* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download annexes
Survey
Document related concepts
Transcript
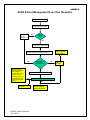
PPHSN Guidelines For The Preparedness, Surveillance And Response To Severe Acute Respiratory Syndrome (SARS) in Pacific Island Countries And Territories March 28th 2003 SARS is a new disease syndrome. Our knowledge about the best way to prevent and treat it is constantly evolving. These guidelines will be continuously updated. Please regularly check PPHSN website for the most up to date guidance PPHSN. SARS Guidelines 28/03/2003 1 Table of Contents BASIC DISEASE FACTS ......................................................................................................... 4 Background ............................................................................................................................ 4 Description of disease ............................................................................................................ 4 Epidemiology ......................................................................................................................... 4 Agent and infectious dose ...................................................................................................... 4 SURVEILLANCE ..................................................................................................................... 5 WHO Case Definitions for hospital based surveillance ........................................................ 5 Surveillance and reporting ..................................................................................................... 6 Minimum dataset ................................................................................................................... 6 PREPAREDNESS – INITIAL ACTION AND RESPONSIBILITIES ..................................... 6 Outbreak response team (EpiNet or CDC other committee) ................................................. 6 Staff responsibilities for the various actions .......................................................................... 6 Clinical assessment of suspected patients .............................................................................. 6 Enhanced surveillance ........................................................................................................... 7 Communications (between members of team and with outside bodies, media etc.) ............. 7 Laboratory diagnosis .............................................................................................................. 7 Initial community interventions ............................................................................................. 7 External (international) reporting, requests for support, and coordination among agencies . 8 CASE MANAGEMENT – the clinical response ....................................................................... 8 Investigations ......................................................................................................................... 8 Management of suspect cases ................................................................................................ 8 Management of probable cases .............................................................................................. 9 Specific Treatment ................................................................................................................. 9 Hospital discharge and follow-up .......................................................................................... 9 HOSPITAL INFECTION CONTROL .................................................................................... 10 Care for patients with probable SARS ................................................................................. 10 MANAGEMENT OF CONTACTS OF SUSPECTED AND PROBABLE CASES .............. 11 General ................................................................................................................................. 11 Contacts of suspected cases on aircraft ................................................................................ 12 REFERENCES AND FURTHER SOURCES OF INFORMATION ..................................... 12 ANNEXES ............................................................................................................................... 13 PPHSN. SARS Guidelines 28/03/2003 2 HISTORY OF GUIDELINE.................................................................................................... 13 ANNEX 1................................................................................................................................. 14 ANNEX 2................................................................................................................................. 15 ANNEX 3................................................................................................................................. 21 ANNEX 4................................................................................................................................. 22 ANNEX 5................................................................................................................................. 23 ANNEX 6................................................................................................................................. 25 ANNEX 7................................................................................................................................. 27 ANNEX 8................................................................................................................................. 28 ANNEX 9................................................................................................................................. 33 ANNEX 10............................................................................................................................... 