* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Ondansetron and the Risk of Cardiac Arrhythmias: A Systematic
Survey
Document related concepts
Transcript
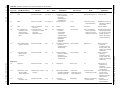
TOXICOLOGY/ORIGINAL RESEARCH Ondansetron and the Risk of Cardiac Arrhythmias: A Systematic Review and Postmarketing Analysis Stephen B. Freedman, MDCM, MSc; Elizabeth Uleryk, BA, MLS; Maggie Rumantir, MD; Yaron Finkelstein, MD* *Corresponding Author. E-mail: yaron.fi[email protected]. Study objective: To explore the risk of cardiac arrhythmias associated with ondansetron administration in the context of recent recommendations for identification of high-risk individuals. Methods: We conducted a postmarketing analysis and systematically reviewed the published literature, grey literature, manufacturer’s database, Food and Drug Administration Adverse Events Reporting System, and the World Health Organization Individual Safety Case Reports Database (VigiBase). Eligible cases described a documented (or perceived) arrhythmia within 24 hours of ondansetron administration. The primary outcome was arrhythmia occurrence temporally associated with the administration of a single, oral ondansetron dose. Secondary objectives included identifying all cases associating ondansetron administration (any dose, frequency, or route) to an arrhythmia. Results: Primary: No reports describing an arrhythmia associated with single oral ondansetron dose administration were identified. Secondary: Sixty unique reports were identified. Route of administration was predominantly intravenous (80%). A significant medical history (67%) or concomitant use of a QT-prolonging medication (67%) was identified in 83% of reports. Approximately one third occurred in patients receiving chemotherapeutic agents, many of which are known to prolong the QT interval. An additional third involved administration to prevent postoperative vomiting. Conclusion: Current evidence does not support routine ECG and electrolyte screening before single oral ondansetron dose administration to individuals without known risk factors. Screening should be targeted to high-risk patients and those receiving ondansetron intravenously. [Ann Emerg Med. 2014;64:19-25.] Please see page 20 for the Editor’s Capsule Summary of this article. A feedback survey is available with each research article published on the Web at www.annemergmed.com. A podcast for this article is available at www.annemergmed.com. 0196-0644/$-see front matter Copyright © 2013 by the American College of Emergency Physicians. http://dx.doi.org/10.1016/j.annemergmed.2013.10.026 INTRODUCTION Background Ondansetron hydrochloride is a potent antiemetic that antagonizes serotonin at 5-hydroxytryptamine3 receptors.1 Between 1995 and 2009, administration in US emergency departments (EDs) increased more than 330-fold, from 38,000 to 12.6 million doses.2 Pediatric usage has also increased, with more than 2 million doses administered to children in US EDs each year, greater than 85% by the oral route.3 In September 2011, the Food and Drug Administration (FDA) issued a communication warning that ondansetron may induce fatal arrhythmias.4 In June 2012, the FDA issued an update linking the risk of QT prolongation to the administration of a 32-mg intravenous dose. Despite this, screening recommendations remain unchanged5-7 and continue to recommend that ondansetron be avoided in patients with congenital long-QT syndrome and that ECG monitoring and serum electrolyte screening be performed in all potentially susceptible patients Volume 64, no. 1 : July 2014 (“electrolyte abnormalities, congestive heart failure, bradyarrhythmias, or patients taking other medicinal products that lead to QT prolongation”).4 Importance The identification of high-risk individuals is important because drugs are frequently administered in the ED without complete knowledge of a patient’s medication or medical history, and the opportunity for monitoring of adverse drug events may be limited.8,9 Consequently, to identify at-risk individuals, diagnostic investigations that might not otherwise be indicated are often required. For example, individuals with vomiting may have electrolyte abnormalities; however, in practice, significant abnormalities are uncommon and testing is not routinely recommended.10 Similarly, long-QT syndrome, a rare and asymptomatic disorder,11 typically remains undiagnosed until a complication occurs. Approximately 16,000 screening ECGs need to be performed to identify a single asymptomatic long-QT syndrome case.12 Annals of Emergency Medicine 19 Ondansetron and Risk of Cardiac Arrhythmias Freedman et al Editor’s Capsule Summary administration of a single oral ondansetron dose. Our secondary objectives were to identify all pediatric (<18 years) and adult (18 years) cases associating ondansetron administration (any dose, frequency, or route) to the development of an arrhythmia and to evaluate causality. What is already known on this topic Recently, the Food and Drug Administration issued a warning that ondansetron may induce fatal arrhythmias, leading to concerns about emergency department use. What question this study addressed This systematic review analyzed available sources to identify cases of arrhythmia temporally associated with the administration of a single oral ondansetron dose. A secondary outcome was identifying all cases associating ondansetron administration to an arrhythmia. What this study adds to our knowledge No reports describing an arrhythmia associated with single oral ondansetron dose administration were identified. How this is relevant to clinical practice This study supports the safety of the clinical practice of administering a single oral ondansetron dose to low-risk patients without the need for screening evaluations. Goals of This Investigation Current regulatory agency communications have resulted in significant uncertainty and confusion among clinicians who use ondansetron frequently.13 Although the ability of high-dose intravenous ondansetron to prolong the QT interval is not in dispute, the need for universal screening is. We sought to identify all reports describing an association between ondansetron administration and arrhythmia occurrence, with a focus on single oral dose administration, a common practice in EDs. MATERIALS AND METHODS Search Strategy We conducted a systematic review of the published literature, in accordance with Preferred Reporting Items for Systemic Reviews and Meta-Analyses guidelines.14,15 Additionally, we explored the grey literature (which includes reports, theses, conference proceedings, translations, bibliographies, and other documents that are “not