* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Sternal Talon® Implants
Survey
Document related concepts
Transcript
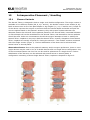
Sternal Talon® Implants
Instructions for Use
V 3.0-EN (09.11)
Instructions for Use
Sternal Talon® Implants
Symbol Explanations
Safety alert symbol
CAUTION:
WARNING:
DANGER:
Indicates a situation which, if not avoided, could result
in minor or moderate injury.
Indicates a situation which, if not avoided, could result
in death or serious injury.
Indicates a situation which, if not avoided, will result in
death or serious injury.
Symbol for "Observe Instructions for Use!"
Symbol for "ORDER NUMBER" / "ARTICLE NUMBER"
Symbol for "LOT DESIGNATION"
Symbol for "NOT TO BE REUSED!"
Symbol for "NON-STERILE"
Symbol for "STERILIZATION USING IRRADIATION"
Symbol for "NOT TO BE RESTERILIZED!"
Symbol for "MANUFACTURING DATE"
Symbol for "DO NOT USE IF PACKAGING IS DAMAGED!"
Symbol for "USE BEFORE"
2
V 3.0
Instructions for Use
Sternal Talon® Implants
Table of Contents
1
General Information .................................................................... 5
2
Safety Instructions / Signal Words................................................. 5
3
Product Liability & Warranty ......................................................... 6
3.1
3.2
3.3
Intended Use ........................................................................................ 6
Warranty ............................................................................................. 6
Hotline................................................................................................. 7
4
Users ......................................................................................... 7
5
Material ..................................................................................... 8
6
Intended Purpose / Indication ....................................................... 8
7
Contraindications ........................................................................ 9
8
Side Effects and Adverse Interactions ........................................... 10
9
Cautionary Notes ....................................................................... 11
10
Packaging.................................................................................. 13
11
Sternal Talon® System Components ............................................. 14
12
Symbols on Product .................................................................... 16
13
Sternal Talon® Storage ............................................................... 17
13.1
13.2
Sternal Talon® Storage Tray ................................................................. 17
Sternal Talon® Implant Storage Box ...................................................... 19
14
Sterility, Cleaning and Disinfection ............................................... 22
15
Intraoperative Placement / Handling ............................................. 24
15.1
15.2
15.3
15.4
15.5
15.6
15.7
Closure Variants ................................................................................. 24
Sternum Measurement for Correct Size Selection .................................... 25
Preparing Site for Sternal Talon® Insertion ............................................. 28
Placing the Sternal Talon® .................................................................... 30
Closing the Sternal Talon® ................................................................... 34
Locking the Sternal Talon® ................................................................... 36
Removing the Sternal Talon® in an Emergency Immediately After the
Operation ........................................................................................... 37
V 3.0
3
Instructions for Use
Sternal Talon® Implants
16
Hospital and Patient Equipment .................................................... 39
17
Removing the Sternal Talon® in a Re-Operation Following Bony
Consolidation ............................................................................. 40
18
Disposal .................................................................................... 41
19
Troubleshooting ......................................................................... 41
20
Accessories................................................................................ 42
21
Contact Details .......................................................................... 43
4
V 3.0
Instructions for Use
Sternal Talon® Implants
1
General Information
WARNING
Non-observance of these Instructions for Use could lead to serious or even lethal
patient injury!
Be sure to read, understand and follow the instructions given below!
Every user must read and follow the Instructions for Use in full.
In particular pay attention to all cautionary, warning and hazard notices.
The instructions for use must be accessible by the user at all times. These Instructions for
Use must be kept accessible to all relevant user groups of the hospital. This especially
includes all users and all persons and departments involved in the intended use and proper
storage, processing and disposal of the product.
The product and its accessories may be used only by persons who have been adequately
trained and have the necessary knowledge and experience with the system.
The user is responsible for ensuring that the patient is informed about and has understood
the information regarding application, risks, warning and hazard notices contained in these
Instructions for Use.
The present text applies equally to both male and female persons. For reasons of legibility
alone, we refrain from referring in every case to both sexes.
2
Safety Instructions / Signal Words
Throughout the manual, the following terms are used to identify advices and precautions that
will help avoid accidental injury to patients or personnel:
NOTICE
Risk of material damage
Indicates a situation which, if not avoided, could lead to material damage (loss of time, data
loss, device/machine failure, etc.)!
WARNING
Danger of death or serious injury!
Indicates a situation which, if not avoided, could result in death or serious injury!
V 3.0
5
Instructions for Use
Sternal Talon® Implants
3
Product Liability & Warranty
3.1
Intended Use
Sternal Talon® is an advanced system for rigid sternal fixation.
Various surgical techniques and materials are optionally available for implant fixation. It is
the user’s (treating physician’s or surgeon’s) responsibility to determine and select the
correct type and configuration of the Sternal Talon® system to be used in compliance with
anatomical and functional patient requirements. The same applies when using the special
instruments and accessories of the Sternal Talon® system.
KLS Martin shall not be responsible for complications caused by wrong indications, wrong
implant selection, incorrect combination of system components, use of inadequate
operating techniques, limitations of the treatment used, or lack of hygiene.
All Sternal Talon® system components, instruments and accessories must be available in
ready-for-use condition.
The product must be used according to indication.
3.2
Warranty
Our Standard Terms and Conditions of Sale effective at the time shall apply. Agreements
diverging from these Standard Terms and Conditions do not restrict the legal rights of the
buyer.
Any warranty exceeding the above provisions shall require a contractual form and shall exclude
component-related vandalism and consumables.
Important Notices
The product may only be repaired by Gebrüder Martin or a qualified person or firm expressly
authorized by Gebrüder Martin to perform such work.
If the repair is carried out by a person or firm specially authorized by Gebrüder Martin, the
operator of the product is required to obtain from the repairer a certificate with details about
the nature and scope of the repair work done. This certificate must be dated and signed and
include the firm’s details.
In all cases where a party other than the product manufacturer performed the work, repaired
products must be additionally marked with the repairer’s ID label.