36 PPHSN. SARS Guidelines 28/03/2003 3 Severe Acute respiratory Syndrome (SARS) BASIC DISEASE FACTS Background As of 27 March 2003, reports of over 1408 cases, including 53 deaths, of Severe Acute Respiratory Syndrome (SARS), an atypical pneumonia of unknown aetiology, have been received by the World Health Organization (WHO) since 16 November 2002. WHO is coordinating the international investigation of this outbreak and is working closely with health authorities in the affected countries to provide epidemiological, clinical and logistical support as required. SARS was first recognised on the 26 February 2003 in Hanoi, Viet Nam, but it the epidemic started in Guangdong in November 2002. Local transmission occurred in China, Vietnam, Singapore and Canada. The worst-affected areas are Guangdong province and the Special Administrative Region of Hong Kong in China, Hanoi in Vietnam, and Singapore. The causative agent has yet to be identified, although the search has been currently narrowed to members of the paramyxovirus and coronavirus families. The main symptoms and signs include high fever (>38 degrees Celsius), cough, shortness of breath or breathing difficulties. Approximately 10 percent of patients with SARS develop severe pneumonia; some of whom have needed ventilator support. As of 27 March the majority of cases have occurred in people who have had very close contact with other cases; for this reason, health care workers are at particular risk. Description of disease The syndrome begins with fever for 1-2 days, then a dry cough or dyspnea for 2-3 days. Atypical pneumonia develops on day 4-5 in the majority of cases. It is initially unilateral but after a further 1-3 days it often becomes bilateral, progressing to extensive "white-out" on chest XRay. The disease then takes 1 of 2 courses: A) the patient improves (80-90% of cases) and recovers over the next 4-7 days; or B) the patient deteriorates severely on day 6-7 with respiratory distress (10-20% of cases). 50% of patients in category B require mechanical ventilation. The mortality rate in this sub-group is high. During the early phase of the outbreak, around 50% of type B cases have died, giving an overall CFR of 5-10%. Risk factors for poor outcome are not clear, apart from the severity of illness and the need for mechanical ventilation. So far SARS has affected predominantly adults aged 20-70 yrs. Very few cases have occurred in children. The modes of transmission and the causative agent have yet to be determined. Aerosol and/or droplet spread is likely as is transmission from body fluids. Respiratory isolation, strict respiratory and mucosal barrier nursing are recommended for cases. Cases should be treated as clinically indicated. (see below for further details). Epidemiology Agent and infectious dose Some laboratories have reported finding paramyxovirus or coronavirus particles on electron microscopy of specimens taken from cases. However these findings await confirmation. The infectious dose is unknown. Source From the knowledge available to date the source of an infection is another person who is ill with SARS. Occurrence So far all cases reported from outside the affected areas have a history of travel in the previous 10 days through an affected area OR close contact with a case of SARS. PPHSN. SARS Guidelines 28/03/2003 4 Mode of transmission It is likely that the agent is spread from person to person by droplet/aerosol spread. However it may also be air-borne and transmission from contact with body fluids has not been excluded. Period of communicability Not known but particularly infectious once respiratory symptoms appear. A lower risk of transmission is likely to be present during the prodromal phase. Incubation period The incubation period is thought to be 2-7 days exceptionally 10 days, most commonly 3-5 days Vulnerable population sub-groups Insufficient information available at this stage. But probably worse outcomes can be expected in individuals with underlying respiratory and cardiac illnesses such as asthma, COPD and heart disease. Risk in the Pacific The main risk in the Pacific is the importation of cases from affected areas with subsequent local transmission to close contacts including health workers. SURVEILLANCE Please note that a SINGLE case of suspected/probable SARS is an outbreak. WHO Case Definitions for hospital based surveillance Suspected case Clinicians should be alert for persons with onset of illness after February 1, 2003 with: Fever (>38° C) AND One or more signs or symptoms of respiratory illness, including: cough, shortness of breath, difficulty breathing, AND A history of either of the following: close contact*, within 10 days of onset of symptoms, with a person who has been diagnosed with SARS. history of travel, within 10 days of onset of symptoms, to an area** (see table below) in which there are reported foci of transmission of SARS. * close contact means having cared for, having lived with, or having had direct contact with respiratory secretions and body fluids of a person with SARS. Affected Areas** - Severe Acute Respiratory Syndrome (SARS) Country Canada Singapore China Viet Nam Area Toronto Singapore Beijing, Guangdong Province, Hong Kong Special Administrative Region of China, Shanxi, Taiwan Hanoi Last revised 27 March 2003 **An "Affected Area" is defined as a region at the first administrative level where the country is reporting local transmission of SARS. PPHSN. SARS Guidelines 28/03/2003 5 Note In addition to fever and respiratory symptoms, SARS may be associated with other symptoms including: headache, muscular stiffness, loss of appetite, malaise, confusion, rash, and diarrhea. Probable case A suspected case with chest X-ray findings of pneumonia or adult respiratory distress syndrome. OR A person with an unexplained respiratory illness resulting in death, with an autopsy examination demonstrating the pathology of Respiratory Distress Syndrome without an identifiable cause. Surveillance and reporting If travel questionnaires are issued to arriving passengers or passengers from affected areas are requested to identify themselves, record number of arrivals with a travel history that puts them in the at risk group (travel to an affected area within the previous 10 days). Report all suspected/probable cases immediately to National Public Health Authorities, using the PPHSN reporting form (see ANNEX 2). Report all suspected/probable cases immediately to PPHSN Coordinating Body (CB) Focal point or WHO Suva using the PPHSN reporting form (a copy of the completed form used to report to the National Public Health Authorities) (see contacts list in ANNEX 1) Report to PacNet or PacNet-restricted. Minimum dataset [Optional: upon arrival, affected area visited in the last 10 days and presence of symptoms] Please see PPHSN reporting form for data items. For PacNet or PacNet-restricted, same as on reporting form, EXCEPT reporter and patient details (you can send the form on PacNet or PacNet-restricted, but delete the 2nd page). PREPAREDNESS – INITIAL ACTION AND RESPONSIBILITIES This will depend on local arrangements within each country. Outbreak response team (EpiNet or CDC other committee) For the purpose of proper SARS control in hospital environment, this team should include a member experienced in hospital infection control, and who can advise on isolation and barrier nursing issues. Priority functions of the team are to: identify facilities where suspected and probable cases of SARS can be nursed. perform an inventory of supplies required for nursing such patients (using WPRO SARS Preparedness Kit contents list). plan how contacts of suspect/probable cases will be managed liaise with customs/immigration authorities on the best way to provide information to arriving passengers, record travel details for surveillance and plan of action if an ill individual arrives