* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 1,0-,1,2 + ½
Bremsstrahlung wikipedia , lookup
Molecular Hamiltonian wikipedia , lookup
Double-slit experiment wikipedia , lookup
Chemical bond wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Ferromagnetism wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Particle in a box wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Tight binding wikipedia , lookup
Auger electron spectroscopy wikipedia , lookup
Matter wave wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Hydrogen atom wikipedia , lookup
Electron scattering wikipedia , lookup
Wave–particle duality wikipedia , lookup
Atomic orbital wikipedia , lookup
Atomic theory wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
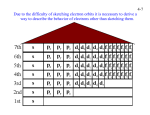
Chapter 5 : Electrons in Atoms Problems with Rutherford’s Model of the Atom Chlorine # 17 Argon # 18 Potassium # 19 Reactive Not reactive Very reactive Does not explain why elements react the way they do! • It’s because they have different arrangements of electrons! Electrons and Light • An element’s chemical behavior is related to its arrangement of electrons • Elements give off different light when burned • We can analyze light to learn about atoms! Atomic Emission Spectra The frequencies of the electromagnetic radiation (EMR) emitted by atoms of an element are unique to each element, like fingerprints on people! The Quest for a Better Model • Electromagnetic radiation (EMR) behaves like a wave. Characteristics of a Wave Wavelength = λ Frequency = f (number of waves that pass a point per second) 1 Hertz (Hz) = 1 wave per second (SI Unit for frequency) Types of Waves Compressional or Longitudinal Wave (sound or slinky) Transverse Wave (light, rope) ://www.youtube.com/watch?v=Rbuhdo0A ZDU Light as a Wave • We can relate the speed, frequency, and wavelength of EMR with the equation: v = fl v is velocity (m/s) f is frequency (Hertz – Hz, 1 Hz = 1 wave/sec) l is wavelength (m) The speed of light (c) is a constant! c = 3.0 x 108 m/s Velocity and Frequency of Light v = λf v (or c for light) = speed of light (3.0 x 108 m/s) ↑ wavelength ↓ frequency ↓ wave length ↑frequency Problems A helium-neon laser emits a light with a wavelength of 633 nm. What is the frequency of this light? Use the equation v = λf First convert nm to m. 633 nm X 1 m 1 x 109 nm = 6.33 x 10-7 m Then do the math. 3.0 x 108 m/s = (6.33 x 10-7 m) (f) (6.33 x 10-7 m) (6.33 x 10-7 m) 4.7 x 1015 1/s or Hz = f An FM radio station broadcasts at a frequency of 98.5 MHz. What is the wavelength of the station’s broadcast signal? Frequency = 98.5 MHz Velocity = 3 x 108 m/s Wavelength = X Use the formula, Rearrange the formula, Convert 98.5 MHz to Hz 98.5 MHz 1 x 106 Hz = 9.85 x 107 Hz 1MHz λ = vf so plugging in our values: λ = 3 x 108 m/s / 9.85 x 107 Hz = 3.05 m v = λf λ = v/f Stop. Light: Particle or Wave? Wave model doesn’t address: Why heated objects emit only certain frequencies of light at a given temperature? Why some metals emit electrons when a colored light of a specific frequency shines on them? (photoelectric effect) http://www.youtube.com/watch?v=WO38qVDGgqw&feature=related Iron Dark gray = room temp Red = hot temp Blue = extremely hot temp Photoelectric Effect – The problem with wave theory Only certain frequencies of light could emit an electron from a plate of Ag. Accumulation of low frequencies couldn’t Einstein and the Dual Nature of EMR (1900) • Electromagnetic Radiation (EMR) acts as a wave of individual particles (photons) Wave vs. Particle Wave Characteristics Particle Characteristics Light can be reflected. Photons have the ability to knock individual electrons off of a conductor-Photoelectric Effect! Light shows interference Metals glow only specific patterns. colors when heated. Wave + Wave RESULT What is the relationship between energy and frequency? Max Planck - 1900 Matter gains or loses energy only in small, specific amounts called quanta. This is why we see specific color lines in emission spectra. A quantum is the minimum amount of energy that can be gained or lost by an atom Equantum = hf E is energy h is Planck’s constant = 6.626 x 