Improper interventions or alterations performed by third parties during the period of limitation
shall void any and all warranty claims. Unauthorized actions performed on the product shall
invalidate any liability claims vis-à-vis Gebrüder Martin.
6
V 3.0
Instructions for Use
Sternal Talon® Implants
3.3
Hotline
Should you have any questions on how to handle the unit/product or question on its
clinical application, please do not hesitate to contact the Product Management
Tel.:
+49 7461 706-234
Fax:
+49 7461 706-312
4
Users
The user is responsible for the correct surgical procedure, as well as for the prevention or
reduction of general risks during surgical procedures. The user must be theoretically and
practically trained in the surgical technique. He is responsible for the correct compilation of
implant components and their correct insertion. He is also responsible for complications
caused by an incorrect diagnosis or an incorrect surgical technique.
The user and the surgical team must be fully familiar with the surgical procedure, the
implant set and the respective instruments. All relevant information must be fully
accessible on site at all times.
All rules of medical practice as well as the state of the art and the contents of relevant
scientific articles published by medical authors must be known.
Before surgery the user must ensure that sufficient Sternal Talon ® implants are available,
that surgery can be performed under aseptic conditions and that all required Sternal
Talon® and other instruments are fully and functionally available in sterile condition.
Before using the equipment, all personnel should become familiar with its handling.
Personnel included in this process are of central sterilization staff, members of the surgical
teams, nurses, ICU staff and the bioengineering departments.
The user must ensure that the products of the Sternal Talon ® hospital kit and the
Sternal Talon® patient kit (refer to section 16 a. and b.) are available according to need.
V 3.0
7
Instructions for Use
Sternal Talon® Implants
5
Material
Implants:
The Sternal Talon® implants are made of a Ti6AL4V titanium alloy according to ISO 5832-3.
This titanium alloy is a biocompatible material.
Instruments:
The instruments are made of stainless steel.
NOTICE
Potential misinterpretation of examination results!
Implant systems can cause irritating artifacts in CT scans and MRIs.
WARNING
Danger of burns or accidental implant movement when using magnetic resonance
imaging (MRIs)!
Due to the further development and increasingly higher energy density of MRI systems, an
adverse effect on implants cannot be ruled out in the future. Therefore, MRIs are not
permitted unless potential patient injury can definitely be ruled out.
Due to the magnetic field, MRIs pose a danger of heating up or dislocating potentially
susceptible implants.
6
Intended Purpose / Indication
The Sternal Talon® is intended for use in primary and secondary closure of the sternum
following midline sternotomy to stabilize the sternum and promote fusion. Moreover, the
Sternal Talon® is effective for reconstruction procedures and improves sternal stability in nonunions.
Sternal Talon® is an advanced system for rigid sternal fixation. In this procedure, Single and
Double Sternal Talons are used to pull the two sternal halves together by way of a ratchet
mechanism. A position screw on the anterior surface of the device rotates from "Open" to
"Closing" to "Locked" position. By pressing the two implant halves medially together using the
instrument supplied with the product, complete sternal closure can be achieved in a short
time.
The system can be used for cardiothoracic patients but is especially suitable for those at high
risk resulting from:
Morbid obesity
Diabetes
Chronic obstructive pulmonary disease
8
V 3.0
Instructions for Use
Sternal Talon® Implants
7
Contraindications
WARNING
Non-observance of these Contraindications could lead to serious or even lethal
patient injury!
The absolute contraindications for this implant system include:
Patients that are still growing
Lack of bone tissue
Sternal anomalies preventing the use of Sternal Talon ® implants
Manifest infections
Patients with latent infection
Inflammations in the implant region
Bone tumors located in the implant region
Hypersensitivity to foreign bodies
Suspected sensitivity to the implant materials
Allergies against the implant materials
Interventions carried out in a non-sterile environment
The relative contraindications for this implant system include:
Osteoporosis or osteomalacia or other severe structural bone damage
Parasternal sternotomy with very low sternal width on one side
Patients unwilling or unable to follow instructions during the postoperative phase due to
their mental, neurological or physical condition
Reduced compliance due to obvious drug or alcohol abuse
The final decision about the patient-specific suitability of the Sternal Talon® system rests with
the surgeon.
V 3.0
9
Instructions for Use
Sternal Talon® Implants
8
Side Effects and Adverse Interactions
General known risks
Any invasive surgical procedure has risks such as infection, hematoma, dysesthesia (changes
in sensation), vascular and nerve injuries, postoperative pain, and delayed wound healing.
Smoking may significantly increase the risk of delayed wound healing.
Specific known risks
In addition to posing general risks, the Sternal Talon® poses specific risks, including:
Tissue damage
Tissue discoloration
Extrusion
Non-union or delayed union
Necrosis of bone
Calcification underneath the implant contact points
Decrease in bone density due to stress shielding
Pain, discomfort or abnormal sensation
Selection of a Sternal Talon® with foot plates that are too long, relative to the depth of the
sternum (sternal thickness), may damage (squeeze) internal chest wall structures, e.g.
vessels, pleura, etc.
Damage of the internal mammary artery or other vessels
Early and late infections, surface and/or deep infections associated with high lethality
Nerve injuries
Hematomas and slow wound healing
Sternal Talon® implants might be palpable
Allergic response or hypersensitivity to the implant material
Increased reaction of the fibrous tissue in the region of the sternotomy and / or implant
Keloid formation in the scar area
Formation of seromas
Poor healing
Insufficient tissue coverage of implant
Wear and tear, fracture, deformation, loosening or migration of the implant due to impact
of inadequately high forces or excessive weight loads
Implant loosening, implant cutting into bone or surrounding tissue, and formation of sternal
fractures due to exposure to inadequately high forces or excessive weight loads and/or
poor bone quality
10
V 3.0
Instructions for Use
Sternal Talon® Implants
Sternal fractures caused by patient falling down
Fragments and microscopically small particles may detach from the fixation implant and
move away from the implantation site. Metal particles may also remain in the patient’s
body even after the removal of a metal implant. The long-term effects of such particles are
presently unknown.
Apart from the side effects described above, it should be noted that any surgical intervention
could lead to complications not necessarily caused by the implant system, such as infections,
nerve injuries and pain.