ill on a plane with suspected SARS. Staff responsibilities for the various actions Individual countries to decide Clinical assessment of suspected patients Clinicians must be aware of the symptoms and signs of SARS. Patients with symptoms of SARS should be triaged immediately to designated examination rooms or wards to minimize exposure to other patients and staff. Patients with suspected SARS should be issued with surgical masks. PPHSN. SARS Guidelines 28/03/2003 6 Medical and nursing staff must take precautions when examining the patient ie barrier nursing. Obtain and record detailed clinical, travel and contact history including occurrence of acute respiratory diseases in contact persons during the last 10 days. Obtain chest X-ray (CXR) and full blood count (FBC). (See example patient management flow chart in ANNEX 4) Enhanced surveillance Complete PPHSN reporting form and send immediately to National Health Authorities, with a cc to PPHSN-CB Focal Point. Also send immediately the form WITHOUT reporter and patient details (i.e. page 2) to PacNet or PacNet-restricted Identify close contacts and give information to contacts. Screen any contacts with compatible symptoms as for suspected cases. Communications (between members of team and with outside bodies, media etc.) Ensure that lines of communication are clear. Identify spokesperson for the team who will be the focal point for media briefings and will liaise with international agencies eg WHO/SPC (this could be the EpiNet team Focal Point or another person). Laboratory diagnosis The agent causing SARS remains to be established. There are no specific diagnostic tests. For suspected cases where the diagnosis of SARS is by exclusion and the patient is not very ill (ie no chest X-ray changes compatible with SARS). It is reasonable to take specimens for diagnostic purposes. However health care workers must take full barrier nursing precautions to protect themselves from aerosols or splashing/splattering of blood or other body fluids. For probable cases where the diagnosis of SARS is very likely and particularly if the patient has significant respiratory symptoms. The clinicians must perform a risk/ benefit analysis. There have been documented cases of transmission to HCWs during diagnostic/therapeutic procedures, particularly those prone to the generation of aerosols. Therefore the priority should be for tests likely to influence the clinical management of the patient. If specimens are collected for diagnostic testing (rather than clinical management), they should be stored under appropriate conditions. At this stage, the two laboratories in our region that have agreed to receive specimens are: o Institute Pasteur, Noumea o WHO Collaborating Centre for Reference and Research on Influenza, Australia (See Contact List in ANNEX 1 for addresses) Initial community interventions Provide suitable information to arriving passengers (particularly those who have traveled through affected countries) about the risks of SARS and where they can go to for advice and assistance (as example, see Advice to Arriving Travelers in ANNEX 5). Simple health education messages should be communicated to the public via appropriate media (see Health Advices from Hong Kong in ANNEXES 6 and 7 for examples). WHO has not recommended restricting travel to any destination in the world. However, all travellers should be aware of the main symptoms and signs of SARS, as given above. On the other hand, the CDC, the French Department of Health, Health Canada, New Zealand Ministry of Health and Singapore advise persons planning elective or nonessential travel to the worst-affected areas to postpone their trips until further notice. This careful attitude helps to avoid SARS long-distance spread through travel to and from infected zones and prevents the importation of SARS "home" (lots of close contacts...). This is particularly important in places where control measures may not be easy to implement (and SARS importation may have serious public health consequences). PPHSN. SARS Guidelines 28/03/2003 7 External (international) reporting, requests for support, and coordination among agencies Report all suspect and probable cases to PPHSN/WHO using the PPHSN reporting form Contact PPHSN-CB Focal Point or WHO South Pacific if additional information or assistance is required (see contact list in ANNEX 1). CASE MANAGEMENT – the clinical response Investigations CXR Chest radiographs might be normal during the febrile prodrome and throughout the course of illness. However, in a substantial proportion of patients, the respiratory phase is characterized by early focal infiltrates progressing to more generalized, patchy, interstitial infiltrates. Some chest radiographs from patients in the late stages of SARS have also shown areas of consolidation. In typical severe cases, chest x-ray findings begin with a small unilateral patchy shadow, and progress over 24 - 48 hours to become bilateral, generalized, interstitial/confluent infiltrates. Patchy chest x-ray changes are sometimes noted in the absence of chest symptoms. Acute respiratory distress syndrome might be observed in the end stage. Post-mortem lung tissue shows generalized alveolar damage and lymphocytosis without obvious viral inclusion bodies. FBC Initially the blood picture is often normal. However, by day 3 - 4 of the illness, lymphopenia is commonly observed (>50%), and less commonly, there might be thrombocytopenia. If SARS is complicated by secondary bacterial infection, neutrophilai may occur. Other Elevated hepatic transaminases and creatine phosphokinase levels are seen early in the respiratory phase of the disease. Management of suspect cases In-flight care of suspected case of SARS If a passenger on a flight from an affected area becomes noticeably ill with a fever and respiratory symptoms, the following action is recommended for cabin crew: o The passenger should be, as far as possible, isolated from other passengers and crew o The passenger should be asked to wear a protective mask and those caring for the ill passenger should follow the infection control measures recommended for cases of SARS o A toilet should be identified and made available for the exclusive use of the ill passenger o The captain should radio ahead to alert the airport of destination so that quarantine or health authorities are altered to the arrival of a suspect case of SARS o On arrival, the ill passenger should be placed in isolation and assessed by port health authorities General care of suspected case of SARS Patients with symptoms of SARS should be triaged immediately to designated examination rooms or wards to minimize exposure to other patients and staff. Patients with suspected SARS should be issued with surgical masks obtain and record detailed clinical, travel and contact history including occurrence of acute respiratory diseases in contact persons