available through the conventional, commercial distribution channels”)16 and global adverse drug reaction and pharmacovigilance registries to identify all relevant reports. Objectives The primary objective of this study was to identify all reported arrhythmias occurring in temporal association with the 20 Annals of Emergency Medicine Study Selection Eligible cases described a documented arrhythmia or event perceived as an arrhythmia and ondansetron administration up to 24 hours preceding the event. We selected this timeframe to ensure comprehensiveness and the inclusion of all potentially relevant cases catalogued by global pharmacovigilance registries, including those associated with long-term use. Two investigators with expertise in outcomes-based research (S.B.F.) and clinical pharmacology (Y.F.) independently screened all abstracts to assess eligibility. Excluded cases were filed with a reason. Disagreements were resolved through discussion until achievement of consensus. Articles deemed appropriate underwent detailed, full-text review. Adverse drug reaction registry reports were reviewed manually. Data Extraction Cases underwent independent data abstraction by 2 authors (S.B.F., M.R.) using a standardized form. Disagreements were resolved through discussion until achievement of consensus. Information extracted included age, indication, dose, route, concomitant medications, event description and timing, and causal association. The presence of a significant medical history was assessed by the 2 reviewers and required a consensus opinion based on the assumptions that would be expected by a physician providing clinical care and was focused on the presence of preexisting cardiac disease (eg, heart failure, personal or family history suggestive of long-QT syndrome, other arrhythmias) or disorders associated with electrolyte abnormalities (eg, short-gut syndrome, diuretic use, renal disease). Data Sources Published Literature. The search strategy was developed through consultation with a professional research librarian (E.U.) with experience in conducting systematic reviews.17 The strategy contained a broad series of subject headings and keywords relating to ondansetron, arrhythmias, and cardiovascular disease. We ran the initial searches (Appendix E1, Table E1, available online at http://www.annemergmed.com) in October 2011 (most recent update, December 19, 2012), using the OvidSP, Thomson Reuters, and SciVerse search platforms in the following databases: MEDLINE, EMBASE, Web of Knowledge/Science (Conference Proceedings Citation Index), and Scopus. Databases were searched from creation to December 2012, with no language restrictions. The reference lists of all included articles and relevant reviews were hand-searched. Grey Literature. The New York Academy of Medicine Grey Literature Report, FDA Web site (http://www.fda.gov/drugs/ drugsafety/default.htm), OpenGrey (System for Information on Volume 64, no. 1 : July 2014 Freedman et al Grey Literature in Europe; http://www.opengrey.eu/), the Canada Vigilance Adverse Reaction Online Database (http:// www.hc-sc.gc.ca/dhp-mps/medeff/databasdon/index-eng.php), Google Scholar, and the Canadian Agency for Drugs and Health Technologies (http://cadth.ca/en/products) were searched on September 5, 2012, with the search term combination of “ondansetron” AND (QT or arrhythmia*). Global ADR Registries. The World Health Organization (WHO) Global Individual Safety Case Reports Database (VigiBase) registry, which holds more than 3,800,000 reports from national pharmacovigilance centers and regulatory authorities of more than 80 countries,18 was searched on August 30, 2012, with the WHO-adverse drug reaction terminology preferred terms “arrhythmia,” “arrhythmia ventricular,” and “arrhythmia atrial” to extract all suspected or interacting arrhythmia reports occurring within 24 hours of ondansetron administration. A search of the FDA Adverse Events Reporting System was conducted on November 26, 2012, using formal MedDRA nomenclature (version 15.1; Glaxosmithkline, Brentford, Middlesex, United Kingdom). Cases reporting “ondansetron,” “ondansetron hydrochloride,” “ondansetron ODT” or “Zofran” and “Arrhythmia,” including “Torsade de Pointes,” were retrieved. We approached GlaxoSmithKline, the company that developed and markets ondansetron (Zofran), to provide all relevant postmarketing adverse drug reaction reports. Data Synthesis and Analysis The primary outcome was the number of reports describing an arrhythmia after single oral ondansetron dose administration. Secondary outcomes included (1) cases linking the administration of ondansetron by any route, dose, and frequency to the development of an arrhythmia; and (2) the strength of causality. To determine the optimal method of assessing causality, we used the Naranjo ADR Probability Scale (Table E2, available online at http://www.annemergmed.com).19 A boardcertified clinical pharmacologist (Y.F.) with previous experience using the scale20,21 assigned probability scores to all published cases. For cases identified in the WHO registry, we documented the scores provided (ie, Council for International Organizations of Medical Sciences scoring system)22; FDA Adverse Events Reporting System did not provide causality scores. RESULTS Case Selection Published Literature. The electronic database searches identified 1,120 citations (Figure). After removal of duplicates and review of titles and abstracts, 71 articles containing potentially eligible cases remained. Of these, 18 reports describing 21 eligible cases were identified. Reference and text review detected 2 additional cases. In total, 23 cases (6 pediatric; 17 adult) (Table E3, available online at http://www. annemergmed.com) were identified from 20 publications.23-42 Grey Literature. No additional eligible reports were identified. Volume 64, no. 1 : July 2014 Ondansetron and Risk of Cardiac Arrhythmias 1120 Articles identified and screened for retrieval from MEDLINE, EMBASE, Web of Knowledge, SCOPUS 91 1029 Titles and abstracts screened for inclusion 958 71 2 Duplicate articles removed Articles excluded based on title/abstract review Potentially relevant articles retrieved for full-text review Relevant reports identified in reference review 20 53 Excluded 38 review articles 11 Ineligible reports 4 Letters/Comments (no case) Articles (23 cases) describing an arrhythmia or an event perceived as possibly being an arrhythmia in an individual administered ondansetron up to 24 hours prior to the event. Figure. Flow of reports identified in the published literature through the systematic review process. Global ADR Registries. A comprehensive VigiBase search