* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Smooth ER - Home - KSU Faculty Member websites
Survey
Document related concepts
Nucleic acid double helix wikipedia , lookup
Epigenetics in stem-cell differentiation wikipedia , lookup
Therapeutic gene modulation wikipedia , lookup
DNA vaccination wikipedia , lookup
Nicotinic acid adenine dinucleotide phosphate wikipedia , lookup
Epigenomics wikipedia , lookup
History of genetic engineering wikipedia , lookup
Cre-Lox recombination wikipedia , lookup
Point mutation wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Extrachromosomal DNA wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Polycomb Group Proteins and Cancer wikipedia , lookup
Primary transcript wikipedia , lookup
Transcript
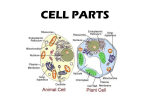
Cell Structure & Function There are two types of cells: 1. Prokaryotic- cells that DO NOT have a membrane around their heredity material 2. Eukaryotic- cells with a NUCLEUS , which is heredity material surrounded by a membrane • Which is more complicated? • EUKARYOTIC! • Examples of Eukaryotes: • plants, animals, fungi. • Examples of Prokaryotes: • Bacteria Organelles • There are many small structures located inside the cell. • These structures are called organelles (little organs). • These organelles perform functions that keep the cell alive. • Some organelles are found only in plant cells, and some only in animal cells. Name some organelles inside an animal cell • • • • • • • • Nucleus Lysosome Golgi bodies Mitochondrion Ribosome Vacuole Cytoplasm Endoplasmic reticulum (ER) Types of Cells Cell Types Eukaryotic Cells Plant Cells Prokaryotic Cells Animal Cells Animal Cells Cellular Anatomy Biochemistry and Evolution • Prokaryotes - do not have a membranebounded nucleus • Eukaryotes - possess nucleus and other complex internal structures • Prokaryotes and eukaryotes appear to have evolved from a common ancestor over three billion years ago The Cell is the Basic Unit of Life • Plasma membrane - surrounds aqueous environment of the cell • Cytoplasm - all materials enclosed by the plasma membrane (except the nucleus) • Cytosol - aqueous portion of the cytoplasm minus subcellular structures • Bacteriophage or phage - viruses that infect prokaryotic cells Prokaryotic Cells: Structural Features • Prokaryotes, or bacteria are usually singlecelled organisms • Prokaryotes lack a nucleus (their DNA is packed in a nucleoid region of the cytoplasm) • Escherichia coli (E. coli) - one of the best studied of all living organisms • E. coli cells are ~0.5mm diameter, 1.5mm long Eukaryotic Cells: Structural Features • Eukaryotes: plants, animals, fungi, protists • Have a membrane-enclosed nucleus containing the chromosomes • Are commonly 1000-fold greater in volume than prokaryotic cells • Have an intracellular membrane network that subdivides the interior of the cell Eukaryotic cell (animal) Eukaryotic cell (plant) NUCLEUS: • Most functions of the cell are controlled by the nucleus. • Functions: “Brain” of the cell. • It houses and protects the cell’s genetic information. The Cell Nucleus • Why have nuclear pores at all? • What materials can pass through the nuclear envelope? What materials are retained? • What is in the nucleolus? • What molecules are in chromatin? Nucleus • Large round structure located inside the cytoplasm. • Contains genetic material (DNA). • Has a nuclear membrane (semipermeable). • Contains a Nucleolus (makes Ribosomes). • Controls the activity of the cell. Name 3 parts of the nucleus • Nuclear membrane • Chromatin • nucleolus nucleolus Nuclear membrane chromatin Chromatin questions • • • • • • Where is chromatin located? In the nucleus. Where is DNA located? In the chromatin. What does DNA do? In controls the activities in the cell. Note! • The shape of chromatin changes when a cell begins to divide. • When a cell begins to divide the chromatin coils and takes the form of chromosomes. Chromosomes • Long thread-like structures found in the nucleus of the cell. • Contain hereditary information. • Genes are hereditary units made up of DNA. Chromosomes • Nucleolus: dense part of the nucleus. • Site where DNA is concentrated. Nuclear Envelope: double layer that covers the nucleus. Also made of 2 phospholipid bilayers. • Nuclear Pores: holes in the nuclear envelope that allow passageways for RNA and other things entering and leaving the nucleus. DNA structure and replication Purine (1) is a heterocyclic aromatic organic compound, consisting of a pyrimidine ring fused to an imidazole ring (pyrimidine + imidazole) + A G Aside from DNA and RNA, purines are biochemically significant components in a number of other important biomolecules, such as ATP, GTP, cyclic AMP, NADH, and coenzyme A. They may also function directly as neurotransmitters, acting upon purinergic receptors. Adenosine, activates adenosine receptors. Purines are found in high concentration in meat and meat products, especially internal organs such as liver and kidney. Plant based diet is generally low in purines Examples of high purine sources include: sweetbreads, anchovies, liver, beef kidneys, scallops A moderate amount of purine is also contained in beef, pork, fish and seafood, asparagus, spinach, mushrooms, green peas, beans, oatmeal, wheat bran and wheat germ.[4] Pyrimidine is a heterocyclic aromatic organic compound similar to benzene containing two nitrogen atoms at positions 1 and 3 of the six-member ring benzene Pyrimidine cytosine thymine uracil C T U Chromosome structure Chromosome Oragnisation What chromosome is made of? - chromosome is made of chromatin (nucleic acid and protein( histone). - when in mitotic and meiotic stages of cell cycle, chromatin (euchromatin & heterochromatin) stains readily. Euchromatin is a lightly packed form of chromatin that is rich in gene concentration Heterochromatin is a tightly packed form of DNA. Its major characteristic is that transcription is limited. As such, it is a means to control gene expression, through regulation of the transcription initiation. Chromosome Packaging Chromatin is organised on three basic levels: - primary (nucloesome) - secondary (solenoid) - tertiary/quaternary (final folding into chromosome shape) A typical eukaryotic chromosome contians 1 to 20 cm of DNA. During metaphase of meiosis/mitosis, this DNA is package into a chromosome with a length of only 1 to 10 mm (a condensation of almost 104-fold in length from the naked DNA molecule). Chromosome Packaging beaded string – nucleosome structure of chromatin Chromosome Packaging - nucleosome contains histones (2 of each H2A, H2B, H3 and H4, and one H1) - the diameter of nucleosome (bead) is 11nm. - 200 bp of DNA associated with one bead, 23 bp protected by H1 and 8 to 114bp (depends on species and type of cells) form a linker between beads. Chromosome Packaging Second level of chromatin organization: solenoid - the nucleosome is supercoiled and organised into a solenoid structure, with 67 nucleosomes per turn. - H1 stabilize the structure of solenoid. - the supercoiling produces a fibre of approximately 30nm in diameter. Chromosome Packaging Higher order folding of chromatin into chromosome Transcription and Translation Nucleic Acids • Polynucleotides - nucleic acid biopolymers are composed of nucleotide monomers • Nucleotide monomers are composed of: (1) A five-carbon sugar (2) A heterocyclic nitrogenous base (3) Phosphate group(s) Deoxyribose • Deoxyribose lacks a hydroxyl group at C-2. It is the sugar found in DNA. Nitrogenous bases • Major Purines: Adenine (A) Guanine (G) • Major Pyrimidines Cytosine (C) Thymine (T) Uracil (U) Adenosine Triphosphate (ATP) • Nitrogenous base (adenine), sugar (ribose) Structure of a dinucleotide • Residues are joined by a phosphodiester linkage Short segment of a DNA molecule • Two polynucleotides associate to form a double helix • Genetic information is carried by the sequence of base pairs Deoxyribonucleic Acid (DNA) • The ‘blueprint of life” • Unique and unrepeatable • Located in genes Genes make up chromosomes Chromosomes in the nucleus of the cell • DNA composed of units called nucleotides Each nucleotide is made up of a sugar, phosphate, and hydrogen base. • Double Helix model of Watson and Crick • X ray photos by Rosalind Franklin • Sides of ladder (S-P) with covalent bonds • Sugar: deoxyribose • Steps of ladder (bases) • Complementary Base Pairs Purines (Adenine/Guanine) Pyrimidines (Cytosine/Thymine) A-T (2 hydrogen bonds) C-G (3 hydrogen bonds) The Race for the DNA Model King’s College London Cavendish Lab London Rosalind Franklin James Watson Francis Crick (X ray Diffraction Studies) Photo #51 Maurice Wilkins Nobel Prize 1962 (Double Helix Model) • Mutation: a mistake in nitrogen base pairs • Pattern of bases codes for a specific amino acid • Codon: 3 letter (bases) code for 1 amino acid • Replication of DNA (Making an identical copy) DNA unzips and hydrogen bases separate (action of DNA polymerase) Free-floating nucleotides match up on each side Hydrogen bonds reform Result: 2 identical DNA molecules (Each contain 1 of the original DNA strands) Semi-Conservative Model of Replication • Each DNA molecule with a new strand and an old strand • DNA never leaves the nucleus Ribonucleic Acid (RNA) • • • • • • Made up of nucleotides Sugar called ribose Nitrogen Bases: A, C,G,U Single-stranded Located in the nucleus and in the ribosomes 3 Types: Messenger RNA (mRNA); carries message of DNA to ribosomes (3 letter code – codon); single strand of letters Transfer RNA (tRNA): reads the mRNA code at the ribosomes (3 letter code – anticodon); carries an amino acid; shamrock shape Ribosomal RNA (rRNA): joins mRNA to tRNA Transcription • Making of mRNA from one side of DNA in nucleus DNA polymerase unzips DNA Hydrogen bonds break and energy is released One side of DNA acts as a “template” (model) for making mRNA Free-floating nucleotides line up and form mRNA DNA reforms mRNA carries the message of DNA to ribosomes Translation • Occurs at the ribosomes • Anticodon of tRNA joins with codon of mRNA (rRNA helps) • Amino acid pops off end of tRNA • Amino acids string together to form a protein (Protein Synthesis) Endoplasmic reticulum (ER) • cytoplasmic channels from the cell membrane to the nuclear membrane • associated with the storage, synthesis, and transport of materials within the cell • “HIGHWAY” for cell transport Endoplasmic Reticulum • What are the two types of ER? • How does the role of each type differ? • What kind of cells would have a lot of rough ER? Smooth ER? Endoplasmic Reticulum • A system of channels within the cytoplasm, that transports materials. • Smooth E.R. has no ribosomes on it. • Rough E.R. has ribosomes. – 2 Types: 1.Rough ER: – Rough appearance because it has ribosomes – Function: helps make proteins, that’s why it has ribosomes 2.Smooth ER: – NO ribosomes – Function: makes fats or lipids The endomembrane system regulates protein traffic and performs metabolic functions in the cell Endoplasmic reticulum Smooth v. Rough Smooth ER Nuclear envelope Rough ER Smooth ER •lacks ribosomes •Involved in synthesis of lipids, metabolism of carbohydrates, and detoxification of poisons ER lumen 200 nm Rough ER Smooth ER Rough ER •Has attached ribosomes •Makes proteins and phospholipids The endomembrane system regulates protein traffic and performs metabolic functions in the cell Endoplasmic reticulum Smooth v. Rough Smooth ER Nuclear envelope Rough ER Smooth ER •lacks ribosomes Transport vesicles-transport materials from the ER to the Golgi •Involved in synthesis of lipids, ER lumen metabolism of carbohydrates, and detoxification of poisons 200 nm Rough ER •Has attached ribosomes •Makes proteins and phospholipids Rough ER Smooth ER Endoplasmic Reticulum (ER) • A system of membranes that is found in a cell’s cytoplasm • Assists in the production, processing, and transport of proteins • Assists in the production of lipids The ER • Rough ER • Part of the ER with ribosomes attached • Where proteins are made and released by a vesicle The ER • Smooth ER • Part of the ER that lacks ribosomes Endoplasmic Reticulum (ER) • • • • • Network of fluid filled tubules (cisternae) Roughly ½ of eukaryotic membrane tissue Continuous with the nuclear envelope Smooth ER = lacks ribsosomes Rough ER = ribosomes bound to cytoplasmic side of ER membrane Structure of the ER The Smooth ER • Synthesis of lipids, steroids (ie: sex hormones) • Carb metabolism (ie: liver cells hydrolysis glycogen into glucose utilizes enzymes in smooth ER) • Detoxification of drugs/poisons (ie: smooth ER enzymes make drugs more soluble add – OH). Tolerance = more Smooth ER • Involved in Ca ion movement during muscle contraction. Functions of Rough ER • Attached ribosomes = protein synthesis • Abundant in cells that make proteins • Manufactured proteins are “threaded” through pore into the cisternal space of ER. • Glycoproteins – covalently bonded to carb. • Secretory proteins are packaged into transport vescicles and sent to various locations in the cell. Rough ER • Also manufactures phospholipids from precursors in cytoplasm. • Assembles phospholipids and proteins into new membrane sections. • ER membrane can expand or transfer new membrane via vescicles to other parts of endomembrane system. MITOCHONDRION: tiny, double-membrane