during the last 10 days obtain chest X-ray (CXR) and full blood count (FBC) if CXR is normal: provide advice on personal hygiene, avoidance of crowded areas and public transportation, remain at home until well with daily clinical follow-up [Singapore teaches patients under "domestic quarantine" to take and record their own temperatures 4-hourly, which the health worker reviews daily]. PPHSN. SARS Guidelines 28/03/2003 8 discharge with advice to seek medical care if respiratory symptoms worsen if CXR demonstrates uni- or bi-lateral infiltrates with or without interstitial infiltration SEE MANAGEMENT OF PROBABLE CASES Management of probable cases hospitalize under isolation or cohorted with other SARS cases (see section on Hospital infection control) Cases need to be in the best isolation facility that can be arranged (this will vary for PICs) and must be nursed using strict barrier techniques including gown or preferably overalls, gloves, boots or over-shoes, HEPA or N95-100 mask (or at least a surgical mask if nothing else available) and goggles - not pleasant to use in PIC climate! samples for laboratory investigation (if possible) and exclusion of known causes of atypical pneumonia: o throat and/or nasopharyngeal swabs 1 o blood for culture and serology (acute specimen and convalescent specimen taken after 3 weeks) o urine o bronchoalveolar lavage o post mortem examination as appropriate Samples should be investigated in laboratories with proper containment facilities (BL3). CXR as clinically indicated treat as clinically indicated Specific Treatment Treatment regimens have included several antibiotics to presumptively treat known bacterial agents of atypical pneumonia. In several locations, therapy has included antiviral agents such as oseltamivir or ribavirin; the effectiveness of these treatments is uncertain.. Steroids have also been administered orally or intravenously to patients in combination with ribavirin and other antimicrobials. Intravenous steroids may be associated with improved outcomes in severe cases. At present, the most efficacious treatment regime, if any, is unknown. Empirical antibiotic therapy should cover causes of community acquired pneumonia including both typical and atypical respiratory pathogens. Hospital discharge and follow-up The period of communicability of the agent that causes SARS is unknown at this time. WHO advises that patients are fit for discharge if: Clinical symptoms/findings: Afebrile for 48 hours No cough Laboratory tests: if previously abnormal White cell (lymphocyte) count returning to normal Platelet count returning to normal Creatinine phosphokinase returning to normal Liver function tests returning to normal Radiological findings: 1 Improving chest x-ray changes best specimen for (known) respiratory viruses is nasal swab PPHSN. SARS Guidelines 28/03/2003 9 Follow-up for convalescent cases Discharged convalescent patients should be asked to return to hospital If they have an elevated temperature of 38 degrees and above on two consecutive occasions they should report to the health care facility from which they were discharged. Follow up is recommended at one week (or before if decided so by the clinician) at which time they should have a repeat chest x-ray, full blood count and any other blood tests that were previously abnormal. The patient should be followed up by the health care facility from which they were discharged. If possible they should not return to their home island. Subsequent follow-ups are recommended until the chest x-ray and patient’s health returns to normal. As part of the follow-up convalescent serology should be taken is taken at 3 weeks (if an acute serum specimen was taken) after the date of the presenting symptoms and provided to the health care facility from which they were discharged. Until more is known about the aetiological agent, and the potential for continued carriage (and hence the risk of continuing transmission) a cautious approach is warranted. WHO advises that following discharge from hospital convalescent cases should be advised to wait for a minimum of 14 days, before considering returning to work/school/college. This is twice the known maximum incubation period. During this period they should stay indoors, keeping contact with others to a minimum. Clear instructions should be given to convalescent cases to return to the health care facility from which they were discharged [see above] if their condition deteriorates and any further symptoms develop. HOSPITAL INFECTION CONTROL Please see NEW ZEALAND MOH Infection Control advice for the management of patients with suspected Severe Acute Respiratory Syndrome (SARS) in ANNEX 8 for an example of measures that can be adapted depending on the resources available. Alternatively see WHO publication Infection Control for VHF in the African Health Care Setting for simple guidance. This is available on the PPHSN website (http://www.spc.int/phs/PPHSN/Outbreak/SARS_Outbreak.htm). Care for patients with probable SARS WHO advises strict adherence with the barrier nursing of patients with SARS using precautions for airborne, droplet and contact transmission. Triage nurses should rapidly divert persons presenting to their health care facility with flu-like symptoms to a separate assessment area to minimise transmission to others in the waiting areas. Suspect cases should wear surgical masks until SARS is excluded. Probable and suspected cases MUST be nursed SEPARATELY, and suspected cases must NEVER be placed with other patients for observation. Patients with probable SARS should be isolated and accommodated as follows in descending order of preference: o Negative pressure rooms with the door closed o Single rooms with their own bathroom facilities o Cohort placement in an area with an independent air supply and exhaust system. Note Turning off air conditioning and opening windows for good ventilation is recommended if an independent air supply is not possible. Disposable equipment should be used wherever possible in the treatment and care of patients with SARS. If devices are to be reused, they should be sterilised in accordance with manufacturers’ instructions. Surfaces should be cleaned with broad spectrum (bactericidal, fungicidal, and virucidal) disinfectants of proven efficacy. PPHSN. SARS Guidelines 28/03/2003 10 Patient movement should be avoided as much as possible. Patients being moved should wear a surgical mask to minimise dispersal of droplets. NIOSH standard masks (N95), often used to protect against other highly transmissible respiratory infections such as tuberculosis, are preferred if tolerated by the patient. All visitors, staff, students and volunteers should wear a N95 mask on entering the room of a patient with confirmed or suspected SARS. Surgical masks are a less effective alternative to N95 masks. Handwashing is the most important hygiene measure in