identified 25 reports, and the FDA Adverse Events Reporting System identified 16; 4 were duplicates, resulting in 37 unique reports. Because safety reports are shared by GlaxoSmithKline with regulatory authorities (eg, the FDA), the company stated that querying regulatory data sets would capture all relevant reports. Primary Outcome We did not identify any reports describing the occurrence of an arrhythmia within 24 hours of the administration of a single oral ondansetron dose in any of the sources searched. Secondary Outcomes Eleven (18%) pediatric and 49 (82%) adult cases were identified (Tables 1 and 2). Forty-eight (80%) involved intravenous administration and 2 (3%) involved long-term oral use in patients with multiple arrhythmia risk factors. A significant medical history (67%; 40/60) or concomitant use of QT-prolonging medications (67%; 40/60) was identified in 83% (50/60). Unique (ie, nonoverlapping) indications for use included prevention of postoperative (35%; 21/60) or chemotherapy-induced (35%; 21/60) vomiting. Adult Reports (n¼49). Published Literature (n¼17) Fifteen (88%) of the cases had readily identifiable proarrhythmogenic risk factors (ie, significant medical history or receiving other QTprolonging medications). Median patient age was 46 years (interquartile range [IQR] 36, 60; range 24 to 67 years). Twelve involved intravenous administration24,25,28-35,42; 1, oral administration37; and 4, an unspecified route.36,38-40 The single oral case involved long-term use37 in a patient with acute cardiomyopathy and electrolyte imbalances. Two patients received multiple intravenous doses24,31 and 1 received a large intravenous dose.36 Nine individuals were treated to prevent Annals of Emergency Medicine 21 Freedman et al Ondansetron and Risk of Cardiac Arrhythmias Table 1. Summary of all secondary outcome reports identified from all sources searched. Pediatric (<18 Years) Reports Source Published literature ADR databases VigiBase FAERS† Grey literature Total Adult (‡18 Years) Reports Intravenous Oral Intramuscular Intravenous Oral Not Documented Total 4 1* 1 12 1* 4 23 4 1 0 9 0 0 0 1 0 0 0 1 19 8 0 39 0 0 0 1 2 3 0 9 25 12 0 60 ADR, Adverse drug report; FAERS, FDA Adverse Event Reporting System. *Involved long-term use (ie, multiple doses) in patients with significant arrhythmia risk factors. † A total of 16 cases were identified in FAERS; however, 4 were duplicates of previously identified cases in VigiBase, yielding 12 unique reports. postoperative nausea and vomiting24,25,29,31-33,35,42 and 3 for cancer chemotherapy-associated vomiting.34,36,39 Subjects were receiving a median of 6 additional medications (IQR 2, 8); only 1 did not receive concomitant medications. Nine individuals received other QT-prolonging medications43 and 2 were receiving 3 such medications.28,40 ADR Registries (n¼32). The WHO VigiBase search identified 21 adult reports (patients aged 25 to 70 years; 9 men). Nineteen patients received intravenous ondansetron (4 to 20 mg/day); the other 2 lacked documentation of route of administration. Twelve patients received long-term ondansetron. Nine were oncology patients; 1 had a pacemaker. Nineteen (91%) received concomitant medications (range 1, 7; median 3; IQR 2, 4); 14 (67%) received QT-prolonging agents. Four patients had an ondansetron rechallenge, and none had arrhythmia recurrence. The FDA Adverse Events Reporting System search identified 13 adult reports; 2 were duplicates and were excluded. The 11 remaining adults were aged 20 to 80 years. Eight patients received intravenous ondansetron (1 both intravenously and orally); 3 lacked documentation of route of administration. Ten patients (91%) had significant underlying conditions, including cardiac (eg, cardiomyopathy, myocarditis, long-QT syndrome), oncologic, renal, and metabolic abnormalities. Ten patients (91%) received additional medications (range 1, 24; median 8; IQR 4, 13); 9 included a QT-prolonging medication. The sole case without a reported risk factor involved a 62-year-old administered ondansetron intravenously. Pediatric Reports (n¼11). Published Literature (n¼6). The mean age of pediatric patients was 12.2 years (SD 2.8 years). A single case describing an arrhythmia in a child administered oral ondansetron was identified. This 16-year-old with significant medical problems (eg, leukemia, sepsis, acute tubular necrosis, hypokalemia, hypomagnesaemia) had been administered longterm ondansetron,27 along with 8 additional QT-prolonging agents. Additional cases were associated with intravenous exposure: 3 described arrhythmias in the operating room in children with long-QT syndrome after concomitant administration of multiple QT-prolonging medications.26,41 Another report described a child becoming apneic and bradycardic after intravenous administration.25 Atropine and oxygen administration led to recovery. Last, 3 hours after intramuscular ondansetron injection, a child developed ventricular tachycardia and died.23 Because no other details were provided in association with this report, causality is uncertain and unlikely. Global ADR Registries (n=5). VigiBase search identified 4 pediatric cases (age range 2 to 14 years; United States n¼2; Germany n¼1, Chile n¼1) involving intravenous ondansetron (1 to 24 mg/day). Three of these children received long-term ondansetron; 2 were oncology patients receiving multiple medications, including chemotherapeutics, whereas the third received 3 general anesthetic agents. The FDA Adverse Events Reporting System search identified 3 reports; 2 were duplicates of cases identified in VigiBase. The unique case involved an Table 2. Summary description of arrhythmia risk factors in all secondary outcome reports identified from all studied sources. Pediatric (<18 Years) Reports (n[11) Source Published literature ADR databases VigiBase* FAERS Total (%)† Adult (‡18 Years) Reports (n[49) Significant Medical History Concomitant QT-Prolonging Medication Prevention of PONV Significant Medical History Concomitant QT-Prolonging Medication Prevention of PONV 3 4 4 11 9 9 3 0 6 (55) 3 1 8 (73) 1 1 6 (55) 13 10 34 (69) 14 9 32 (65) 3 3 15 (31) PONV, Postoperative nausea and vomiting. *WHO global individual case safety report database system. † Totals may underrepresent actual frequency of occurrence because all cases with missing or unclear information, particularly the ADR databases, had the item under consideration coded as negative (ie, not present). 