10-34 J·s f is frequency J is joule, SI Unit for energy Calculating the energy in a Photon Ephoton = hf E = (6.626 x 10-34 J·s) x (7.23 x 1014 s-1) E = 4.79 x 10-19 J Niels Bohr - 1913 • Worked in Rutherford’s lab • Proposed a quantum model of the atom • Explains why emission spectra were discontinuous • Predicted frequencies of light in Hydrogen’s atomic emission spectra Bohr’s Explanation • Ground state – lowest energy state of an atom’s electrons • Excited state – when an atom’s electrons gain energy • Electrons move in circular orbits – Smaller orbit – lower energy state, “energy level” – Larger orbit – higher energy state, “energy level” An explanation for Emission Spectra http://www.mhhe.com/phys sci/astronomy/applets/Bohr/ applet_files/Bohr.html Excited States Ground state Atoms absorb energy and are excited. As the electron returns to the ground state they give off energy “photon” equal to the difference in energy levels. Bohr’s Model Electrons move around the nucleus in set orbits in specific energy levels. When excited, electrons give off a discrete amount of energy as an emission spectrum. This discrete amount of energy is a quantum. Click the link to make an electron jump! http://www.mhhe.com/physsci/astronomy/applets/Bohr/applet_files/Bohr.html Bohr used work of others… • Balmer—made an equation (math) to connect the lines of the hydrogen spectrum to each other. • Planck—Energy is directly proportional to the frequency of light. Problem: Bohr’s Model Only explains Hydrogen • Louis de Broglie (1924) – proposed that the energy levels are based on the wave like nature of electrons Heisenberg Uncertainty Principle • It is impossible to know the velocity and position of an electron at any given time • Bohr gives a specific place for electrons, while Heisenberg says you can’t know where the electrons are at a particular time. Photon and electron are about the same mass. Erwin Schrodinger - 1926 • Developed the quantum mechanical model of the atom – Assigns electrons to energy levels like Bohr – Does not predict the path of the electron – It predicts the probability of finding an electron • An electron’s “atomic orbital” is the most probable location of the electron at any point in time. Each dot is a picture of an electron during a given amount of time. Where does the electron spend most of the time? Boundary represents the location of an electron 90% of the time. Stop. Each dot is a picture of an electron during a given amount of time. Where does the electron spend most of the time? Boundary represents the location of an electron 90% of the time. Principle Energy Levels • 7 energy levels • Lowest energy is 1 – greatest energy 7 • Each level consists of sublevels The second energy level is larger and the electrons are farther from the nucleus. s Orbitals p and d Degenerate Orbitals Degenerate orbitals have exactly the same amount of energy. f Degenerate Orbitals Quantum Number Symbol Possible Values Definition Principle n 1,2,3… Major energy Level or shell Angular Momentum l 0 to n-1 Sublevel or subshell Magnetic ml -l to +l Orbital orientation Spin ms +½ to -½ Spin direction If Principle (n) = Then Angular (l) = And Magnetic (ml) = 1 0 (s) 0 2 0 (s) 1 (p) 0 -1, 0, +1 3 0 (s) 1 (p) 2 (d) 0 (s) 1 (p) 2 (d) 3 (f) 0 -1, 0, +1 -2,-1,0,+1,+2 0 -1, 0, +1 -2,-1,0,+1,+2 -3,-2,-1,0,1,2,3 4 0 = s, 1 = p, 2 = d, 3 = f To Write Ground State Electron Configurations… 1. Lowest energy is the most stable 2. 3 principals or rules to follow—Pauli Exclusion Principle, Aufbau Principle, and Hund’s Rule Pauli Exclusion Principle For electrons to occupy the same orbital, they must have opposite spin. • That limits 2 electrons per orbital, written as up or down: Aufbau Principle – each electron must occupy the lowest energy state possible. What that means is: 1. Within a principle energy level, the energy sublevels have different energies. 2. The sublevels increase in energy from s,p,d,f 3. Orbitals in a given energy sublevel have equal energy (they are degenerate). 4. Principal energy levels can overlap. Hund’s Rule In a given sublevel, electrons must occupy each degenerate orbital before additional electrons can be added. Pauli Exclusion Principle Aufbau Diagram Tells you the order orbitals are filled with electrons, in order of increasing energy! 