9
Cautionary Notes
WARNING
Improper handling and care as well as non-intended use can lead to premature
wear and/or pose a risk for patients and users!
Be sure to read, understand and follow the instructions given below!
Prior to use, inspect the Sternal Talon® for: loose, bent, broken, cracked, worn or fractured
components.
Make sure that the Sternal Talon® implants to be used are neither deformed nor damaged
in any other way.
Never use the product if it is damaged or defective in any way. Set aside the product if it is
damaged.
Do not use the system if malfunction becomes apparent during the operation.
Do not use the system if there is reason to suspect that it has been damaged by being
dropped or mishandled.
The implant must not be adapted to bone contours (no bending whatsoever!).
Aseptic operating conditions are a prerequisite.
Once the Sternal Talon® implants are in place, the stability of the sternum fixation must be
checked.
Implants that have been removed may never be reimplanted. Even if the implant is
apparently intact, there may still be small defects and internal weak points that might lead
to failure or fracture of the fixing implant.
Adequate patient instruction and patient compliance are essential factors for the success of
the surgical intervention. Therefore, postoperative guidance is extremely important. The
patient must know that a metal implant is less resistant than normal bone tissue Excessive
physical activities or carrying loads may lead to implant loosening, migration, deformation
or fracture.
V 3.0
11
Instructions for Use
Sternal Talon® Implants
As with any implant, there is a danger of contamination by foreign matter and coarse
particles such as glove talc or lint from drapes, cleaner residues or other surface
impurities. Therefore, the implants should be touched as little as possible. Implants that
have come in contact with body liquids may never be reused!
Implants are intended for supporting the healing process in bone tissue, they are not
substitutes for normal tissue. Be sure to inform the patient about the risks and potential
side effects associated with the use of implants.
Exposing the fixed sternum to excessive loads must be avoided after the operation.
If the sternum is exposed to abnormal loads (e.g. in wheelchair drivers or patients with
walking aids) or if bone quality is poor, there is an increased risk of implant loosening,
implant cutting into bone and development of sternal fractures.
Exposure to continuous vibrations must be avoided after the operation – e.g. during
rehabilitation measures or when operating machines (drilling machines, vibrating grinders,
etc.).
12
V 3.0
Instructions for Use
Sternal Talon® Implants
10
Packaging
After product delivery, check the original packaging for damage.
Implants delivered non-sterile: Products with damaged PE pouches cannot be taken back.
Implants delivered sterile: Products with damaged blister packs cannot be taken back.
Sternal Talon® implants delivered non-sterile must be removed from their packaging prior
to cleaning and sterilization.
The transportation/storage packaging of sterile implants is non-sterile. Therefore, sterile
implants must be unpacked outside of the sterile OR area and transferred to the sterile
environment in impeccably sterile condition.
Implants packaged with the label
can be used directly from their sterile packaging
without further preparatory treatment. The expiry date is printed on the outer protective
packaging.
Sterile implants with (even unintentionally) opened or damaged sterile packaging, as well
as implants removed from packaging with an elapsed expiry date, must be regarded as
non-sterile and must be subjected to a validated treatment procedure before use.
NOTICE
All packaging is marked with a lot number.
In case of complaints, always specify this number together with the article number.
The use of any implant components should be documented in the patient record, complete
with article number, item designation, lot and/or serial number (if available). Be aware that
reliable traceability will be guaranteed only if this information is available from the patient
file.
V 3.0
13
Instructions for Use
Sternal Talon® Implants
11
Sternal Talon® System Components
The device is available in a Single and a Double version. All Sternal Talon® implants consist of
two parts, a male and a female portion.
Single Sternal Talon®
Double Sternal Talon®
(two legs)
(four legs)
female
–
male
female
–
male
Thanks to a ratchet mechanism, opening and closing of the implants is an easy process. The
front side of the implant features a blue screw offering three different setting options
(positions); depending on the position selected, the ratchet mechanism engages or
disengages. Rotating the screw clockwise to the 7 o’clock ("Open") position unlocks the ratchet
mechanism and the two parts of the implant can then be disconnected from each other.
Rotating the screw counterclockwise to the 3 o’clock ("Closing") position engages the lock
mechanism. At the 11 o'clock ("Locked") position, the implant is securely locked.
Single Sternal Talon®
The Single Sternal Talon® implants have two legs and are available in five widths – XS, S, M,
L, XL – and four foot plate depths: 11 mm, 14 mm, 17 mm and 20 mm.
Large
(34–45mm)
Xtra-Large
(46–52 mm)
11 mm
24-008-11-71
24-009-11-71
24-010-11-71
24-011-11-71
-
14 mm
24-008-14-71
24-009-14-71
24-010-14-71
24-011-14-71
24-012-14-71
17 mm
-
24-009-17-71
24-010-17-71
24-011-17-71
24-012-17-71
20 mm
-
-
24-010-20-71
24-011-20-71
24-012-20-71
11
14
20
Medium
(32–38 mm)
17
Small
(23–31 mm)
14
Xtra-Small
(17–22 mm)
Foot Plate Depth
V 3.0
Instructions for Use
Sternal Talon® Implants
Double Sternal Talon®
The Double Sternal Talon® implants have four legs and are available in two widths – S and M –
and four foot plate depths: 11 mm, 14 mm, 17 mm and 20 mm.
11 mm
24-019-11-71
24-020-11-71
14 mm
24-019-14-71
24-020-14-71
17 mm
24-019-17-71
24-020-17-71
20 mm
-
24-020-20-71
V 3.0
20
Medium
(32–38 mm)
14
Small
(23–31 mm)
11
Foot Plate Depth
17
Overall Sternal Width
15
Instructions for Use
Sternal Talon® Implants
12
Symbols on Product
Foot Plate Depth
in mm (14)
Sternal Talon®
Width (L)
Three Position
Screw
Setting options of the
3-position screw and
corresponding
functions (quick
reference)
(Cut Points)
Marking
Meaning
Key to setting options of the 3-position screw and corresponding
functions
Safe Zone (safe closing position)
M
Sternal Talon® Width
14
Foot Plate Depth (in mm)
24-021-14-71
Article Number
European Conformity Mark
(at the bottom side of the Sternal Talon)
3-Position Screw
LOT Number (at the bottom side of the Sternal Talon®)
cut points
16
Points at which the implant can be cut open in case of need
V 3.0
Instructions for Use
Sternal Talon® Implants
13
Sternal Talon® Storage
13.1
Sternal Talon® Storage Tray
a. Opening and Closing the Sternal Talon® Storage Tray (55-172-00-04)
Opening:
Press the black pushbutton,
then pull off the lid as
indicated by the arrows.