preventing the spread of infection. Gloves are not a substitute for handwashing. Hands should be washed before and after significant contact with any patient, after activities likely to cause contamination and after removing gloves. Alcohol-based skin disinfectants formulated for use without water may be used in certain limited circumstances. Health care workers (HCWs) are advised to wear gloves for all patient handling. Gloves should be changed between patients and after any contact with items likely to be contaminated with respiratory secretions (masks, oxygen tubing, nasal prongs, tissues). Particulate filter personal respiratory protection devices capable of filtering 0.3um particles (N95) or other relevant mask should be worn at all times when attending patients with suspected or probable SARS. Gowns (waterproof aprons) and head covers should be worn during procedures and patient activities that are likely to generate splashes or sprays of respiratory secretions. HCWs must wear protective eyewear or face-shields during procedures where there is potential for generation of aerosols or splashing, splattering or spraying of blood or other body substances. Standard precautions should be applied when handling any clinical wastes. All waste should be handled with care to avoid injuries from concealed sharps (which may not have been placed in sharps containers). Gloves and protective clothing should be worn when handling clinical waste bags and containers. Where possible, manual handling of waste should be avoided. Clinical waste must be placed in appropriate leak-resistant biohazard bags or containers labelled and disposed of safely. MANAGEMENT OF CONTACTS OF SUSPECTED AND PROBABLE CASES General SARS has only been reported to have been transmitted from cases to close contacts. These include people living with or caring for cases at home or in hospital, who have prolonged exposure to droplet spread and/or body fluids including respiratory secretions. The incubation period is reported to be short, typically 2 to 7 days. Close contacts should be informed that they may be at risk of contracting the infection. They should be advised to seek medical care urgently if they develop fever and respiratory symptoms within ten days of their contact with a case. No restriction of activities is recommended for close contacts who have no symptoms. (NB in Hong Kong and Singapore the authorities are recommending that the children of probable cases be kept away from school for 10 days from the date the parent was hospitalised as a precaution Provide reassurance. Record name and contact details. Provide advice in the event of fever or respiratory symptoms to: o immediately report to doctor/physician/health authority o not report to work until advised by health authority o avoid public places until advised by health authority o minimize contact with family members and friends PPHSN. SARS Guidelines 28/03/2003 11 Contacts of suspected cases on aircraft If a passenger or member of aircrew has suspected/probable SARS on arrival and the immediate medical assessment of the ill passenger excludes SARS as a possible cause of his/her illness, the passenger should be referred to local health care facilities for any necessary follow up. If however, the initial medical assessment conducted in the airport concludes that the passenger is a suspect or probable case of SARS the following action should be taken: Contacts All contacts of the ill passenger should be identified during the flight. For the purposes of air travel a contact is defined as: o passengers sitting in the same seat row or 2 rows in front or behind the sick passenger o all flight attendants on the flight o anyone having intimate contact, providing care or otherwise having contact with respiratory secretions of the sick passenger o any one on the flight living in the same household as the ill passenger o If it is a flight attendant who is considered to be a suspect or probable SARS case all the passengers are considered to be contacts. Contacts should provide, to the health authorities, identification and details of their place(s) of residence for the next 14 days. Contacts should be given information about SARS and advised to seek immediate medical attention if they develop any symptoms of SARS within 10 days of the flight. When seeking medical attention they should ensure that all those treating them are aware that they have been in contact with a suspect case of SARS. Contacts should be allowed to continue to travel so long as they are well. If over time it becomes apparent that the suspect case is a probable case of SARS, the health authority where the case is being cared for should inform other health authorities in those areas in which the contacts reside that active surveillance of each contact should be undertaken until 10 days after the flight. Active surveillance may consist of: o If possible daily temperature check and interview by health care worker o At the minimum the contacts should be contacted by a health worker and the advice about seeking medical attention if any of the symptoms of SARS occur reinforced. In some circumstances it may be recommended that contacts remain at home and minimise contact with others including their own families until 10 days after the flight. REFERENCES AND FURTHER SOURCES OF INFORMATION http://www.spc.int/phs/PPHSN/Outbreak/SARS_Outbreak.htm http://www.info.gov.hk/dh/ap.htm http://www.phls.co.uk/topics_az/SARS/menu.htm http://www.cdc.gov/ncidod/sars/ http://www.who.int/csr/sars/en/ http://www.moh.govt.nz/sars PPHSN. SARS Guidelines 28/03/2003 12 ANNEXES ANNEX 1 – List of Key Contacts ANNEX 2 – PPHSN Reporting Form ANNEX 3 – Affected Areas – SARS ANNEX 4 – SARS Patient Management Flow Chart (example) ANNEX 5 – Advice to arriving travellers (2 examples, incl. The Solomon Islands) ANNEX 6 – Health Advice on the Prevention of Respiratory Tract Infections (example from Hong Kong MOH) ANNEX 7 – Health Advice on the Prevention of Respiratory Tract Infections in Public Places (example from Hong Kong MOH) ANNEX 8 – New Zealand infection control advice for managing patients with SARS ANNEX 9 – Assessment of Risk and Capacities to Respond to a Multi-country outbreak of Severe Acute Respiratory Syndrome ANNEX 10 – WPRO SARS Preparedness Kit contents list. HISTORY OF GUIDELINE Compiled by: Dr Kevin Carroll on behalf of PPHSN Reviewed by: Dr Tom Kiedrzynski Epidemiologist (Ag) Secretariat of the Pacific Community, PPHSN-CB Focal Point The PPHSN SARS Task Force consists of: Dr Alain Berlioz, Pharmacist-biologist, New Caledonia Pasteur Institute Dr Mary Beers Deeble, Senior Lecturer, Director, MAE, NCEPH, ANU Dr Kevin Carroll, MO/Epidemiologist, WHO South Pacific Dr Rob Condon, Public Health Physician/Epidemiologist, SARS Outbreak Response Group, WPRO Dr Tom Kiedrzynski, Notifiable Disease Specialist/Epidemiologist (Ag), SPC Dr Kamal