen Plasma
... into hydrogen atoms is exactly taken into account. Such low-density expansions have been more recently completed up to order ρ 5/2 [4–7]. Those results have been checked afterwards in Ref. [37], and their high-temperature form in the one-component case does coincide with that derived in Ref. [20]. I ...
... into hydrogen atoms is exactly taken into account. Such low-density expansions have been more recently completed up to order ρ 5/2 [4–7]. Those results have been checked afterwards in Ref. [37], and their high-temperature form in the one-component case does coincide with that derived in Ref. [20]. I ...
Quantitative Comparison of the Hydrogen Bond
... FIGURE 1: Hydrogen bond topologies as derived by detected h3JNC′ H-bond correlations for ubiquitin in the native state (A) and in the A-state (B). Top: two-dimensional maps representing the detected h3JNC′ scalar correlations (filled diamonds). Secondary structure elements are indicated as filled ar ...
... FIGURE 1: Hydrogen bond topologies as derived by detected h3JNC′ H-bond correlations for ubiquitin in the native state (A) and in the A-state (B). Top: two-dimensional maps representing the detected h3JNC′ scalar correlations (filled diamonds). Secondary structure elements are indicated as filled ar ...
Ph. D. Thesis
... Systems with competing orders are of great interest in condensed matter physics. When two phases have comparable energies, novel interplay effects such can be induced by tuning an appropriate parameter. In this thesis, we study two problems of competing orders - (i) ultracold atom gases with competi ...
... Systems with competing orders are of great interest in condensed matter physics. When two phases have comparable energies, novel interplay effects such can be induced by tuning an appropriate parameter. In this thesis, we study two problems of competing orders - (i) ultracold atom gases with competi ...
CHAPTER 3 STOICHIOMETRY
... Solution: Let's first calculate the number of N atoms in 1.68 10 g of urea. First, we must convert grams of urea to number of molecules of urea. This calculation is similar to Problem 3.26. The molecular formula of urea shows there are two N atoms in one urea molecule, which will allow us to conve ...
... Solution: Let's first calculate the number of N atoms in 1.68 10 g of urea. First, we must convert grams of urea to number of molecules of urea. This calculation is similar to Problem 3.26. The molecular formula of urea shows there are two N atoms in one urea molecule, which will allow us to conve ...
Syllabus Cambridge International A & AS Level Chemistry Syllabus code 9701
... Candidates can take the course either as an AS Level, A Level or staged assessment to A Level. Practice of experimental skills Candidates should be directed towards the practice of experimental skills throughout the whole period of their course of study. Candidates’ experimental skills will be teste ...
... Candidates can take the course either as an AS Level, A Level or staged assessment to A Level. Practice of experimental skills Candidates should be directed towards the practice of experimental skills throughout the whole period of their course of study. Candidates’ experimental skills will be teste ...
ANNEX (Manuscrits posteriors a la Comissió de Doctorat de Juliol del...
... Dimethyl substitution of [1]- at the 8, 8’ positions was achieved by B-C cross coupling reaction employing a modified Kumada reaction (Scheme 2). Addition of 5 equivalents of methylmagnesium bromide to a cooled (0°C) solution of Cs[2] in THF, followed by a catalytic amount of [PdCl2(PPh3)2] and CuI ...
... Dimethyl substitution of [1]- at the 8, 8’ positions was achieved by B-C cross coupling reaction employing a modified Kumada reaction (Scheme 2). Addition of 5 equivalents of methylmagnesium bromide to a cooled (0°C) solution of Cs[2] in THF, followed by a catalytic amount of [PdCl2(PPh3)2] and CuI ...
Chemistry 134 Problem Set Introduction
... 14.74 (a) List each of the elements in the nitrogen family and classify it as a metal, metalloid, or nonmetal. (b) Which member of the nitrogen family is the most abundant element in the Earth's atmosphere? (c) List the highest and lowest oxidation state for each member of the nitrogen family. 14.75 ...
... 14.74 (a) List each of the elements in the nitrogen family and classify it as a metal, metalloid, or nonmetal. (b) Which member of the nitrogen family is the most abundant element in the Earth's atmosphere? (c) List the highest and lowest oxidation state for each member of the nitrogen family. 14.75 ...
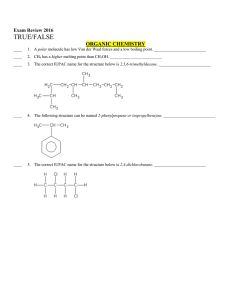
document
... 0.0119 mole NaHCO3 reacts with 0.0119 mole HCl 0.0171 mole Mg(OH)2 reacts with 0.0342 mole HCl ...
... 0.0119 mole NaHCO3 reacts with 0.0119 mole HCl 0.0171 mole Mg(OH)2 reacts with 0.0342 mole HCl ...
OCR AS Level Chemistry B (Salters) H033
... AS Level Chemistry B (Salters) (from September 2015) Chemistry B (Salters) was first examined in 1992 as a new concept project examination. In contrast to the traditional ‘topic-based’ approach, Chemistry B (Salters) is ‘context-led’. Chemical concepts are introduced within a relevant context, the c ...
... AS Level Chemistry B (Salters) (from September 2015) Chemistry B (Salters) was first examined in 1992 as a new concept project examination. In contrast to the traditional ‘topic-based’ approach, Chemistry B (Salters) is ‘context-led’. Chemical concepts are introduced within a relevant context, the c ...
L-edge X-ray absorption study of mononuclear vanadium complexes
... transition metal L-edge spectra are typically richly structured. They are, however, also difficult to interpret since in addition to ligand-field and covalency effects, one must consider the spin– orbit coupling (SOC) interaction between the potentially many final state multiplets. The SOC interaction ...
... transition metal L-edge spectra are typically richly structured. They are, however, also difficult to interpret since in addition to ligand-field and covalency effects, one must consider the spin– orbit coupling (SOC) interaction between the potentially many final state multiplets. The SOC interaction ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.