22 Annals of Emergency Medicine Volume 64, no. 1 : July 2014 Freedman et al 11-year-old child who received ondansetron and 8 concomitant intravenous medications, including 4 arrhythmogenic anesthetics. Strength of Causality. There were no reported arrhythmias that developed after administration of a single oral ondansetron dose in any of the studied sources (primary outcome). All 23 secondary outcome reports from the literature were assigned low probability Naranjo scores (pediatric 2 to 3, adult 2 to 4; “possible” range). Council for International Organizations of Medical Sciences probability scores were reported in VigiBase for 1 child (“possible”) and 6 adults (5 “possible” and 1 “probable”); the remainder of adverse drug reaction registry cases could not be assigned probability scores because of insufficient data provided to the registry. LIMITATIONS Our study has several potential limitations. First, it is possible that not all cases have been reported. To mitigate this, we used a comprehensive search strategy and a low inclusion threshold of all published and unpublished reports. Second, we used the Naranjo Scale to assess the causality of adverse reactions because it is the most widely accepted causality scoring system and has been in use for more than 30 years.44 During study planning, we conducted a literature search and identified its use in 938 publications, including 45 assessments of drug-induced arrhythmias. However, this score was not designed to evaluate adverse events resulting from the interaction of multiple drugs,45 as was often the case in our reports. Nonetheless, the associations reported for the secondary outcomes were scarce and weak, with original authors usually attributing causality to other factors, such as baseline conditions or concomitant drugs. Although our ability to assess causality related to reports in the unpublished literature was limited because of missing data in some cases, our search yielded a paucity of reports, with none involving a single oral ondansetron dose. Although the FDA warning in 2011 may have limited the use of ondansetron in patients with risk factors, thereby decreasing the risk of complications since its publication, the absence of reports associating single oral dose ondansetron use with arrhythmia throughout the study period implies that such events rarely if ever occur. DISCUSSION Despite extensive use for more than 22 years and several hundred million patient treatment-days,46 we did not find any reports of an arrhythmia occurring in a patient after the administration of a single oral ondansetron dose. Our findings indicate that universal ECG screening and electrolyte testing before ondansetron administration are likely unwarranted. The evidence is particularly weak as it relates to the typical pediatric ED scenario in which a single oral dose is often administered to an otherwise healthy individual who is not prescribed other QT-interval prolonging medications. Individuals at risk of developing an arrhythmia included those receiving ondansetron intravenously in the setting of underlying arrhythmogenic Volume 64, no. 1 : July 2014 Ondansetron and Risk of Cardiac Arrhythmias conditions and those administered other QT-prolonging medications concomitantly. Ondansetron is routinely used, often off label, to treat a variety of ambulatory conditions in the ED, such as gastroenteritisinduced vomiting47 and hyperemesis gravidarum.48-50 The unwarranted withholding of ondansetron when clinically indicated and the performance of universal ECG and electrolyte screening may lead to increased morbidity, resource consumption, impaired ED patient flow, and costs. Recently, a study of 100 children administered therapeutic doses of intravenous ondansetron for the treatment of nausea and vomiting in the ED found no statistical or clinically significant evidence of QT-interval prolongation at peak (3 minutes) or 1 hour postpeak effect.51 The pharmacokinetic differences between oral and intravenous ondansetron administration provide a biologically plausible mechanistic explanation for the absence of reports of arrhythmias after single oral dose administration. The peak serum level after oral administration, the most relevant factor affecting arrhythmia risk, is only 3% to 34% of that achieved after a similar intravenous dose.52,53 Although the maximum QTc lengthening is 17 to 20 msec after intravenous administration of a 32-mg ondansetron dose, it is only 5.8 msec after an 8-mg intravenous dose in adults,54-56 and to our knowledge there are no reports documenting QTc prolongation after oral administration. The time to peak serum level is much longer after oral (2.3 hours) compared with intravenous (5 minutes) administration.53 Recently, the intentional ingestion of 60 mg of ondansetron by a child (ie, 8 times the therapeutic dose) did not result in ECG changes.57 Thus, oral administration of a therapeutic ondansetron dose is highly unlikely to cause a clinically significant prolongation of the QT interval and even less likely to cause an arrhythmia, a finding supported by our analysis. Proposed Approach In accordance with the data presented in this article, it is our opinion that a patient-tailored, evidence-based approach to risk-stratify patients would serve to optimize care. Consideration should be given to performing ECG and electrolyte screening in patients with known arrhythmia risk factors, including cardiac (eg, heart failure, personal or family history suggestive of longQT syndrome, other arrhythmias), medication (eg, multiple dose or intravenous ondansetron, concomitant QT-prolonging or cardiac medications), or electrolyte (eg, short-gut syndrome, diuretic use, renal disease); individuals receiving a single oral dose in the absence of risk factors should not be screened. This balanced approach enables the implementation of regulatory agency recommendations without compromising or delaying care. It minimizes the potential harms and costs (estimated at $5.5 million per 1 true long-QT syndrome case) associated with screening low-risk patients while allowing patients to benefit from an effective medication. In summary, we did not identify any reports of arrhythmias developing in otherwise healthy patients after the administration of a single oral ondansetron dose. Clinicians should be cognizant of ondansetron’s potential to prolong Annals of Emergency Medicine 23 Freedman et al Ondansetron and Risk of Cardiac Arrhythmias the QT interval when administered intravenously, particularly in high doses and in high-risk patients. A risk-stratification strategy should be used to individualize how these recommendations are adapted into clinical practice to identify potentially vulnerable patients who are likely to benefit from screening. In patients with no known risk factors who are administered a single oral ondansetron dose, routine ECG and electrolyte screening are not warranted. to conduct the study or produce the article. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the article. Supervising editor: Matthew D. Sztajnkrycer, MD, PhD Publication dates: Received for publication July 31, 2013. Revision received October 2, 2013. Accepted for publication October 24, 2013. Available online December 4, 2013. Author affiliations: From the Sections of Pediatric Emergency Medicine and Gastroenterology, Alberta Children’s Hospital, Alberta Children’s Hospital Research Institute, University of Calgary, Calgary, Alberta, Canada (Freedman); the Hospital Library and Archives (Uleryk), Child Health Evaluative Sciences (Finkelstein), Hospital for Sick Children Research Institute (Finkelstein, Rumantir), and Divisions of Paediatric Emergency Medicine and Clinical Pharmacology and Toxicology, Department of Paediatrics (Finkelstein), The Hospital for Sick Children, Toronto, Ontario, Canada; and the Faculty of Medicine, University of Toronto, Toronto, Ontario, Canada (Uleryk, Finkelstein). Author contributions: SBF and YF contributed equally to this article and are therefore co-Principal Investigators. SBF and YF had full access to all of the data in the study, take responsibility for the integrity of the data and the accuracy of the data analysis, serve as study guarantors, and were responsible for study concept, statistical analysis, and study supervision. SBF, MR, and YF were responsible for analysis and interpretation of data and administrative, technical, and material support. SBF, EU, and YF were responsible for drafting the article. All authors were responsible for acquisition of data and critical revision of the article for important intellectual content, had full access to all of the data (including statistical reports and tables) in the study, and take responsibility for the integrity of the data and the accuracy of the data analysis. SBF takes responsibility for the paper as a whole. Funding and support: By Annals policy, all authors are required to disclose any and all commercial, financial, and other relationships in any way related to the subject of this article as per ICMJE conflict of interest guidelines (see www.icmje.org). All authors have completed the Unified Competing Interest form at www.icmje.org/ coi_disclosure.pdf (available on request from the corresponding author) and declare that (1) none of the authors have received support for the conduct of the submitted work; (2) Dr. Freedman has a relationship with GlaxoSmithKline that might have an interest in the submitted work in the previous 3 years; (3) the authors’ spouses, partners, or children have no financial relationships that may be relevant to the submitted work; and (4) Drs. Freedman, Finkelstein, and Rumantir and Ms. Uleryk have no nonfinancial interests that may be relevant to the submitted work. Specifically, Dr. Freedman acknowledges receiving in-kind study drug/placebo from GlaxoSmithKline, the manufacturer of ondansetron, for the conduct of an independently funded (Bill and Melinda Gates Foundation and Thrasher Research Fund) clinical trial not related to the current study. No other form of funding or research support was provided by GlaxoSmithKline. The first draft of the article was written by Drs. Freedman and Finkelstein. No honorarium, grant, or other form of payment was given to anyone 24 Annals of Emergency Medicine The data provided by the WHO Collaborating Centre for International Drug Monitoring, Uppsala, are not homogeneous at least with respect to origin or likelihood that the pharmaceutical product caused the adverse reaction, and the information we report from the WHO Collaborating Centre does not represent the opinion of the World Health Organization. Presented as an abstract at the 2013 Pediatric Academic Society annual meeting, May 2013, Washington, DC. REFERENCES 1. Bryson JC. Clinical safety of ondansetron. Semin Oncol. 1992;19:26-32. 2. Tay KY, Ewald MB, Bourgeois FT. Use of QT-prolonging medications in US emergency departments, 1995-2009. Pharmacoepidemiol Drug Saf. 2013;. http://dx.doi.org/10.1002/pds.3455. 3. Sturm JJ, Hirsh DA, Schweickert A, et al. Ondansetron use in the pediatric emergency department and effects on hospitalization and return rates: are we masking alternative diagnoses? Ann Emerg Med. 2010;55:415-422. 4. Food and Drug Administration. FDA drug safety communication: abnormal heart rhythms may be associated with use of Zofran (ondansetron). Available at: http://www.fda.gov/Drugs/DrugSafety/ ucm271913.htm. Accessed April 23, 2013. 5. FDA drug safety podcast for healthcare professionals: new information regarding QT prolongation with ondansetron (Zofran). Available at: http://www.fda.gov/Drugs/DrugSafety/DrugSafetyPodcasts/ ucm310546.htm. Accessed September 5, 2012. 6. Zofran (ondansetron)—association with changes in electrical activity in the heart—for health professionals. Available at: http://www. healthycanadians.gc.ca/recall-alert-rappel-avis/hc-sc/2012/15080aeng.php. Accessed April 23, 2013. 7. Medicines and Healthcare Products Regulatory Agency. Ondansetron (Zofran): risk of QTc prolongation—important new intravenous dose restriction. Available at: http://www.mhra.gov.uk/Safetyinformation/ DrugSafetyUpdate/CON180635. Accessed April 23, 2013. 8. Hohl CM, Dankoff J, Colacone A, et al. Polypharmacy, adverse drugrelated events, and potential adverse drug interactions in elderly patients presenting to an emergency department. Ann Emerg Med. 2001;38:666-671. 9. Banerjee A, Mbamalu D, Ebrahimi S, et al. The prevalence of polypharmacy in elderly attenders to an emergency department—a problem with a need for an effective solution. Int J Emerg Med. 2011;4:22. 10. Olshaker JS, Mason JD. The usefulness of serum electrolytes in the evaluation of acute adult gastroenteritis. Ann Emerg Med. 1989;18:258-260. 11. Garson A Jr, Dick M 2nd, Fournier A, et al. The long QT syndrome in children. An international study of 287 patients. Circulation. 1993;87:1866-1872. 12. Rodday AM, Triedman JK, Alexander ME, et al. Electrocardiogram screening for disorders that cause sudden cardiac death in asymptomatic children: a meta-analysis. Pediatrics. 2012;129:e999-e1010. 13. Bruzzese E, Vecchio AL, Guarino A. Hospital management of children with acute gastroenteritis. Curr Opin Gastroenterol. 2013;29:23-30. 14. Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151:264-269, W64. Volume 64, no. 1 : July 2014 Freedman et al 15. Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009;339:b2700. 16. Alberani V, De Castro Pietrangeli P, Mazza AM. The use of grey literature in health sciences: a preliminary survey. Bull Med Libr Assoc. 1990;78:358-363. 