Closing:
Place the lateral lugs of the lid
into the guide grooves, then
push lid in place as indicated
by the arrows.
V 3.0
17
Instructions for Use
Sternal Talon® Implants
b. Loading the Instrument Section of the Sternal Talon® Storage Tray
(55-172-00-04)
The Sternal Talon® Storage Tray offers dedicated space for storing the following instruments
securely.
Quantity
Article No. (REF)
Designation
2
24-008-01-07
Screwdriver
2
24-003-04-07
Introduction/Removal Clamp
2
24-004-01-07
Tenaculum Clamp
2
24-005-01-07
Reduction Clamp
1
24-006-01-07
Caliper
1
24-006-02-07
Depth Gauge
1
24-007-01-07
Cutter
18
V 3.0
Instructions for Use
Sternal Talon® Implants
NOTICE
Implants that have been removed from their sterile packaging but are still unused and noncontaminated may be processed in the same way as implants delivered non-sterile (see
section 14) using an appropriate cleaning, disinfecting and sterilization procedure. To this
end, the Sternal Talon® Storage Tray offers dedicated storage space for 6 Single Sternal
Talon® implants or 6 Double Sternal Talon® implants.
Please note that the Sternal Talon® Storage Tray (55-172-00-04) comprises only the storage
facility shown above. The instruments and implants additionally depicted are just included
here for illustration. In other words, the Sternal Talon ® instruments required for surgical
interventions must be ordered separately in addition to the storage tray (55-172-00-04).
WARNING
Avoid intraoperative delays!
If parts required for the intervention are missing, this can prolong the operation or make it
even impossible to perform the planned procedure. Therefore, make sure prior to each
intervention that enough Sternal Talon® implants are at hand and all Sternal Talon® and
other instruments required are available complete, fully functional and in sterile condition.
13.2
Sternal Talon® Implant Storage Box
a. Opening and Closing the Sternal Talon® Implant Storage Box (55-171-51-04)
V 3.0
19
Instructions for Use
Sternal Talon® Implants
Opening: To open the box, press the legs of the locking pins together (photo 1) and pull out
the pins (photo 2). Once all pins have been removed (photo 3), the cover can simply be lifted
off.
1
2
3
Closing: To close the box, put the cover in place, then insert the locking pins into the
respective openings (photo 4) and push them back down in place (photos 5 and 6).
4
5
6
b. Charging the Sternal Talon® Implant Storage Box (55-171-51-04)
The Sternal Talon® Implant Storage Box is designed for storing the following sterile-packed
Sternal Talon® implants:
Quantity
Article Number (REF)
Designation
3
24-008-11-71
Single Sternal Talon, 11 mm, XtraSmall, Sterile
3
24-008-14-71
Single Sternal Talon, 14 mm, XtraSmall, Sterile
3
24-009-11-71
Single Sternal Talon, 11 mm, Small, Sterile
3
24-009-14-71
Single Sternal Talon, 14 mm, Small, Sterile
3
24-009-17-71
Single Sternal Talon, 17 mm, Small, Sterile
3
24-010-11-71
Single Sternal Talon, 11 mm, Medium, Sterile
3
24-010-14-71
Single Sternal Talon, 14 mm, Medium, Sterile
3
24-010-17-71
Single Sternal Talon, 17 mm, Medium, Sterile
3
24-010-20-71
Single Sternal Talon, 20 mm, Medium, Sterile
20
V 3.0
Instructions for Use
Sternal Talon® Implants
Quantity
Article Number (REF)
Designation
3
24-011-11-71
Single Sternal Talon, 11 mm, Large, Sterile
3
24-011-14-71
Single Sternal Talon, 14 mm, Large, Sterile
3
24-011-17-71
Single Sternal Talon, 17 mm, Large, Sterile
3
24-011-20-71
Single Sternal Talon, 20 mm, Large, Sterile
3
24-012-14-71
Single Sternal Talon, 14 mm, XtraLarge, Sterile
3
24-012-17-71
Single Sternal Talon, 17 mm, XtraLarge, Sterile
3
24-012-20-71
Single Sternal Talon, 20 mm, XtraLarge, Sterile
1
24-019-11-71
Double Sternal Talon, 11 mm, Small, Sterile
1
24-019-14-71
Double Sternal Talon, 14 mm, Small, Sterile
1
24-019-17-71
Double Sternal Talon, 17 mm, Small, Sterile
1
24-020-11-71
Double Sternal Talon, 11 mm, Medium, Sterile
1
24-020-14-71
Double Sternal Talon, 14 mm, Medium, Sterile
1
24-020-17-71
Double Sternal Talon, 17 mm, Medium, Sterile
1
24-020-20-71
Double Sternal Talon, 20 mm, Medium, Sterile
NOTICE
This implant configuration has proved suitable as a basic set providing all the implants
normally required for Sternal Talon® interventions.
Please note that the Sternal Talon® Implant Storage Box (55-171-51-04) comprises only the
storage facility shown above. The implants additionally depicted are just included here for
illustration. In other words, the Sternal Talon® implants required for surgical interventions are
not included but must be ordered separately in addition to the storage box (55-171-51-04).
WARNING
Avoid intraoperative delays!
If parts required for the intervention are missing, this can prolong the operation or make it
even impossible to perform the planned procedure. Therefore, make sure prior to each
intervention that enough Sternal Talon® implants are at hand and all Sternal Talon® and other
instruments required are available complete, fully functional and in sterile condition.