4f 3d 4d 5d 2p 3p 4p 5p 6p 1s 2s 3s 4s 5s 6s 7s 5f 6d 7p 8s 14 e- max 10 e- max 6 e- max 2e- max 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, etc. How can we write this easily? Only list the total electrons in each orbital, as a superscript 1. H – 1s1 2. He –1s2 3. Li - 1s2, 2s1 4. Be - 1s2, 2s2 5. B - 1s2, 2s2, 2p1 6. C - 1s2, 2s2, 2p2 7. N - 1s2, 2s2, 2p3 8. O - 1s2, 2s2, 2p4 9. F - 1s2, 2s2, 2p5 10.Ne - 1s2, 2s2, 2p6 Orbital Notation • Very similar to electron configuration, only this one takes spin of the electron into account and may include all of the electrons or just the valence (outer energy level) electrons sublevel Symbol Be Energy Level 1S 2S ↑↓ ↑↓ + and - spin 2Px 2Py 2Pz Representing Electron Configurations Electron Configurations Sub level diagram – indicates the order that orbitals are filled What are the orbital diagrams and electron configuration notation for Al and Cl? Electron Configuration Shorthand • Substitute noble gases from preceding energy levels in the notation Li – [He] 2s1 C – [He] 2s2 2p2 Stop. Valence Electrons • Electrons in the outer most energy levels S: [Ne] 3s2 3p4 Sulfur has 6 valence electrons (add 2 from s and 4 from p) How many valence electrons do Ne, Al, and Cl have? Valence Electrons Ne: 1s2, 2s2, 2p6 8 e- on 2nd energy level Al: 1s2, 2s2, 2p6, 3s2, 3p1 3 e- on 3rd energy level Cl: 1s2, 2s2, 2p6, 3s2, 3p5 7 e- on 3rd energy level Lewis Dot Models • Only valence electrons – outermost electrons (highest energy level) • Octet rule – all atoms want to have 8 electrons (H and He want 2) in outer orbit 1 2 5 8 X 7 4 Symbol 3 6 Writing Electron Dot Structures • Fill the valence electrons 1 at a time in any particular order. * Ca * * C * * * *O** * * * What are the electron dot diagrams for K, Ar and F? Electron Dot Structures Valence electrons are used in reactions and are represented by an electron dot structure. O H Ne N Periodic Table Blocks • Where an element is on the periodic table can give you clues about its electron configuration. • For the representative elements (A groups), the A-group number tells you how many valence electrons! – Alkali earth metals (2A*) have 2 valence e– Halogens (7A*) have 7 valence e*Note: A different method of naming the groups numbers the columns 1-13 starting on the left side of the table and includes the transition metals. In this system group 2A = group 2, group 3A = group 13 END OF CHEM 1 UNIT 5!!! (the rest is for Pre-AP) Modern Atomic Theory • Any electron in an atom can be described by 4 quantum numbers • Principal Quantum Number • Azimuthal (Angular Momentum) Quantum Number • Magnetic Quantum Number • Spin Quantum Number Principal Quantum Number (n) Related to the size and energy of principal energy level. The farther away from the nucleus the more energy the electron has 1 < 2 < 3 < 4 < 5 < 6 etc…. Azimuthal Quantum Number (Angular Momentum) = l • Refers to the subshells in each principal energy level (n) n 1 2 3 • • • • S=0 P=1 D=2 F=3 4 l 0 0 1 0 1 2 0 1 2 3 Magnetic Quantum Number (ml) • Specifies the orbital within a energy level where an electron is likely to be found n l Orbital designation ml 1 0 1s 0 2 0 2s 0 1 2p -1,0,+1 0 3s 0 1 3p -1,0,+1 2 3d -2,-1,0-,1,2 0 4s 0 1 4p -1,0,+1 2 4d -2,-1,0-,1,2 3 4f -3,-2,-1,0,1,2,3 3 4 Spin Quantum Number (ms) • + ½ or – ½ • Electrons in the same orbitals must have opposite spins (Pauli Exclusion Principle) n l Orbital designation ml ms 1 0 1s 0 + ½, - ½ 2 0 2s 0 + ½, - ½ 1 2p -1,0,+1 + ½, - ½ 0 3s 0 + ½, - ½ 1 3p -1,0,+1 + ½, - ½ 2 3d -2,-1,0-,1,2 + ½, - ½ 0 4s 0 + ½, - ½ 1 4p -1,0,+1 + ½, - ½ 2 4d -2,-1,0-,1,2 + ½, - ½ 3 4f -3,-2,-1,0,1,2,3 + ½, - ½ 3 4 A n l ml ms 2 1 -1 + ½ or What are the quantum numbers for A? B? 2,1,0,-½ 3,0,0,+½ B So to summarize There are four quantum numbers that describe every electron – Principal Quantum # - Energy Level (1-7) – Angular Momentum or Azimuthal Quantum # Type of Orbital (s,p,d,f) – Magnetic Quantum # - Orientation of the orbital (x, y, z, xy, xz, yz, etc.) – Spin Quantum # - Spin (+ or -)