17. Wahi G, Parkin PC, Beyene J, et al. Effectiveness of interventions aimed at reducing screen time in children: a systematic review and metaanalysis of randomized controlled trials. Arch Pediatr Adolesc Med. 2011;165:979-986. 18. Lindquist M. VigiBase, the WHO Global ICSR Database System: basic facts. Drug Inf J. 2008;42:409-419. 19. Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30:239-245. 20. Avner M, Finkelstein Y, Hackam D, et al. Establishing causality in pediatric adverse drug reactions: use of the Naranjo probability scale. Paediatr Drugs. 2007;9:267-270. 21. Rezvani M, Finkelstein Y, Verjee Z, et al. Generalized seizures following topical lidocaine administration during circumcision: establishing causation. Paediatr Drugs. 2007;9:125-127. 22. Meyboom RH, Hekster YA, Egberts AC, et al. Causal or casual? the role of causality assessment in pharmacovigilance. Drug Saf. 1997;17:374-389. 23. Chandrakala R, Vijayashankara C, Kumar K, et al. Ondansetron induced fatal ventricular tachycardia. Indian J Pharmacol. 2008;40:186-187. 24. Havrilla PL, Kane-Gill SL, Verrico MM, et al. Coronary vasospasm and atrial fibrillation associated with ondansetron therapy. Ann Pharmacother. 2009;43:532-536. 25. Afonso N, Dang A, Namshikar V, et al. Intravenous ondansetron causing severe bradycardia: two cases. Ann Card Anaesth. 2009;12:172-173. 26. McKechnie K, Froese A. Ventricular tachycardia after ondansetron administration in a child with undiagnosed long QT syndrome. Can J Anaesth. 2010;57:453-457. 27. Bagatell R, Hainstock M, Lowe MC, et al. The perfect storm: torsades de pointes in a child with leukemia. Pediatr Blood Cancer. 2007;49:996-999. 28. Choo EK, Weber FS, Schmidt TA. Torsade de pointes after administration of droperidol for nausea and vomiting. Prehosp Emerg Care. 2009;13:261-265. 29. Baguley WA, Hay WT, Mackie KP, et al. Cardiac dysrhythmias associated with the intravenous administration of ondansetron and metoclopramide. Anesth Analg. 1997;84:1380-1381. 30. Chakraborti C, Egan J. The lesser of two adverse reactions. Joint Commission J Qual Pat Saf. 2010;36:22-27. 31. Kasinath NS, Malak O, Tetzlaff J. Atrial fibrillation after ondansetron for the prevention and treatment of postoperative nausea and vomiting: a case report. Can J Anaesth. 2003;50:229-231. 32. Moazzam MS, Nasreen F, Bano S, et al. Symptomatic sinus bradycardia: a rare adverse effect of intravenous ondansetron. Saudi J Anaesth. 2011;5:96-97. 33. Dolenska S. Intraoperative cardiac arrest in acquired long QT syndrome. Br J Anaesth. 2009;102:503-505. 34. Perez-Verdia A, Angulo F, Hardwicke FL, et al. Acute cardiac toxicity associated with high-dose intravenous methotrexate therapy: case report and review of the literature. Pharmacotherapy. 2005;25:1271-1276. 35. Bosek V, Hu P, Robinson LA. Acute myocardial ischemia after administration of ondansetron hydrochloride. Anesthesiology. 2000;92:885-887. 36. Ballard HS, Bottino G, Bottino J. Ondansetron and chest pain. Lancet. 1992;340:1107. 37. Purvis JA, Cunningham EL, McGlinchey PG, et al. Drugs, electrolytes and tako-tsubo cardiomyopathy: triple aetiology of acquired long QT syndrome and torsades de pointes. Ulster Med J. 2009;78:188-189. Volume 64, no. 1 : July 2014 Ondansetron and Risk of Cardiac Arrhythmias 38. Hussain A, Ghazal S. After more than 300 defibrillation shocks, patient still alive 12 years later refractory torsade de pointes due to polypharmacy and persistent vomiting. J Saudi Heart Assoc. 2010;22:149-151. 39. Philips JA, Marty FM, Stone RM, et al. Torsades de pointes associated with voriconazole use. Transpl Infect Dis. 2007;9:33-36. 40. Boxall E, Milne J, Peters E, et al. Sudden unexplained death in a patient with HIV and MDR-TB. Journal of the International AIDS Society Conference: 10th International Congress on Drug Therapy in HIV Infection; Conference Start 2010; 20101107; Glasgow, Scotland, November 7-11, 2010. 41. Nathan AT, Berkowitz DH, Montenegro LM, et al. Implications of anesthesia in children with long QT syndrome. Anesth Analg. 2011;112:1163-1168. 42. Saxena A, Chand T, Arya SK, et al. Ondansetron-induced ventricular tachycardia in a patient of caesarian section. J Obstet Anaesth Crit Care. 2012;2:103-104. 43. Roden DM. Drug-induced prolongation of the QT interval. N Engl J Med. 2004;350:1013-1022. 44. Gallagher RM, Kirkham JJ, Mason JR, et al. Development and interrater reliability of the Liverpool adverse drug reaction causality assessment tool. PLoS One. 2011;6:e28096. 45. Kane-Gill SL, Kirisci L, Pathak DS. Are the Naranjo criteria reliable and valid for determination of adverse drug reactions in the intensive care unit? Ann Pharmacother. 2005;39:1823-1827. 46. GlaxoSmithKline Inc., inventor Product Monograph - Zofran (ondansetron hydrochloride dihydrate). Mississauga, ON January 26, 2012. 47. Kharbanda AB, Hall M, Shah SS, et al. Variation in resource utilization across a national sample of pediatric emergency departments. J Pediatr. 2013;163:230-236. 48. Madjunkova S, Maltepe C, Koren G. The leading concerns of American women with nausea and vomiting of pregnancy calling Motherisk NVP Helpline. Obstet Gynecol Int. 2013;2013:752980. 49. Pasternak B, Svanstrom H, Hviid A. Ondansetron in pregnancy and risk of adverse fetal outcomes. N Engl J Med. 2013;368:814-823. 50. Raymond SH. A survey of prescribing for the management of nausea and vomiting in pregnancy in Australasia. Aust N Z J Obstet Gynaecol. 2013;53:358-362. 51. The effect of intravenous ondansetron on the QT interval of patients’ electrocardiograms in the pediatric emergency department. Available at: http://www2.aap.org/sections/pem/PDF/ 2013SOEMNCEProgram7-13.pdf. Accessed July 29, 2013. 52. Pritchard JF, Bryson JC, Kernodle AE, et al. Age and gender effects on ondansetron pharmacokinetics: evaluation of healthy aged volunteers. Clin Pharmacol Ther. 1992;51:51-55. 53. VanDenBerg CM, Kazmi Y, Stewart J, et al. Pharmacokinetics of three formulations of ondansetron hydrochloride in healthy volunteers: 24-mg oral tablet, rectal suppository, and i.v. infusion. Am J Health Syst Pharm. 2000;57:1046-1050. 54. Charbit B, Albaladejo P, Funck-Brentano C, et al. Prolongation of QTc interval after postoperative nausea and vomiting treatment by droperidol or ondansetron. Anesthesiology. 2005;102:1094-1100. 55. Charbit B, Alvarez JC, Dasque E, et al. Droperidol and ondansetroninduced QT interval prolongation: a clinical drug interaction study. Anesthesiology. 2008;109:206-212. 56. GlaxoSmithKline Inc. A randomized, double-blind, four-period crossover study to investigate the effect of intravenous ondansetron, a 5-HT3 antagonist, on cardiac conduction as compared to placebo and moxifloxacin in healthy adult subjects. Available at: http://www. gsk-clinicalstudyregister.com/result_detail.jsp?protocolId¼ 115458&studyId¼9B2760D1-7F0C-4871-992F-EAF86227CE3E& compound¼ondansetron. Accessed October 1, 2012. 