V 3.0
21
Instructions for Use
Sternal Talon® Implants
14
Sterility, Cleaning and Disinfection
Processing: Cleaning, Disinfecting and Sterilization
Implants that have been removed from their sterile packaging but are still unused and
uncontaminated, as well as implants that are purchased non-sterile, may be reprocessed as
long as suitable cleaning, disinfecting and sterilization procedures are employed. These
implants must always be sent through the entire processing cycle (using suitable cleaning,
disinfecting and sterilization methods) before usage.
Instruments and metallic implants are suitable for machine processing/thermal disinfection.
They can be treated with programs released for (re)processing surgical instruments. The
Sternal Talon® storage trays include components made of hard-anodized and anodized
aluminum which may only be cleaned with neutral-pH or alkaline detergents explicitly
approved for use on anodized aluminum parts. However be sure to observe the instructions
given by the manufacturer of your cleaning/disinfecting machines (washer-disinfectors) and
the treatment agents (cleaner, disinfectant) used. The processing cycle – including proper
loading – must guarantee adequate removal of residues.
To ensure an effective cleaning process, both implant components (male and female
portion) have to be separated before being placed and treated in the washerdisinfector. Upon completion of the cleaning process, take care that only implant
halves with identical lot number (see rear side of the implant halves) are joined
together and that the devices are arranged properly in the corresponding storage
tray.
Sterilization must be carried out in accordance with valid vapor-sterilization procedures,
e.g. in a sterilizer in accordance with EN 285:2008 and validated in accordance with
ISO 17665-1:2006.
Only for sterile-packed implants:
Implants delivered sterile are marked as such:
. They must be stored in their
original packaging in a place suitable for storing sterile supplies and may be removed from
their packaging only immediately prior to use. The expiration date and integrity of the sterile
package must always be checked before use. If the expiration date is exceeded or the
packaging is found to be defective, do not use the implant components until properly
resterilized!
22
V 3.0
Instructions for Use
Sternal Talon® Implants
Storage conditions (only for implants in sterile packaging)
Temperature:
5–25°C (41–77°F)
Relative humidity:
45% – 85%
WARNING
Danger of infection from non-sterile handling!
Improper sterilization and non-sterile handling of the implants and the accessories can pose a
serious health hazard to patients.
The responsibility for proper cleaning, disinfection and sterilization of implant components and
accessories lies with the operator or product user. Be sure to observe all local regulations
(including potential restrictions).
Sternal Talon® implants are intended for single use only and must not be reused under any
circumstances. Never reuse contaminated and/or used implant components!
V 3.0
23
Instructions for Use
Sternal Talon® Implants
15
Intraoperative Placement / Handling
15.1
Closure Variants
The Sternal Talon® is designed in both a single- and double configuration. The single version is
available in five different widths (XS, S, M, L and XL), the double version in two widths (S, M).
Both versions come in four foot plate depths or leg lengths (11, 14, 17 and 20 mm). Proper fit
of the device requires the surgeon to measure both the sternal depth and width and pick the
correct implant based on the patient’s anatomy. Only with proper implant selection can
adequate fixation be achieved. Some patients, based on the sternal width, intercostal distance
or sternal depth will not be candidates for the Sternal Talon ® and alternative closure methods
should be used. Under no circumstances should the surgeon attempt to bend or modify the
Sternal Talon® implants in any way! With the Sternal Talon® system, altogether three Sternal
Talon® implants are usually required for closing the sternum – two Double versions combined
with one Single version (Figure 1). Further stabilization of the bone can be achieved with
sternal wires or sternal plates.
Alternative Closure: Due to the patient’s anatomy and/or surgeon preference, three or more
single devices may be used in lieu of 2 double devices and one single device configuration. Any
of the intercostal spaces one thru five can be used for placement of a single device. Further
stabilization of the sternum can be achieved with sternal wires or sternal plates. A
configuration consisting of three Single Sternal Talon® implants is depicted in Figure 2.
1
24
2
V 3.0
Instructions for Use
Sternal Talon® Implants
15.2
Sternum Measurement for Correct Size Selection
a. Determination of the total width of the sternum
Use the caliper (24-006-01-07) at the application sites to determine the total width of the
sternum. Measure the two halves separately. To determine the total width, continue to add
the width on the left and the width on the right respectively. Enter the established
measurement results into the Sternal Talon® measurement and documentation table
(wall chart, see section 15.2 c.).
Excerpt from the Measurement and
Documentation Table (wall chart)
(refer to section 15.2 c)
b. Determination of the thickness of the sternum
After determining the total width, use the caliper (24-006-01-07) to measure the depth of
the sternum (sternal thickness) at the placement points and enter the results in the
Sternal Talon® Measurement and Documentation Table (wall chart, see section 15.2 c.).
The sternal thickness will determine whether a device with 11-mm, 14-mm, 17-mm or
20-mm foot plates is appropriate, depending on anatomical conditions. This must always
be decided by the surgeon in each individual case. The correct implant size can then be
determined with the help of the following table (see section 15.2 c.).
Excerpt from the Measurement and
Documentation Table
(wall chart) (see section 15.2 c.)
V 3.0
25
Instructions for Use
Sternal Talon® Implants
c.
Sternal Talon® Measurement and Documentation Table
To determine the correct Sternal Talon® size to be used, enter the measuring results from
steps a.) and b.) into the measurement and documentation table (wall chart)
90-728-01-04, then evaluate them using the auxiliary table below.
Note:
1) If two adjacent intercostal spaces have a total width difference greater than 2 mm, a
double Sternal Talon® may not be indicated.
2) If sternum thickness (sternum depth) between left and right halves is not equal, the
surgeon may choose to use larger/deeper device. Care should be exercised to ensure
device is in close proximity to inferior table of the sternum and poses no harm to vital
structures.
NOTICE
The measurement and documentation table (wall chart) 90-728-01-04 is not only intended
for selecting the correct implants, but also for recording important information such as the lot
and article numbers of all the implants used.
26
V 3.0
Instructions for Use
Sternal Talon® Implants
d. Case example for selecting the correct Sternal Talon® size
Helpful brief instructions with a case example on correct size selection are printed on the
rear of the measurement and documentation table (90-728-01-04).