Kishore, Senior Lecturer, Medical Microbiology, FSMed Dr Joe Koroivueta, Consultant Virologist, Fiji MoH Dr Ilisapeci Kubuabola, MAE student, Fiji MOH Dr Michael O'Leary, Regional Medical Epidemiologist, CDC/PIHOA Dr Hitoshi Oshitani Regional Adviser in Communicable Disease Surveillance and Response / Team Leader of SARS Preparedness Team, WPRO Dr Salanieta Saketa, National Epidemiologist/MO, Fiji MoH Revision date(s) 25.03.2003 Review date 28.03.2003 PPHSN. SARS Guidelines 28/03/2003 13 ANNEX 1 CONTACTS PPHSN-CB Focal Point Dr Tom Kiedrzynski Epidemiologist (Ag) Secretariat of the Pacific Community, PPHSN-CB Focal Point SPC PO Box D5 98848 Noumea cedex New Caledonia Tel: +(687) 26 20 00 or 01 43 Fax: +(687) 26 38 18 Out of hours +(678) 25 92 36 E-mail: [email protected] WHO South Pacific Dr Kevin Carroll MO/Epidemiologist WHO South Pacific PO Box 113 Suva, Fiji Tel +(679) 3304 600 Fax +(679) 3300 462 Out of hours +(679) 99202971 Email [email protected] Emergency Hotline to WPRO WHO WPRO Outbreak Ops Room After Hours Duty Officer Dr. Hitoshi Oshitani Dr. Robert Condon +63-2-528 9833; +63-2-528 9781 +63-2-528 9782; +63-2-528 9650 +63-920 640 9696 +63-918 921 4217 +63-919 3922023 Institute Pasteur Alain Berlioz-Arthaud, Laboratoire de Biologie Medicale, Institut PASTEUR de NouvelleCaledonie, BP 61, 98845 Noumea, New Caledonia. Tel : +(687) 27.02.85 Fax : +(687)27.33.90 Email : [email protected] WHO Collaborating Centre for Reference and Research on Influenza, Australia Ian Barr WHO Collaborating Centre for Reference and Research on Influenza 45 Poplar Road, Parkville. Victoria, Australia 3052 Tel +(61) 3 9389 1761 Fax + (61) 3 9389 1881 Email [email protected] PPHSN. SARS Guidelines 28/03/2003 14 ANNEX 2 PPHSN Reporting Form Severe acute respiratory syndrome (SARS) outbreak PPHSN. SARS Guidelines 28/03/2003 15 PPHSN Reporting Form Severe acute respiratory syndrome (SARS) outbreak Report of suspected or probable cases This form is to be used for reporting suspected or probable cases of respiratory illness, which may be associated with the outbreaks of respiratory illness in Hong Kong SAR, Guangdong province (China) and Hanoi (Vietnam). The cases to be reported are those conforming to the case definitions provided by the WHO and reproduced at the bottom of this form. Please return this form to PPHSN-CB Focal Point at SPC Fax: +(687) 26 38 18 e-mail: [email protected] AND/OR WHO South Pacific Fax: +(679) 330 04 62 e-mail: [email protected] WHO case definitions for hospital based surveillance Suspected case A person presenting to a health care facility after 1st February 2003 with a history of: High fever (>38 o C) And: One or more respiratory symptoms (cough, shortness of breath, difficulty breathing) And one or more of the following: Close contact*, within 10 days of onset of symptoms, with a person who has been diagnosed with SARS. History of travel, within 10 days of onset of symptoms to an area in which there are reported foci of transmission of SARS (see ANNEX 3). * Close contact means having cared for, having lived with, or having had direct contact with respiratory secretions and body fluids of a person with SARS. Probable Case A suspect case with chest x-ray findings of pneumonia or Adult Respiratory Distress Syndrome OR A person with an unexplained respiratory illness resulting in death, with an autopsy examination demonstrating the pathology of Respiratory Distress Syndrome without an identifiable cause. Note In addition to fever and respiratory symptoms, SARS may be associated with other symptoms including: headache, muscular stiffness, loss of appetite, malaise, confusion, rash, and diarrhea. PPHSN. SARS Guidelines 28/03/2003 16 Reporter details Name of person completing this form: _ _ _ _ _ _ _ _ _ _ _ _ _ Date of report to PPHSN _ _/ _ _ /_ _ _ Name of reporter: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ Position: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ Institution/Organisation: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ Country:____________ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ Contact telephone number: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ E-mail: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ Mobile phone number: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ Fax no. _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ Patient details Name: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ Sex: Female Male Date of birth: Surname: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _/ _ _/ _ _ _ _ Country of residence (If different from home address) ___________ Home address: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ City/town: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ Postcode: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ Country: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ Home telephone: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ PPHSN. SARS Guidelines 28/03/2003 Mobile phone: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ 17 Patient history In the last 10 days, has the patient been in contact with anyone who is a suspect or probable case of SARS? Yes No If yes: Type of contact: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ (e.g. family member, friend, etc) Name of the SARS contact: _____________________________________ Place of the contact: _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ Has the patient traveled since 1 February 2003 in one of the countries reported in the ANNEX? Yes No If yes Country visited Length of the stay From 1 2 3 4 To _____ _____ _____ ____ __/__/____ __/__/____ _____ _____ _____ ____ __/__/____ __/__/____ _____ _____ _____ ____ __/__/____ __/__/____ _____ _____ _____ ____ __/__/____ __/__/____ Date of return to (name country) Country of departure ____________ PPHSN. SARS Guidelines 28/03/2003 __/__/____ Airport of arrival in Country ___________ 18 Clinical details Date of onset of illness Clinical diagnosis _ _/ _ _ / _ _ _ _ Pneumonia Fever 38C Yes No ARDS Cough Yes No Other: _ _ _ _ _ _ _ _ _ Myalgia Yes No Fully recovered (symptom free) Recovering Stable Getting worse Not very ill Moderately ill Severely ill Died Current status: Shortness of breath/difficulty breathing Yes No Severity of illness: Other, please specify: ____________ Admitted to hospital? Yes __/__/__ No Admission status: Currently admitted Date of admission _ _ / _ _/ _ _ _ _ Hospital _________________ Transferred to other hospital Date of admission _ _ / _ _/ _ _ _ _ Hospital _________________ Discharged Date of discharge _ _ / _ _/ _ _ _ _ PPHSN. SARS Guidelines 28/03/2003 (Name and country) (Name and country) 19 Tests performed and laboratory results Chest X-ray ____________________________ Date _ _/ _ _/ _ _ _ _ Results: Normal Abnormal: ____________________________ ____________________________ Date _ _/ _ _/ _ _ _ _ Results: Normal