* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Antiporter-lika proteinsubenheter i andningskedjans Komplex I
Homology modeling wikipedia , lookup
Circular dichroism wikipedia , lookup
Protein domain wikipedia , lookup
Protein folding wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
Bimolecular fluorescence complementation wikipedia , lookup
Protein structure prediction wikipedia , lookup
Protein mass spectrometry wikipedia , lookup
P-type ATPase wikipedia , lookup
Polycomb Group Proteins and Cancer wikipedia , lookup
Protein moonlighting wikipedia , lookup
Trimeric autotransporter adhesin wikipedia , lookup
SNARE (protein) wikipedia , lookup
Protein purification wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
List of types of proteins wikipedia , lookup
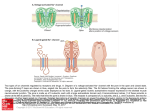
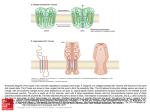
Examensarbete i kemi, naturvetenskapliga fakulteten, Lunds universitet Antiporter-lika proteinsubenheter i andningskedjans Komplex I Benoit Combaluzier All organisms need energy to survive. The energy comes either from sunlight (photosynthesis) or from burning food molecules (respiration), but in both cases the energy is converted to ATP by the organism. ATP is an energy stock that can be used by a multitude of processes in the cell. Eukaryote aerobic organisms produce ATP in the mitochondria by an assembly of protein complexes called respiratory chain or electron transport chain. The respiratory chain is composed by 4 protein complexes which pump protons across the membrane and a fifth complex that uses this proton gradient to build ATP from ADP and Pi. The Complex I, also called NADH:quinone oxidoreductase, is the least understood in the respiratory chain. The bacterial Complex I is composed by 14 protein subunits. The mammalian Complex I has 32 accessory subunits. Since the bacterial Complex I is a simple model, it has been chosen to study the structure and the function of Complex I. It is not understood how the complex works on the molecular level and how electron transfer through Complex I drives proton pumping across the membrane. Three subunits, NuoL, NuoM and NuoN, located in the membrane part of the complex I have structure similarities to one class of antiporters. Antiporters are membrane proteins in charge of transporting ions across the membranes. These antiporters, called MrpA and MrpD, are responsible for salt balance and for maintaining a neutral cytoplasmic pH at alkaline growth conditions. Since NuoL, NuoM and NuoN show similarities to that kind of antiporters, they are most likely involved in the proton transport machinery. NuoL is more similar to MrpA and NuoM and NuoN are more similar to MrpD. Bacillus subtilis lacking the MrpA protein cannot grow at high Na+ concentration but they could be rescued if NuoL protein was expressed in vivo. The same observations were made with MrpD-deleted strains rescued by NuoM. This demonstrated that NuoL and NuoM have Na+ transport ability. In this work, we cloned the NuoN, MrpA and MrpD genes into a plasmid DNA vector. The plasmid containing the gene encoding for NuoN protein could be used to investigate if NuoN has also an ion transport ability and can replace MrpD in B. subtilis lacking MrpD protein. The plasmids containing MrpA and MrpD genes could be used to control that the cells lacking MrpA or MrpD in their genome can retrieve their functionality when these proteins are expressed from the plasmids. Swedish official title: Antiporter-lika proteinsubenheter i andningskedjans Komplex I Swedish credits: 20p E-mail address of first author: [email protected] Supervisor: Cecilia Hägerhäll, Biochemistry Submission date/time: 5/4/2004 Examensarbete i kemi, naturvetenskapliga fakulteten, Lunds universitet Antiporter-like subunits in respiratory chain complex I Benoit Combaluzier Chemistry, Biochemistry Autumn 2003 Abstract in English NADH:quinone oxidoreductase or Complex I is the largest and most complex of the respiratory chain enzymes. The enzyme oxidizes NADH to NAD+ and reduces quinone to quinol. Electron transfer through the enzyme is coupled to proton translocation by an unknown mechanism. The membrane spanning part of the enzyme contains three subunits, NuoL, NuoM and NuoN that show primary sequence similarity to a certain type of antiporters, MrpA and MrpD, and are thus predicted to play a role in the proton translocation machinery. NuoL is more closely related to MrpA and NuoM and NuoN are more closely related to the MrpD antiporter. In a previous work the function of the NuoL and NuoM subunits from Rhodobacter capsulatus Complex I has been investigated by expressing the proteins in Bacillus subtilis strains where the homologous MrpA and MrpD antiporters had been genetically deleted. It appeared that NuoL and NuoM proteins have Na+ translocation ability in vivo. In this work we will clone the MrpA, MrpD and NuoN genes and express the proteins in the two deletion strains. We could then compare the growth of Bacillus subtilis MrpD-deleted strain alone or with expression of NuoN from the pCW6 vector under several pH conditions. We could also check that the functionality is recovered by Bacillus subtilis MrpD-deleted strain when MrpD is expressed from the pCW6 vector. The same study could be carried out with Bacillus subtilis MrpA-deleted strain.