V 3.0
27
Instructions for Use
Sternal Talon® Implants
15.3
a.
Preparing Site for Sternal Talon® Insertion
Retract the skin and soft tissue and expose the rib-sternum connection. Use blunt
dissection or a high-frequency surgical device to create passages in the intercostal space,
so that the foot plates of the Sternal Talon® can be inserted and the implant halves fixed
to the sternum. Direct vessel visualization helps to prevent injury.
WARNING
Danger of death if arterial injury is not avoided!
Caution should be exercised to avoid injuring the internal mammary artery or other
intercostal vessels. Should vessel damage occur or be suspected, the device should be
removed and the vessel repaired.
28
V 3.0
Instructions for Use
Sternal Talon® Implants
b. Widen the created track as appropriate using the Depth Gauge (24-006-02-07). The depth
gauge features an adjustable measuring device that can be set to a foot plate depth of
11 mm, 14 mm, 17 mm or 20 mm. To adjust the device, loosen the setscrew, set the
desired foot plate depth, then retighten the screw.
Selectable Foot
Plate Depths
Setscrew
NOTICE
As various foot plate lengths (11, 14, 17 or 20 mm) can be set on the instrument, the Depth
Gauge can also be used to check the implant for correct foot plate depth once again before
putting it in place on the sternum.
V 3.0
29
Instructions for Use
Sternal Talon® Implants
15.4
a.
Placing the Sternal Talon®
Once the correct implant size has been selected from the Sternal Talon® set, the threeposition screw must be set to the 7 o’clock "Open" position and the two halves of the
implant disengaged.
7 o’clock "Open" position
7 o’clock "Open" position
Subsequently set the setscrew at the female portion to the 3 o’clock "Closing" position.
3 o’clock "Closing" position
Before the two halves are passed on to the surgeon, check that the Lot number is the
same on the male and the female portion and note the number down on the Sternal Talon®
wall chart (90-728-01-04). Check the implants for correct size and identical lot number on
both halves. Never join two implant halves with different lot numbers!
NOTICE
Sterile packaged implants are already packed separately, the setscrew is in the 3 o’clock
"Closing" position. Implants delivered in sterile packaging can be used directly from the
sterile packaging.
30
V 3.0
Instructions for Use
Sternal Talon® Implants
b. Hold the Sternal Talon® at a 45-degree angle to the sternum, then guide the legs into the
tracks prepared in the intercostal space while pressing the plate downwards (see
illustrations 1–3). Note that the female implant half (the half with the screw) must always
be placed on the right side of the sternum (as seen by the patient). If required, turn the
implant halves a little to align the plates parallel to the sternum, making also sure that the
hooks of the legs are correctly placed under the inferior table of the sternum. Proceed to
install all implant halves in this way.
1
2
3
NOTICE
Check that the implant halves with the screw (female portions) are all placed on the right side
of the sternum (as seen by the patient), that all implants have the correct size and that each
Sternal Talon® is placed in the correct intercostal space as intended. Be aware that these
factors are essential for reliable sternum closure.
WARNING
Danger of death if arterial or vascular injury is not avoided!
Caution should be exercised to avoid injuring the internal mammary artery of other
intercostal vessels. Should vessel damage occur or be suspected, the device should be
removed and the vessel repaired.
V 3.0
31
Instructions for Use
Sternal Talon® Implants
c.
Instead of inserting the Sternal Talons with your hands, you can also use the Sternal
Talon® Introduction/Removal Clamp (24-003-04-07) to apply the Sternal Talon® halves to
the sternum.
The introduction/removal clamp can be used for putting Single Sternal Talon® halves (see
Figure 1) as well as Double Sternal Talon® halves (see Figure 2) in place on the sternum.
1: Single Sternal Talon
2: Double Sternal Talon
If it is not possible to place the implant halves correctly on the sternum either manually or with
the Sternal Talon® Introduction/Removal Clamp (24-003-04-07), the tracks created may be
too small (see section 15.3 a.) or the foot plates of the selected implant may be too short.
In such case, it may be necessary to widen the track and check with the depth gauge
(24-006-02-07). The gauge can also be used to check the leg length of the selected device for
correctness once again (see section 15.3 b.). If necessary, use an implant with longer foot
plates.
32
V 3.0
Instructions for Use
Sternal Talon® Implants
WARNING
Danger of death if arterial or vascular injury is not avoided!
Caution should be exercised to avoid injuring the internal mammary artery of other
intercostal vessels. Should vessel damage occur or be suspected, the device should be
removed and the vessel repaired.
WARNING
Danger of death in the event of re-operation
Only use linear fluted drains with the Sternal Talon®. Do not use round-perforated drains in
conjunction with Sternal Talon® devices, as the foot plates could get caught in the drain
holes, requiring rotational manipulation of the drain or even reopening of the sternum in
order to remove the chest tube.
Always ensure drains are not engaged and mobile prior to closure. If the chest tube appears
to be caught on the footplates of the implant during removal, do not force removal. The chest
tube will need to be removed under direct visualization in the operating room.
V 3.0
33
Instructions for Use
Sternal Talon® Implants
15.5
a.
Closing the Sternal Talon®
Make sure that the 3-position screw is turned to the 3 o’clock or "Closing" position. Reduce
the sternum with the Tenaculum Clamp 24-004-01-07 or by wire placement in the
manubrium and xyphoid. Now align the male and female portions of all Sternal Talons®
and slightly engage the two halves in each case, using the Sternal Talon ®
Introduction/Removal Clamp 24-003-04-07 designed for this purpose (see Figure 1).
Once the two sides of the Sternal Talon® have engaged, move the Reduction Clamp
24-005-01-07 to the designated insertion points on the Sternal Talon® (see Figures 2 and
3) and use it to firmly lock the male and female portions together Talon by Talon, thus
closing the sternum completely. During this process, be careful to observe the midline for
protruding internal tissue and proper bony alignment.
