* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Ch. 1-- Matter and Change
Water pollution wikipedia , lookup
Abundance of the chemical elements wikipedia , lookup
Click chemistry wikipedia , lookup
Isotopic labeling wikipedia , lookup
Transition state theory wikipedia , lookup
Condensed matter physics wikipedia , lookup
Chemical element wikipedia , lookup
American Chemical Society wikipedia , lookup
Computational chemistry wikipedia , lookup
Al-Shifa pharmaceutical factory wikipedia , lookup
Process chemistry wikipedia , lookup
Institute of Chemistry Ceylon wikipedia , lookup
Organic chemistry wikipedia , lookup
Nuclear chemistry wikipedia , lookup
Chemical weapon proliferation wikipedia , lookup
Chemical potential wikipedia , lookup
Molecular dynamics wikipedia , lookup
Chemical industry wikipedia , lookup
Freshwater environmental quality parameters wikipedia , lookup
Chemical weapon wikipedia , lookup
Chemical reaction wikipedia , lookup
Chemical plant wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Chemical Corps wikipedia , lookup
Green chemistry wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
History of molecular theory wikipedia , lookup
Drug discovery wikipedia , lookup
Analytical chemistry wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
California Green Chemistry Initiative wikipedia , lookup
Safety data sheet wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Stoichiometry wikipedia , lookup
Registration, Evaluation, Authorisation and Restriction of Chemicals wikipedia , lookup
Physical organic chemistry wikipedia , lookup
VX (nerve agent) wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
History of chemistry wikipedia , lookup
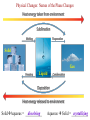
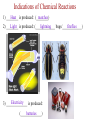
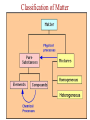
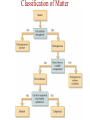
Ch. 1—Chemistry: An Introduction What is chemistry? • Chemistry is the study of the ___________________ composition of substances and the changes they undergo. alchemy • It began from “_______________”... the attempts of alchemists to change common metals into _________ through trial and error. gold Divisions of Chemistry There are several divisions or branches of chemistry: 1) _________ Organic chemistry: the study of substances that contain ________ carbon Example: how gasoline is produced from oil Inorganic without 2) _______________ chemistry: the study of substances __________ ___________ carbon Example: how table salt reacts with different acids Click below for the link to the lecture • http://danreid.wikispaces.com/file/view/Ch. %201%20Notes%20%28teacher%29v2.s wf Divisions of Chemistry (continued) 3) _______________ Analytical chemistry: the study of the _______________ quantitative composition of substances Example: how much chlorine is in a sample of tap water Biochemistry living 4) ____________________: the study of the chemistry of _________ __________________ organisms Example: how sugar in the blood stream of cats affect insulin production The Scientific Method • The scientific method is way to solve a scientific problem. It is an approach to a solution (using mostly common sense.) Example: Your flashlight doesn’t work. (Not necessarily a “scientific problem.”) Steps to the Scientific Method observations (1) Make _________________-Use your 5 senses to gather information. hypothesis (2) Propose a ______________-Make an “educated guess” for what is happening. experiments (3) Perform _______________-This tests your hypothesis. Many experiments are sometimes needed to test a hypothesis. The same experiment must give similar results if the experiment is to be reliable. Steps to the Scientific Method (continued) (4) Make a _____________-theory This should explain the results of your experiments. Theories may ___________ or be ___________ change rejected over time because of results from new experiments. Scientific Laws • • • Laws describe ____________ natural ___________________. phenomena why the A law tells what happens. It does not attempt to explain _____ phenomena occurs. (That is the job of a theory.) math equation Laws can often be summarized by a _____________ __________. Ideal Gas ______”) Law Example: PV = nRT (The “_________ ______ This equation is used in Ch. 12. The law shows the relationship between the pressure, volume and temperature of a given quantity of gas in a container. Chapter 2--Matter & Change Matter vs. Mass • mass and takes up ___________. space Matter is anything that has _________ solids __________, liquids and ________... gases Examples: ________, (the three phases of matter) • • • light Things that are NOT matter: __________, heat, emotions ________, sound... Mass is the amount of ___________ in an object. matter The standard metric unit for mass is the ______________. kilogram Plasma • • Plasma is a high energy electrically charged mixture of ions and electrons. __________ are made of plasma. Stars While plasma is the most abundant phase of matter in the universe, on earth it only occurs in a few limited places: – Lightning bolts – Flames – Fluorescent lights – Aurora Borealis (Northern Lights) • • • Bose-Einstein Condensate Predicted in 1924 and created in 1995, the BEC is a small group of atoms clumped together when taken down to a few billionths of a degree above absolute zero. This group of atoms takes up the same place, creating a "super atom." There are no longer thousands of separate atoms. They all take on the same qualities and for our purposes become one blob. Einstein Bose http://www.colorado.edu/physics/2000/bec/ 400 nanoKelvins 200 nK 50 nK States of Matter phase fixed shape fixed volume compressible solid YES YES NO liquid NO NO YES NO NO YES gas Particle Motion • • • • vibrate Solids-- particles ____________ back and forth in fixed positions slide past each other in the container Liquids-- clumps of particles ______ fly all over the place and collide Gases-- individual particles _____ _________ Vapor is a term used for a gaseous substance that is normally a ________ solid or _______ liquid at room temperature.) Example: _______ water vapor States of Matter Physical Properties and Physical Changes • Physical properties can be determined/measured without changing the substance’s composition. Examples: _______, color odor, __________, taste mass, ________, density _________ point, hardness, boiling point, ____________ melting solubility, etc. • Physical Changes alter a substance without changing its composition. Examples: crushing, ripping, breaking, and any _________ phase changes…(boiling, freezing, melting, etc.) • Most physical changes just alter the size of the particles and are usually reversible. Physical Properties Physical Changes: Names of the Phase Changes Solid Gas Liquid SolidAqueous = ___________ dissolving Aqueous Solid = ___________ crystallizing • Mixtures Mixtures are a physical blend of two or more substances mixed together.” The parts can be separated by _____________ means or physical ____________ changes. physical There are 2 types of mixtures: (1) _________________ Heterogeneous Mixtures: the parts mixed together can still be distinguished from one another...NOT uniform in composition. Examples: chicken soup, fruit salad, _____, dirt sand in water Homogeneous (2) _________________ Mixtures: the parts mixed together cannot be distinguished from one another...completely uniform in composition. Examples: ______, Air Kool-aid, ________, Brass salt water, milk • solution Another term for a homogeneous mixture is a “______________.” Heterogeneous Mixtures Homogeneous Mixtures • Distillation One way to separate a _________ solid from a ___________ liquid in a solution is by distillation. There are 2 steps to the process: Step 1: _________ Boil the solution. Condense Step 2: ________________ the vapor as is escapes and collect it. Distillation Animation • Chemical Properties and Chemical Changes Chemical properties cannot be determined/measured without changing the substance’s composition Examples: ____________, burning whether or not it reacts with an acid or a base. Chemical Changes • Chemical changes will alter a substance and change its composition. rusting Examples: burning, ___________, rotting or decomposing, fermenting __________________, and other chemical reactions. • Most, but not all, chemical changes are irreversible. – You can’t “reverse” the burning of paper. – _____________ use a reversible chemical reaction Rechargeable ____________ batteries Indications of Chemical Reactions 1) ________ Heat is produced: (________) matches Light is produced: (_____________ lightning fireflies 2) ________ bugs/_____________) Electricity 3) ______________ is produced: (______________) batteries Indications of Chemical Reactions 4) ___________________ Precipitate forms: (_______ soap ________) scum Two liquids chemically react to form a solid. 5) gas/smoke/odor/bubbles produced: soda fizz (________ ______) • • Elements vs. Compounds An element cannot be ___________ broken down or _____________ changed into simpler substances by chemical means. Elements are the _________ simplest forms of matter that can exists in normal laboratory conditions. Examples: _______, Gold Helium, __________ Mercury • • 2 or ________ more different elements A compound is made up of ____ ______________ chemically bonded together. Compounds can only be broken down into simpler substances by ____________ chemical ____________. reactions Water Sand, _______________ NaCl (table salt) Examples: _______, Classification of Matter Classification of Matter Classification of Matter • • • Chemical Symbols Chemists use chemical symbols for the elements involved in a chemical reaction. The symbols are a shorthand way of representing the ______________. (See the Periodic Table for a list of all the elements symbols.) The first letter of the chemical symbol for an element is always capitalized _________________. The next letter, if needed, is _______________. Each capital letter lowercase in a formula, therefore, represents another element. H ____, Ne Hg, ___, S NaBr, ________, H2O LiC2H3O2 Examples: ____, • Some symbols come from _______ Latin names: Au=Aurum (Gold) • • Chemical Reactions When writing chemical reactions, the substances that ___________ react with each other are written on the _______ left and are called “reactants”. The substances that are ____________ produced are written on the _______ right and are called the “products.” Reactants Products • yields or “reacts to The “ ” symbol can be read as “_______” produce.” Example: two 2Hhydrogen 2H2O 2 + O2 molecules plus one oxygen which means “____________________________________ molecule yields two water molecules ________________________________________________.” • • • • Conservation of Mass During chemical (or physical) reactions, mass (or matter) is neither _____________ nor _________________. created destroyed The mass of all the reactants _________ equals the mass of all the products. number of each kind of atom is the same. The ___________ Sometimes it appears that the reactant and product masses are not gas was probably a reactant or product in the equal, but a _______ reaction, and that is making the difference! Example: • • 2H2 + O2 2H2O If 4 grams of hydrogen reacted with oxygen to produce 36 grams of 32 water, how many grams of oxygen were used? _______ # of H’s and O’s on each side is __________! constant Notice that the ____ Conservation of Mass CaCl2 + Na2SO4 CaSO4 + 2NaCl mass before = mass after # atoms before = # atoms after