organelles that transfer ENERGY from organic molecules to ATP. ATP powers most of the cell’s chemical reactions. Found in large amounts in muscle cells and cells requiring ENERGY. Function: Powerhouse of the cell. Mitochondria • The “powerhouse” of the cell. • Food molecules are broken down and energy is released. • Functions in Cellular Respiration. The Citric Acid (Krebs) Cycle consists of eight steps TCA Cycle Control • Citrate Synthase (Synthetase) – Condensing Enzyme – Inhibited By: • ATP • NADH • Succinyl CoA TCA Cycle Control-Cont: • Isocitrate Dehydrogenase – Activated By: • ADP – Inhibited By: • ATP • NADH TCA Cycle Control-Cont: • α-Ketoglutarate Dehydrogenase – Inhibited by: • Succinyl CoA • NADH • ATP – Contains tightly bound Tpp, lipoamide, FAD – Similar to PDH complex • E3 subunit the same TCA Cycle Control-Cont: • Succinyl CoA Synthetase – Coupled reaction with GTP – Enzyme that catalyses coupled reaction is called Nucleotidediphosphate Kinase TCA Cycle Control-Cont: • Succinate Dehydrogenase – Has Iron-Sulfur Centers – Covalently Bound with FAD • Name 2 parts of mitochondria: • Outer membrane and folded inner membrane • What types of cells you expect to have more mitochondria? why • More active cells like muscle cells because they need more energy. Mitochondrion • How many membranes? Why? • What cells would have high numbers of mitochondria? • What do mito. have to do with cloning? • What is the current theory on mito. origin? What are mitochondria? • • • • • • An intracellular organelle. There are 100 to 1000s of mitochondria/cell. All mitochondria come from the mother. Mitochondria have their own DNA. Found in all cell types, except the RBC. Major functions of mitochondria: – Makes energy in the form of ATP. – Programmed cell death (apoptosis). Chemical Energy Cars Cells Gasoline ATP Fate of Ingested Food Intermediary Energy Metabolism Fats Proteins Glucose Glycolysis TCA Cycle ETC b-oxidation TCA Cycle ETC ATP Pool Deamination TCA Cycle ETC Why is energy so important? • Role of ATP (energy) – Mechanical Work • Muscle contraction – Chemical Work • Na+/K+ Ion Pump – Synthetic Work [Anabolism] • Macromolecules – – – – Nucleic Acids Proteins Lipids Complex carbohydrates Bioenergetics: Energy • 1 teaspoon of sugar weighs 5 gm and contains 20 calories of energy • 1 teaspoon of sugar contains 10 X 1021 molecules of sugar or sucrose – 10,000,000,000,000,000,000,000 molecules • 1 teaspoon of sugar forms about 3.6 X 1023 molecules of ATP – 360,000,000,000,000,000,000,000 molecules Bioenergetics: Energy • At rest, the average adult male will need 3.0 x 1018 molecules of ATP per second for normal organ functioning. • The body produces and makes approximately 70 Kg of ATP daily (average adult male). • The brain uses approximately 70% of all ATP produced. Bioenergetics Bioenergetics Bioenergetics: Summary • Mitochondria function is to produce ATP for energy. • The mitochondria use electrons and protons from metabolism and molecular oxygen to reduce water and generate proton-motive force to produce ATP from ADP: oxidative phosphorylation. • When this process is dysfunctional, then disease can occur. • Bottomline: mitochondrial cytopathies are diseases of energy production. Bioenergetics: Summary • What happens when an organ does not get enough ATP or energy? – Brain dysfunction: when the brain doesn’t get it’s 70% of energy required: • • • • Seizure Mental Retardation Cognitive dysfunctions Psychological dysfunctions? Lysosomes • How many membranes? • Where are lysosomes formed? • Contain what? • Describe the internal environment of a lysosome. • List three major functions. • What is the relationship between Tay Sachs disease and lysosomes? Lysosomes • Internal sacs bound by single membrane • Originate by budding from Golgi based on sorting of mannose-6-phosphate “tags” on proteins • Responsible for degrading cell components that have become obsolete for cell or organism— digestive sys of cells • Internal pH about 5 (very acidic) • Compartmentalization ESSENTIAL! Failure can lead to many known disease states that result from waste accumulation in the organelle Lysosomes • contain a mixture of some 40 types of digestive enzymes, all with optimum activity at about pH 5 • this acid pH is maintained in lysosomes, as in endosomes, by proton pumps in the membrane • membrane of the lysosome is resistant to action of its own digestive enzymes due to the