Abnormal: ____________________________ ____________________________ Date _ _/ _ _/ _ _ _ _ Results: Normal Abnormal: ____________________________ Cell blood count Date _ _/ _ _ /_ _ Result Normal Thrombocytopenic Leucocytopenic Date _ _/ _ _ /_ _ Result Normal Thrombocytopenic Leucocytopenic Date _ _/ _ _ /_ _ Result Normal Thrombocytopenic Leucocytopenic (If performed) PPHSN. SARS Guidelines 28/03/2003 20 ANNEX 3 Affected Areas* - Severe Acute Respiratory Syndrome (SARS) Last revised by WHO – 27 March 2003 Country Area Canada Toronto Singapore Singapore China Beijing, Guangdong Province, Hong Kong Special Administrative Region of China, Shanxi, Taiwan Hanoi Viet Nam *An "Affected Area" is defined as a region at the first administrative level where the country is reporting local transmission of SARS. PPHSN. SARS Guidelines 28/03/2003 ANNEX 4 SARS Patient Management Flow Chart (Example) Patient arrival at hospital Patient Triaged on arrival Using SARS case definitions Reassurance Advice Usual care No Potential Case of SARS Yes Place mask on Patient and transfer to isolation area HCWs should wear mask or N95 respirator mask PRIOR to entering room to examine patient No •Provide advice on personal hygiene, avoidance of crowded areas and public transportation, remain at home until well. •Discharge with advice to seek medical care if respiratory symptoms worsen SUSPECTED case Of SARS (WHO definition) Yes Yes CXR and FBC, other specimen collection No Inform Public health authorities PPHSN. SARS Guidelines 28/03/2003 PROBABLE case of SARS (WHO definition) Yes Admit to designated isolation facility. Treat as indicated Contact tracing Advice to contacts Health Education Public health authorities informed ANNEX 5 Advice To Arriving Travelers (examples) During your recent travel if you have traveled through the affected areas listed below during the past 10 days, you may have been exposed to cases of severe acute respiratory syndrome. The usual incubation period for this illness is 2-7 days. You should monitor your health. If you become ill with fever accompanied by cough or difficulty in breathing, and you have been in one of the affected areas during the past 10 days you should seek medical advice (please see list below). To help the doctor make a diagnosis, tell him or her about your recent travel to these regions and whether you were in contact with someone who had these symptoms. Please save this card and give it to your physician if you become ill. Affected Areas* - Severe Acute Respiratory Syndrome (SARS) Country Area Canada Toronto Singapore Singapore China Guangdong Province, Hong Kong Special Administrative Region of China, Taiwan Province Viet Nam Hanoi Last revised 24 March 2003 *An "Affected Area" is defined as a region at the first administrative level where the country is reporting local transmission of SARS. List of Medical Practioners/Facilities where Assistance Can Be Obtained Name PPHSN. SARS Guidelines 28/03/2003 Address Telephone Fax SOLOMON ISLANDS - MINISTRY OF HEALTH AND MEDICAL SERVICES ADVISORY ON SEVERE ACUTE RESPIRATORY SYNDROME (SARS) The Centre for Disease Control and Prevention (CDC) and the World Health Organization (WHO) have received reports of patients with severe acute respiratory syndrome (SARS) from Canada, China, Hong Kong Special Administrative Region of China, Indonesia, Philippines, Singapore, Thailand, Vietnam, Germany and the United Kingdom. The cause of these illnesses is unknown and is being investigated. Early manifestations in these patients have included influenza-like symptoms such as fever, muscle pains, headache, sore throat, dry cough, shortness of breath, or difficulty breathing. Based on currently available evidence, close contact with an infected person is needed for the infective agent to spread from one person to another. This illness can be severe and, due to global travel, has spread to several countries in a relatively short period of time. Therefore, there is cause for concern. PPHSN. SARS Guidelines 28/03/2003 WHO has not recommended restricting travel to any destination in the world. However, all travelers should be aware of the main symptoms and signs of SARS. People who have these symptoms and within the previous 10 days have been in close contact with a person who has been diagnosed with SARS, or a history of travel to areas where cases of SARS have been spreading (see list), should seek medical attention and inform health authorities of recent travel. Travelers who develop these symptoms are advised not to undertake further travel until fully recovered. Please note that this situation is rapidly evolving and that the advice given will be constantly changing as more evidence about the causation and options for treatment becomes available. The public is advised to consult the home page of the WHO SARS website: http://www.who.int/csr/sars/en/ for daily updates on the outbreak and relevant press releases. This advisory is given by the Ministry of Health in collaboration with Immigration, Quarantine and Travel Agencies in Solomon Islands, aimed at preventing introduction of SARS into the country and ensuring the health and well being of the local population and visiting friends. To ensure that appropriate health measures are provided in case of an emergency, please respond to the following questions: Have you been to the following places / countries within the last 10 days? Guangdong Prov., China Hongkong, SAR, China Singapore Hanoi, Vietnam ROC / Taiwan Toronto, Canada Yes / No Yes / No Yes / No Yes / No Yes / No Yes / No If “Yes” are you experiencing the following symptoms? High Fevers Coughs Breathing Difficulties Yes / No Yes / No Yes / No If “Yes” to any of the above locations AND “Yes” to any of the above symptoms, please report to the Quarantine Desk at the arrival lounge. If you state “Yes” to any of the above locations and “No” to all of the above symptoms, you are advised to report to the nearest hospital or see your doctor in the event that you start to have these symptoms within the next 7 days and present this advisory. Full Name……………………………….……………. Passport No.