1: Introduction/Removal Clamp
2: Reduction Clamp applied to Single Sternal Talons
34
3: Reduction Clamp applied to Double Sternal Talons
V 3.0
Instructions for Use
Sternal Talon® Implants
NOTICE
Ensure parallel alignment before joining the implant halves.
If the sternal halves cannot be properly aligned to each other, the foot plates of the implant
are probably too short. In this case, measure the depth of the sternum (sternal thickness)
again (see section 15.2) and use an implant with longer foot plates as appropriate. The depth
gauge (24-006-02-07) can be used to verify correct foot plate length once again (see
section 15.3 b.).
WARNING
Danger of death if implants are not correctly closed!
Make sure that the 3-position screws of all implants are set to the 3 o’clock position
("Closing") before starting the closing procedure. Failure to set the screws to this position
may prevent closure altogether or lead to implant damage. Be sure to use the Reduction
Clamp (24-005-01-07) for closing the implants. Depending on the stability of the sternum,
the surgeon should not apply excessive force during closure.
Make sure that the two halves of the sternum are apposed well and the closure is stable with
no motion. Use only moderate pressure when closing the implants in order to prevent bone
fracture, maintain good blood flow and avoid healing troubles.
b. All Sternal Talon® devices feature a "SAFE ZONE" mark indicating the safe "Closing"
position. If the "SAFE ZONE" arrow is not in this area after closing the device, or if the two
halves of the sternum are not properly joined together, you selected the wrong size. The
device should be removed in this case and replaced with a correctly sized Sternal Talon®.
V 3.0
35
Instructions for Use
Sternal Talon® Implants
15.6
Locking the Sternal Talon®
Once all Sternal Talon® implants have been closed, the 3-position screw of all implants must
be rotated counterclockwise to the 11 o’clock "Locked" position in each case. This locks the
Sternal Talons reliably.
11 o’clock Locked position
NOTICE
You’ll feel some resistance just before you reach the locking (11 o’clock) position. This is
intended to ensure that the 3-position screw is securely locked in place. To set the 3-position
screw to the 11 o’clock ("Locked") position, you therefore need to turn it beyond the point of
resistance. Only then will the implant be locked in place reliably.
WARNING
Danger of death if implants are not correctly closed!
Prior to soft-tissue or skin closure, verify that all implants are securely locked and that the
two halves of the sternum make a perfect fit with the gap between them securely closed with
no motion. Be aware that this is essential for reliable sternum closure.
36
V 3.0
Instructions for Use
Sternal Talon® Implants
15.7
a.
Removing the Sternal Talon® in an Emergency Immediately
After the Operation
Removal of the Sternal Talon® is accomplished by rotating the screw clockwise to the
7 o’clock "Open" position. This unlocks the device, enabling the two halves of the implant
to be pulled apart and removed.
7 o’clock "Open" position
NOTICE
We recommend using the specialized KLS Martin screwdriver 24-008-01-07. Almost any flat
screwdriver or cross-drive screwdriver can be used to rotate the screw.
V 3.0
37
Instructions for Use
Sternal Talon® Implants
b. A second re-entry method is to insert a flat screwdriver in the lock mechanism and rotate
superiorly to disengage teeth.
NOTICE
We recommend using the specialized KLS Martin screwdriver
24-008-01-07. However, the lock mechanism can be disengaged in the above way with
almost any flat screwdriver or similar suitable instrument.
c.
Alternatively, a suitable wire/plate cutter can be used to unlock the device by cutting it
open at the intended "cut points".
cut point
cut point
NOTICE
We recommend using the KLS Martin Wire Cutters (24-007-01-07).
38
V 3.0
Instructions for Use
Sternal Talon® Implants
16
Hospital and Patient Equipment
a. Sternal Talon® Hospital Kit
In order to ensure a fast re-entry immediately after the operation, each patient has to be
equipped during hospitalization with a separately packed sterile screwdriver 24-008-01-07 and
a laminated wallet card 90-152-52-21 showing removal instructions. Another individually
sterile-packed screwdriver (24-008-01-07) plus a laminated card (90-152-52-21) must be
stored in the OR area and kept constantly available for the OR staff. These items can be
ordered separately or as a set under article number 24-001-01-02 (hospital kit).
b. Sternal Talon® Patient Kit
Prior to discharge, each patient must be handed out a completed Sternal Talon ® implant
passport (90-151-52-30) containing important information on the implants used. Be sure to
instruct the patient that this passport must be carried along at all times. A sticker
(90-150-52-21) with essential Sternal Talon® information is optionally available for inclusion in
the implant passport to be issued by the hospital. Moreover, the patient is provided with an
ID tag (24-009-02-07) featuring a flat edge (like a flat screwdriver) that can be used for
turning the 3-position screw. These items can be ordered separately or as a set under article
number 24-001-00-02 (patient kit).
c.
Sternal Talon® Brief Guide
A laminated Sternal Talon® Brief Guide (90-245-51-10) is available to the practicing doctors
and surgical staff and can be placed in the surgical area as an application aid.
NOTICE
The laminated Sternal Talon® Brief Guide must not be considered a substitute for these
Instructions for Use
V 3.0
39
Instructions for Use
Sternal Talon® Implants
17
Removing the Sternal Talon® in a Re-Operation
Following Bony Consolidation
To expose the implant for access to the three-position screw, it is necessary to dissect the
surrounding soft tissue in a first step. Thereafter, rotate the 3-position screw clockwise to the
7 o’clock or "Open" position to unlock the device.
7 o’clock "Open" position
If the setscrew does not work, you can also open the implants by introducing a suitable flat
screwdriver into the lock mechanism or by separating the cut points with the use of suitable
wire cutters (refer to section 15.7 b. and c.).
After opening the implants and laterally dissecting approx. 4-5 mm (along the Sternal Talon®
foot plates), apply the Sternal Talon® Introduction/Removal clamp (24-003-04-07) at the
female implant half first (implant half with setscrew), as shown in Figure 1. Remove this
implant half with an arching/turning movement as shown in Figure 2-3. The male implant half
(implant half without screw) can then be removed with an arching/turning movement in the
same way as shown in Figure 4.
