* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download E2 Stellar radiation and stellar types
Survey
Document related concepts
Transcript
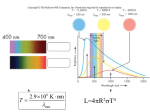
E2 Stellar radiation and stellar types Fusion Life-cycle of a star Equilibrium between radiation pressure and gravity Luminosity (symbol L) Luminosity is defined as the amount of energy radiated by the star per second (The power radiated by the star) Measured in Watts (J.s-1) Black-body radiation Black-body radiation Need to “learn” this! Black-body radiation • Black Body - any object that is a perfect emitter and a perfect absorber of radiation • object does not have to appear "black" • Stars behave approximately as black bodies Black-body radiation The amount of energy per second (power) radiated from a star (its luminosity) depends on its surface area and absolute temperature according to L = σAT4 where σ is the Stefan-Boltzmann constant (5.67 x 10-8 W.m-2.K-4) Example • The sun (radius R = 7.0 x 108 m) has a luminosity of 3.9 x 1026 W. Find its surface temperature. • From L = σAT4 and A = 4πR2 we find T = (L/σ 4πR2)¼ = 5800 K Wien’s law – Finding the temp of a star • λmaxT = constant (2.9 x 10-3 mK) Example • The sun has an approximate black-body spectrum and most of its energy is radiated at a wavelength of 5.0 x 10-7 m. Find the surface temperature of the sun. • From Wien’s law 5.0 x 10-7 x T = 2.9 x 10-3 T = 5800 K Apparent brightness (symbol b) Apparent brightness is defined as the amount of energy per second per unit area of detector b= where 2 L/4πd d is the distance from the star (in m) L is the luminosity (in W) Apparent brightness - CCD Apparent brightness is measured using a charge-coupled device (used also in digital cameras) Read the final paragraph of page 495. Apparent brightness and Luminosity Note that the apparent brightness b and luminosity L are proportional b= 2 L/4πd bαL α 4 T Spectral Class Colour Temperature/K O Blue 25 000 – 50 000 B Blue - white 12 000 – 25 000 A White 7 500 – 12 000 F Yellow - white 6 000 – 7 500 G Yellow 4 500 – 6 000 K Yellow - red 3 000 – 4 500 M Red 2 000 – 3 000 You need to remember the classes and their order Spectral classes Oh be a fine girl….kiss me! More information from spectra The spectrum of a star can have dark absorption lines across it. Each dark line represents the absorption of light at a specific frequency by a chemical element in the star Niels Bohr In 1913, a Danish physicist called Niels Bohr realised that the secret of atomic structure lay in its discreteness, that energy could only be absorbed or emitted at certain values. At school they called me “Bohr the Bore”! The Bohr Model Bohr realised that the electrons could only be at specific energy levels (or states) around the atom. The Bohr Model We say that the energy of the electron (and thus the atom) can exist in a number of states n=1, n=2, n=3 etc. (Similar to the “shells” or electron orbitals that chemists talk about!) n=1 n=2 n=3 The Bohr Model The energy level diagram of the hydrogen atom according to the Bohr model Energy eV 0 High energy n levels are very close to each other n=5 n=4 n=3 n=2 Electron can’t have less energy than this -13.6 n = 1 (the ground state) The Bohr Model An electron in a higher state than the ground state is called an excited electron. Energy eV 0 High energy n levels are very close to each other n=5 n=4 n=3 electron n=2 -13.6 n = 1 (the ground state) Atomic transitions If a hydrogen atom is in an excited state, it can make a transition to a lower state. Thus an atom in state n = 2 can go to n = 1 (an electron jumps from orbit n = 2 to n = 1) Energy eV 0 n=5 n=4 Wheeee! n=3 electron n=2 -13.6 n = 1 (the ground state) Atomic transitions Every time an atom (electron in the atom) makes a transition, a single photon of light is emitted. Energy eV 0 n=5 n=4 n=3 electron n=2 -13.6 n = 1 (the ground state) Atomic transitions The energy of the photon is equal to the difference in energy (ΔE) between the two states. It is equal to hf. ΔE = hf Energy eV 0 n=5 n=4 n=3 electron n=2 ΔE = hf -13.6 n = 1 (the ground state) Atomic transitions An electron can also absorb a photon of the same energy and jump to a hjgher level. Energy eV 0 n=5 n=4 n=3 electron n=2 ΔE = hf -13.6 n = 1 (the ground state) More information from spectra The absorption spectrum thus gives us information about a star’s chemical composition Very hot stars Very hot stars do not show an absorption spectrum as all the gas is ionised so there are no bound electrons orbiting around the nuclei in the star. Thus absorption spectrums can also tell us something about the temperature of a star. Doppler effect on spectra Radial velocity Rotation Different types of stars Binary stars Spectroscopic binaries Eclipsing binaries Eclipsing binaries Cepheids • A type of variable start whose luminosity changes with time (more later!) Red giants and red supergiants • Large in size and red in colour. • Large luminosity • Since they are red, they are comparatively cool. • The source of energy is the fusion of some elements other than hydrogen. White dwarfs • Small and white in colour. • Since they are white they are comparatively hot. • Fusion is no longer taking place, and a white dwarf is just a hot remnant that is cooling down. Hertzsprung – Russell diagram Hertzsprung – Russell diagram • The point of classifying the various types of stars is to see is any patterns exists. A useful way of making the comparison is the H-R diagram. Each dot on the diagram represents a different star. • The vertical axis is the luminosity of the star. It should be noted that the scale is not a linear one. • The horizontal axis is the spectral class of the star in the order OBAFGKM. This is the same as a scale of decreasing temperature. Once again the scale is not a linear one. • The result of such a plot is shown on the next slide Cepheids! Hertzsprung – Russell diagram • A large number of stars the fall on the line that goes from the top left to bottom right. This line is known as the MAIN SEQUENCE and stars that are on it are known as the main sequence stars. Our sun is a main sequence star. These stars are ‘normal’ stable starsthe only difference between them is their mass. They are fusing hydrogen to helium. The stars that are not on the main sequence can also be put into categories. Questions • Page 504 Questions 1, 2, 3, 4, 5, 6, 7, 9.