57. Ghafouri N, Darracq MA, Cantrell FL. Ondansetron-associated hypotension following pediatric self-poisoning. Pediatr Emerg Care. 2012;28:596-597. Annals of Emergency Medicine 25 Ondansetron and Risk of Cardiac Arrhythmias APPENDIX E1. ONDANSETRON DRUG SAFETY COMMUNICATION The U.S. Food and Drug Administration (FDA) is informing the public of an ongoing safety review of the antinausea drug Zofran (ondansetron, ondansetron hydrochloride, and their generics). Ondansetron may increase the risk of developing abnormal changes in the electrical activity of the heart, which can result in a potentially fatal abnormal heart rhythm. Changes in the electrical activity of the heart (prolongation of the QT interval of the ECG)—see data summary below—can lead to an abnormal and potentially fatal heart rhythm (including torsade de pointes). Patients at particular risk for developing torsade include those with underlying heart conditions, such as congenital long-QT syndrome, those who are predisposed to low levels of potassium and magnesium in the blood, and those receiving other medications that lead to QT prolongation. FDA has reviewed all available information and is making interim changes to the drug labels. The manufacturer of Zofran (GlaxoSmithKline) is being required to conduct a thorough QT study to assess the potential for the drug to prolong the QT interval. The results from this study are expected to be available in the summer of 2012. Additional label changes may result after the additional information has been reviewed. The Zofran (ondansetron) drug labels already contain information about the potential for QT prolongation. The labels are being revised to include a warning to avoid use in patients with congenital 25.e1 Annals of Emergency Medicine Freedman et al long-QT syndrome because these patients are at particular risk for torsade. Additionally, recommendations for ECG monitoring in patients with electrolyte abnormalities (eg, hypokalemia, hypomagnesemia), congestive heart failure, or bradyarrhythmias, or in patients receiving other medications that can lead to QT prolongation, are being included in the labels. Additional Information for Health Care Professionals ECG changes including QT-interval prolongation have been observed in patients receiving Zofran (ondansetron). In addition, torsade de pointes, an abnormal heart rhythm, has been reported in some patients receiving ondansetron. The use of Zofran (ondansetron) should be avoided in patients with congenital long-QT syndrome. ECG monitoring is recommended in patients with electrolyte abnormalities (eg, hypokalemia, hypomagnesemia), congestive heart failure, or bradyarrhythmias, or patients receiving concomitant medications that prolong the QT interval. Advise patients to contact a health care professional immediately if they experience signs and symptoms of an abnormal pulse rate or rhythm while receiving Zofran (ondansetron). Report adverse events involving Zofran (ondansetron) to the FDA MedWatch program, using the information in the “Contact Us” box at the bottom of the page. Volume 64, no. 1 : July 2014 Freedman et al Volume 64, no. 1 : July 2014 Table E1. Search strategies. Set History Results Annals of Emergency Medicine 25.e2 Ondansetron and Risk of Cardiac Arrhythmias Databases: OvidSP, MEDLINE, 1946 to April 13, 2012. Initial search: October 12, 2011. Update search: December 11, 2012. 1 Ondansetron/or Ondansetron (nm) or (ondansetron or “gr38032f” or “gr 38032f” or “sn 307” or sn307 or “gr-38032f” or “sn-307” or bryterol or 3,452 cedantron or ceramos or emeset or “c507 75” or narfoz or onsia or sakisozin or vomceran or zetron or zofran or zydis or zofrene or zofron or zophran or zophren).mp. 2 exp Arrhythmias, Cardiac/or exp cardiovascular diseases/ 1,775,166 3 1 and 2 87 Databases: OvidSP, EMBASE (1980 to 2012 week 14). Initial search: October 12, 2011. Update search: December 11, 2012. 2,862 1 Ondansetron/po or (po.fs. and (ondansetron or “gr38032f” or “gr 38032f” or “sn 307” or sn307 or “gr-38032f” or “sn-307” or bryterol or cedantron or ceramos or emeset or “c507 75” or narfoz or onsia or sakisozin or vomceran or zetron or zofran or zydis or zofrene or zofron or zophran or zophren).mp.) 2 exp heart arrhythmia/ 2,634,057 3 1 and 2 2,387 Databases: ISI Web of Knowledge, conference proceedings, Citation Index–Science (CPCI-S) 1990 to present, and conference proceedings, Citation Index–Social Science & Humanities (CPCI-SSH) 1990 to present. Initial search: October 12, 2011. Update search: December 11, 2012. 1 TS¼(ondansetron OR bryterol OR cedantron OR ceramos OR emeset OR narfoz OR onsia OR sakisozin OR vomceran OR zetron OR zofran OR zydis 404 OR zofrene OR zofron OR zophran OR zophren) 2 ts¼arrythmia* 80 3 #1 and #2 0 Databases: SciVerse, Scopus to October 12, 2011. Initial search: October 12, 2011. Update search: December 11, 2012. 1 (ondansetron OR “gr38032f” OR “gr 38032f” OR “sn 307” OR sn307 OR “gr-38032f” OR “sn-307” OR bryterol OR cedantron OR ceramos OR 17 emeset OR “c507 75” OR narfoz OR onsia OR sakisozin OR vomceran OR zetron OR zofran OR zydis OR zofrene OR zofron OR zophran OR zophren) AND (arrhythmia*) AND (LIMIT-TO(DOCTYPE, “cp”)) Freedman et al Ondansetron and Risk of Cardiac Arrhythmias Table E2. Elements and point values of the Naranjo Adverse Drug Reaction Probability Scale. Score Criteria Yes No Do Not Know Are there previous conclusive reports on this reaction? Did the adverse event occur after the suspected drug was administered? Did the adverse reaction improve when the drug was discontinued or a specific antagonist was administered? Did the adverse reaction reappear when the drug was readministered? Are there alternative causes (other than the drug) that could have on their own caused the reaction? Did the reaction reappear when a placebo was given? Was the drug detected in the blood (or other fluids) in concentrations known to be toxic? Was the reaction more severe when the dose was increased or less severe when the dose was decreased? Did the patient have a similar reaction to the same or similar drugs in any previous exposure? Was the adverse event confirmed by any objective evidence? þ1 þ2 þ1 þ2 1 1 þ1 þ1 þ1 þ1 0 1 0 1 þ2 þ1 0 0 0 0 0 0 0 0 0 0 0 0 0 0 25.e3 Annals of Emergency Medicine Volume 64, no. 1 : July 2014 Age, Years Past Medical History Indication Dose Route Prevention of PONV 0.1 mg/kg 825 None Prevention of PONV 0.13 mg/kg IV 1423 None 1627 ALL, pseudomonas