1
2
3
4
WARNING
Danger of death if arterial or vascular injury is not avoided!
The time required for opening the chest in the event of an emergent or elective reoperation
after bony consolidation may be higher than usual.
Be sure not to damage any vital structures during dissection. Utmost caution should be used
with regard to the internal mammary artery and other intercostal vessels.
40
V 3.0
Instructions for Use
Sternal Talon® Implants
18
Disposal
When disposing of packaging material and potentially infectious material (e.g. after implant
removal), ensure adherence to local regulations and disposal guidelines.
19
Troubleshooting
Problem/Symptom
Cause
Correction
The setscrew may be
incorrectly positioned
(refer to section 15.5).
Turn the setscrew to the 3 o’clock
"Closing" position to allow both
halves to interconnect (refer to
section 15.5).
Possibly one of the
Sternal Talon® halves is
bent.
A bent Sternal Talon® implant must
be removed and may not be reused
(refer to section 9).
The Sternal Talon®
cannot be closed.
Turn the three-position screw
to the 7 o’clock "Open" position (refer
to section 15.7 a.) in order to be able
to disconnect the two Sternal Talon®
halves.
The Sternal Talon®
cannot be opened.
3-position screw possibly
not set to correct position.
The Sternal Talon®
buckles during closure.
The implant halves
may have been incorrectly
aligned (refer to
section 15.5).
Align implant halves parallel to each
other before closure
(refer to section 15.5). A bent
Sternal Talon® implant must be
removed and may not be reused
(refer to section 9).
The implant halves
cannot be aligned
parallel to each other.
Overly short foot plates
may have been selected, so
that the feet cannot grip
under the sternum correctly
(refer to section 15.5).
Measure sternum thickness again
(refer to section 15.5) and select a
longer footplate, if required (refer to
section 15.5).
V 3.0
41
Instructions for Use
Sternal Talon® Implants
20
Accessories
Item
Name
Art. Number
1
Introduction/Removal Clamp
24-003-04-07
2
Tenaculum Clamp
24-004-01-07
3
Reduction Clamp
24-005-01-07
4
Caliper
24-006-01-07
5
Depth Gauge
24-006-02-07
6
Wire / Plate Cutter
24-007-01-07
7
Screwdriver
24-008-01-07
8
Patient ID Tag, English
9
24-009-02-07
®
Storage Tray
55-172-00-04
®
Implant Storage Box
55-171-51-04
Sternal Talon
10
Sternal-Talon
11
Measurement and Documentation Table (Wall Chart), English
12
13
90-728-01-04
®
Card, small, English (sticker)
90-150-51-21
®
Implant Passport, English
90-151-51-30
®
Card, big, English (laminated)
90-152-51-21
Sternal Talon
Sternal Talon
14
Sternal Talon
15
Sternal Talon® Brief Guide
90-245-51-10
®
16
Sternal Talon Patient Kit, English
(consisting position 8, 11, 12 and 13)
24-001-00-01
17
Sternal Talon® Hospital Kit, English
(consisting of 2 items no. 7 and 2 items no. 14)
24-001-01-01
Introduction/Removal Clamp
24-003-04-07
Tenaculum Clamp
24-004-01-07
Reduction Clamp
24-005-01-07
Caliper
24-006-01-07
Depth Gauge
24-006-02-07
Wire / Plate Cutter
24-007-01-07
42
Screwdriver
24-008-01-07
V 3.0
Instructions for Use
Sternal Talon® Implants
21
Contact Details
For US market and Canada:
KLS Martin L.P.
Jacksonville, Fl 32246 · USA
Tel.: 1-800-625-1557 or +1-904-641-7746
E-Mail: [email protected]
For other countries please contact:
Gebrüder Martin GmbH & Co. KG
KLS Martin Platz 1, D-78532 Tuttlingen
Tel.: +49 7461 706-0 / Fax +49 7461 706-193
E-Mail: [email protected]
V 3.0
43
KLS Martin Group
Karl Leibinger GmbH & Co. KG
78570 Mühlheim · Germany
Tel. +49 74 63 838-0
[email protected]
KLS Martin France SARL
68000 Colmar · France
Tel. +33 3 89 21 66 01
[email protected]
KLS Martin L.P.
Jacksonville, Fl 32246 · USA
Office phone +1 904 641 77 46
[email protected]
KLS Martin GmbH + Co. KG
79224 Umkirch · Germany
Tel. +49 76 65 98 02-0
[email protected]
Martin Italia S.r.l.
20059 Vimercate (MB) · Italy
Tel. +39 039 605 67 31
[email protected]
Gebrüder Martin GmbH & Co. KG
Representative Office · Russia
121471 Moscow
Tel. +7 (499) 792-76-19
[email protected]
Stuckenbrock Medizintechnik GmbH
78532 Tuttlingen · Germany
Tel. +49 74 61 16 58 80
[email protected]
Nippon Martin K.K.
Osaka 541-0046 · Japan
Tel. +81 6 62 28 90 75
[email protected]
Gebrüder Martin GmbH & Co. KG
Representative Office · China
201203 Shanghai
Tel. +86 21 2898 6611
[email protected]
Rudolf Buck GmbH
78570 Mühlheim · Germany
Tel. +49 74 63 99 516-30
[email protected]
Martin Nederland/Marned B.V.
1270 AG Huizen · The Netherlands
Tel. +31 35 523 45 38
[email protected]
Gebrüder Martin GmbH & Co. KG
A company of the KLS Martin Group
KLS Martin Platz 1 · 78532 Tuttlingen · Germany
Postfach 60 · 78501 Tuttlingen · Germany
Tel. +49 7461 706-0 · Fax 706-193
[email protected] · www.klsmartin.com
Date of Release: 09.11 ·
90-147-52-30 · Printed in Germany · Copyright by Gebrüder Martin GmbH & Co. KG · Alle Rechte vorbehalten ·
Technische Änderungen vorbehalten · We reserve the right to make alterations · Cambios técnicos reservados · Sous réserve de modifications techniques
Ci riserviamo il diritto di modifiche tecniche · Revision Number: V 3.0