extensive glycosylation of the proteins on the lumenal side of the membrane. • A lysosome is a membranous sac of hydrolytic enzymes that can digest macromolecules • Lysosomal enzymes can hydrolyze proteins, fats, polysaccharides, and nucleic acids Endocytosis/Exocytosis • Some types of cell can engulf another cell by phagocytosis; this forms a food vacuole – pinocytosis; exocytosis • A lysosome fuses with the food vacuole and digests the molecules • Lysosomes also use enzymes to recycle the cell’s own organelles and macromolecules, a process called autophagy Fig. 6-14a Nucleus 1 µm Lysosome Lysosome Digestive enzymes Plasma membrane Digestion Food vacuole (a) Phagocytosis Fig. 6-14b Vesicle containing two damaged organelles 1 µm Mitochondrion fragment Peroxisome fragment Lysosome Peroxisome Vesicle (b) Autophagy Mitochondrion Digestion Peroxisome • How do the enzymes in peroxisomes differ from the enzymes in lysosomes? • What cells have many peroxisomes? Why? • Plants have special peroxisomes called glyoxysomes. What is their function? Major Metabolic Functions of the Peroxisome in Plants 1. 2. 3. 4. 5. b-oxidation of fatty acids Glyoxylate cycle Photorespiration (Glycolate pathway) Degradation of purines Decomposition of hydrogen peroxide Two Types of Peroxisomes in Plants • Leaves – Catalyzes oxidation of side product of CO2 fixation in photorespiration • Germinating seeds – Converts fatty acid in seed lipids into sugars needed for growth in the young plant b-oxidation occurs in mitochondria and peroxisomes in mammals, but exclusively in the peroxisome in plants and yeast. Glyoxysomes and Leaf Peroxisomes are Interconverted During Development • Immunogold particles of 2 sizes bound to: – Enzymes of glyoxylate cycle – Peroxisomal enzymes • The same population of peroxisomes assumes different metabolic roles depending on developmental stage of cotelydon Greening cotelydons Photorespiration and Glycolate Oxygenase activity of rubisco Consumption of O2 Glycolate cycle Production of CO2 Involves 3 organelles (chloroplasts, peroxisomes, & mitochondria) The Glycolate Cycle Glycolic acid oxidase H2O2 production The Glycolate Cycle Purine Degradation • Nucleic acid purine moieties (adenine and guanine) are degraded to uric acid O2 xanthine H2O2 O2 H2O2 uric acid Xanthine oxidase allantoin Urate oxidase Urate Oxidase • High urate oxidase concentrations contribute to formation of crystalline inclusions • All purine degradation leads to uric acid H2O2 O purines N H N H C FAD O2 C hypoxanthine H2 O xanthine oxidase O FAD N H N N H O FADH2 C H N H2O2 O2 N H H xanthine C H2 O xanthine oxidase O H N H N C H N O FADH2 C O N N H H uric acid Oxidases • The oxidases use molecular oxygen to remove hydrogen atoms from specific organic substrates • A variety of compounds, including L-amino acids, D-amino acids, polyamines, methanol, urate, xanthine, and very-long-chain fatty acids, serve as substrates for the different oxidases Peroxide Detoxification Oxidases use O2 to oxidize organic substances and produce hydrogen peroxide (H2O2) -- e.g., H2O2 generated by glycolate oxidase reaction, b-oxidation of fatty acids Peroxisomes also contain catalase, the enzyme that degrades H2O2. Importance of H2O2 degradation • 2H2O2 catalase 2H2O + O2 • Peroxisomes contain a high concentration of catalase, a heme protein H - - O - - O - -H HO- -OH (?) • Other reactive oxygen species (ROS) are formed in peroxisomes Reactive Oxygen Species 1e- 1eO2 O-2 1eH2 O2 1eOH- + ·OH H2 O .. . .. . .• .. .. .. .. - .. : H H•: O :H : : : : : : : • :O : : : + O O O O H O O H H O . .. .. . .. ..• .. .. .. Superoxide anion (radical) Hydrogen peroxide Hydroxyl radical • Cause damage to lipids, proteins, DNA • Amount ROS is reduced by catalase, and superoxide dismutase (SOD) 2O2O2 + H2O2 Radical Chemistry Initiation: RH + O2 -->R· + ·OH Propagation: Termination: R· + O2 --> · + ROO· R· + R· --> RR ROO· + RH --> R· + ROOH R· + ROO·--> ROOR ROOH--> RO· + HO· ROO· + ROO· --> ROOR + O2 Other Peroxisomal Enzymes Conclusions • Compartmentalize! To protect the cell from these destructive byproducts, such reactions are segregated. Ribosomes • Non-membrane bound! • Composed of ______ and ________. • Sites to synthesize __________. • How are prokaryotic ribosomes different from eukaryotic ribosomes? • Antibiotics, including tetracycline and streptomycin, paralyze prokaryotic ribosomes. Why don’t these drugs harm eukaryotic ribosomes?