bacteremia, acute tubular necrosis, Clostridium difficile colitis Vomiting, abdominal 4 mg pain8 h Intractable emesis N/A secondary to C difficile colitis and Helicobacter pylori 1341 LQTS, Andersen-Tawil (LQTS 7) syndrome Prevention of PONV N/A IV 1141 Jervell and LangeNielsen (LQTS) syndrome Prevention of PONV N/A Prevention of PONV Adult cases 2442 Pregnancy IV Time to Event Event Explanation Sevoflurane, nitrous oxide, remifentanil, rocuronium, dexamethasone, dimenhydrinate Not documented 2 min PVCs developing into VT Congenital LQTS 2 min Unconscious, apnea, severe bradycardia IM Antacid 300 min VT then VF Bradycardia as a result of attenuation of BezoldJarisch reflex Autopsy declined PO Clarithromycin, amoxicillin, metronidazole, trimethoprimsulfamethoxazole, diphenhydramine, hydroxyzine, pantoprazole, potassium Sevoflurane, desflurane, vecuronium, anticholinesterase, anticholinergic “Several hours” PVCs, prolonged QT Hypokalemia (2.0 mEq/L) interval, ventricular and hypomagnesemia (1.1 mg/dL) as a result ectopy; 60 min later developed VT then TdP of renal dysfunction, diuretic use, diarrhea; multiple medications “Close proximity” Bigeminy, VT IV Sodium pentothal, propofol, isoflurane, pancuronium anticholinesterase, anticholinergic “Close proximity” TdP 4 mg IV Bupivacaine Within 1 min VT 4728 Hypertension, aortic stenosis, GERD, anxiety, depression Abdominal pain, nausea, 4 mg vomiting IV HRT, morphine sulphate, “Shortly after” cimetidine, nortriptyline, droperidol; 30 min fluoxetine, droperidol after ondansetron 3729 Leg injury, hematocrit¼24% Prevention of PONV IV Oxycodone, metoclopramide 4 mg Preoperative During emergence (increased sympathetic activity), in LQTS patient, with multiple QT-prolonging medications During emergence (increased sympathetic activity), in LQTS patient, with multiple QT-prolonging medications Adverse effect of ondansetron or inadvertent intravascular injection of bupivacaine Ventricular ectopy Stress cardiomyopathy, followed by VT or TdP coronary artery disease with TdP related to droperidol administration Bigeminy, ST depression, Concurrent T-wave inversion administration of metoclopramide Ondansetron and Risk of Cardiac Arrhythmias Annals of Emergency Medicine 25.e4 Pediatric cases 1126 None Concomitant Medications Freedman et al Volume 64, no. 1 : July 2014 Table E3. Pediatric and adult case reports identified in the literature. Age, Years Past Medical History Indication Dose Route Concomitant Medications Time to Event Event Reduction of bilateral calcaneal fractures, palpitations Prevention of PONV 2 mg IV 6025 Gastric carcinoma, hemoglobin¼7 g/dL Erosive gastritis, GERD, hypertension Prevention of PONV 4 mg IV Nausea, vomiting, anorexia N/A IV 5124 Hyperlipidemia Prevention of PONV 4 mg2 IV 4731 Benign breast lump Prevention of PONV 4 mg2 IV 4332 Cholecystitis Prevention of PONV 4 mg IV 3533 Asthma Prevention of PONV 4 mg IV Fentanyl, propofol, Immediately after 5 mL cocaine paste, nitrous of 2% lidocaine with oxide, sevoflurane, epinephrine (1 in diclofenac, lidocaine 80,000) with epinephrine VT, VF 3634 Osteosarcoma Medication induced nausea and vomiting 8 mg IV Bradycardia, junctional escape beats, VT 6035 Pulmonary adenocarcinoma Prevention of PONV 2 mg IV Cisplatin, doxorubicin, 2 h postinitiation of sodium bicarbonate, methotrexate infusion methotrexate, leucovorin, lorazepam, dexamethasone, enoxaparin, pantoprazole, sertraline, zolpidem Ropivacaine, fentanyl, Immediate droperidol 6236 Locally advanced pancreatic carcinoma Medication-induced nausea and vomiting 15 mg N/A 4630 Propofol, lidocaine, Immediate fentanyl, nitrous oxide, succinylcholine, cefazolin, desflurane, metoclopramide Glycopyrrolate 2 min Valsartan, pantoprazole, hydrochlorothiazide, midazolam, fentanyl Atorvastatin, midazolam, multivitamin, glucosamine, chondroitin, cefazolin, rocuronium, isoflurane, propofol, fentanyl, glycopyrrolate Midazolam, propofol, isoflurane, nitrous oxide, fentanyl, ketorolac None Explanation Bradycardia, junctional rhythm, VT, SVT Previous administration of metoclopramide Bradycardia Attenuated BezoldJarisch reflex Idiosyncratic adverse effect 60 min Second-degree AV block Immediately after second dose ST-segment elevation and alternans, atrial fibrillation Coronary artery vasospasm “probably” related to ondansetron 15 min after second dose Atrial fibrillation Cannot conclusively establish causality 2 to 3 min Sinus bradycardia Attenuated BezoldJarisch reflex Injection of epinephrine after recent cocaine paste application, during volatile anesthetic administration in patient with LQTS “Probable” relationship between cardiotoxicity and methotrexate Fluorouracil, doxorubicin, N/A dexamethasone, diphenhydramine SVT, PVCs, VT Ventricular arrhythmia Ondansetron-induced acute myocardial ischemia Association is not proof of a relationship to ondansetron Freedman et al Volume 64, no. 1 : July 2014 3429 Ondansetron and Risk of Cardiac Arrhythmias 25.e5 Annals of Emergency Medicine Table E3. Continued. Depression, ileostomy, diverticular disease Long-term nausea treatment 4 mg BID PO Fluoxetine N/A TdP 3638 Hysterectomy, nonulcer dyspepsia, GERD Infected rectus sheath hematoma N/A N/A N/A VT, TdP 6239 AML, invasive Aspergillus Medication-induced nausea and vomiting infection, pleural effusions, cardiomyopathy 8 mg Qday N/A N/A TdP 2540 TB, HIV N/A N/A Loperamide, metronidazole, nitrofurantoin, trimethoprimsulfamethoxazole, tetracycline, metoclopramide, ranitidine, cisapride Daunorubicin, cytarabine, furosemide, amphotericin b, voriconazole, esomeprazole Amikacin, linezolid, moxifloxacin, prothionamide, ethambutol, cycloserine, pyrazinamide, darunavir-ritonavir, clofazamine, enoxaparin, valganciclovir, fluconazole N/A Fatal arrhythmia Long-term nausea treatment Hypomagnesemia, daily ondansetron and fluoxetine, cardiomyopathy Multifactorial cause including cisapride, drug-drug interaction, and electrolyte disturbance Freedman et al Volume 64, no. 1 : July 2014 6737 Voriconazole caused QT prolongation in setting of cardiomyopathy, electrolyte disturbance, and esomeprazole Medications causing QT prolongation, HIV, and TB infection IV, Intravenous; PVC, premature ventricular contraction; VT, ventricular tachycardia; LQTS, long-QT syndrome; IM, intramuscular; VF, ventricular fibrillation; ALL, acute lymphoblastic leukemia; N/A, not available; PO, per os; TdP, torsades de pointes; GERD, gastroesophageal reflux; HRT, hormone replacement therapy; SVT, supraventricular tachycardia; AV, atrioventricular; BID, twice daily; AML, acute myelogenous leukemia; Qday, daily; TB, tuberculosis. Ondansetron and Risk of Cardiac Arrhythmias Annals of Emergency Medicine 25.e6