………………………………………….. Nationality:…………………………………………… Address in Solomon Islands: ……………………………………………………… ……………………………………………………… ANNEX 6 Health Advice on the Prevention of Respiratory Tract Infections (Hong Kong MOH) Background In view of a recent outbreak of febrile respiratory illness among health care staff in Prince of Wales Hospital, the Department of Health is conducting a detailed investigation with the Hospital Authority and the Hong Kong University and Chinese University of Hong Kong to identify the cause of infection. The situation will be monitored closely. The Department of Health has informed the World Health Organization (WHO) about the latest developments. Hong Kong is working closely with the WHO on disease control and prevention. Advice applicable to all As a precautionary measure, members of the public are advised to take precautionary measures to prevent respiratory tract infections: o Build up good body immunity. This means taking a proper diet, having regular exercise and adequate rest, reducing stress and avoiding smoking o Maintain good personal hygiene, and wash hands after sneezing, coughing or cleaning the nose o Maintain good ventilation o Avoid visiting crowded places with poor ventilation o Consult their doctor promptly if they develop respiratory symptoms For schools and pre-school institutions Child Care Centres / Kindergartens / Schools are advised to refer to the 'Guidelines on Prevention of Communicable Diseases in Child Care Centres / Kindergartens / Schools' published by the Department of Health in 2000 for general information on the prevention of communicable diseases in school and institutional settings. This is downloadable from the DH's website http://www.info.gov.hk/dh. Specific advice in the school and institutional setting that helps to prevent respiratory tract infections includes : o Cleanse used toys and furniture properly o Keep hands clean and wash hands properly o Cover nose and mouth when sneezing or coughing o Wash hands when they are dirtied by respiratory secretions e.g. after sneezing o Use liquid soap for hand washing and disposable towel for drying hands o Do not share towels For other institutional settings A 'Guidelines on Prevention of Communicable Diseases in Residential Care Homes for the Elderly and People with Disabilities' published by the Department of Health is also available for general information on the prevention of communicable diseases in the PPHSN. SARS Guidelines 28/03/2003 particular institutional setting. This is downloadable from the DH's website http://www.info.gov.hk/dh. For health care workers in clinic setting There is as at date no unusual upsurge of pneumonia cases in the community. All clinic staff should enforce strict infection control measures appropriate for their particular setting, especially observance of good personal hygiene. If staff fall sick, they should report to their seniors and take sick leave as appropriate. Where considered necessary, for example, treating or nursing a patient with respiratory symptoms, staff may wear masks. The Department of Health will continue to monitor the situation of the pneumonia cases and issue advice accordingly. Patients with respiratory symptoms are advised to wear mask to reduce the chance of spread of the infection. Caring for sick family members with respiratory illness Patients should consult a doctor if they are unwell. They should follow instructions given by the doctor including the use of drugs as prescribed and taking adequate rest as appropriate. Adhere to good personal hygiene practices. Ensure adequate ventilation. Patients should put on masks to reduce the chance of spread of infection to carers. Carers may also put on masks to reduce the chance of acquiring infection through the airways. Advice to relatives visiting patients with pneumonia Visitors to warded patients are advised to take due precautions in infection control, e.g. wearing face mask and gowns and to wash hands thoroughly afterwards before coming into contact with other people. Notification of infections If unusual patterns of illnesses/sick leave in any setting are detected, please notify the respective Regional Office of the Department of Health. The contact numbers are as follows : PPHSN. SARS Guidelines 28/03/2003 ANNEX 7 Health Advice on the Prevention of Respiratory Tract Infections in Public Places (Hong Kong MOH) Members of the public are advised to avoid frequenting crowded public places to prevent the spread of respiratory tract infections. When visiting crowded places such as cinemas and restaurants, the following precautionary measures should be taken : Maintain good personal hygiene. Cover nose and mouth when sneezing or coughing Dispose of used tissue paper properly Keep hands clean. Wash hands when they are dirtied by respiratory secretions e.g. after sneezing Do not share towels Consult your doctor promptly if you develop respiratory symptoms, and follow instructions given by your doctor including the use of drugs as prescribed and adequate rest as appropriate Patients should put on masks to reduce the chance of spread of infection Workers in public places should take the following precautionary measures to reduce the chance of spread of infection : Maintain good personal hygiene. Cover nose and mouth when sneezing or coughing Wash hands after sneezing, coughing or cleaning the nose Consult your doctor promptly if you develop respiratory symptoms Allow plenty of fresh air into the indoor environment If the facilities are mechanically ventilated, ensure frequent air exchanges and proper maintenance and cleansing of the system Ensure that toilet flushing apparatus is functioning properly Provide toilets with liquid soap and disposable tissue towels or hand dryers Cleanse and disinfect the facilities (including furniture and toilet facilities) regularly (at least once a day), using diluted household bleach (i.e. adding 1 part of household bleach to 99 parts of water), rinse with water and then mop dry If the facilities are contaminated with vomitus, wash / wipe with diluted domestic bleach (mixing 1 part of bleach with 49 parts of water) immediately PPHSN. SARS Guidelines 28/03/2003 Infection Control ~ Doing it Right, Each Time Prepared by the Infection Control team, C&C DHB, 19 March 2003. ANNEX 8 New Zealand MOH Infection Control advice for the management of patients with suspected Severe Acute Respiratory Syndrome (SARS)* For detailed case definition and clinical management recommendations please consult the bulletin sent electronically to all C&C DHB staff on 18 March. Notify the Clinical Microbiologist/Infectious Diseases physician on call of any new suspected cases. SARS – should be suspected in patients presenting with symptoms of atypical pneumonia and a story within the last 10 days of recent travel or stay in South-East Asia or recent close contact with unwell travellers to that region or other areas of reported transmission of SARS. The pattern of illness suggests that SARS is infectious but at this stage the causative organism is unknown – the emphasis must therefore be on minimising contact by good infection control procedures as detailed below. To minimise exposure of other patients, staff and hospital visitors, the World Health Organisation and the US Centers for Disease Control & Prevention advise: 1. That all suspected SARS patients are managed in isolation using: • Standard (blood/body fluid) Precautions especially use of barrier protection when dealing with any blood or body fluids; use of masks (see airborne precautions below) plus eye protection if patients are coughing or procedures are done which are likely to generate splashes or sprays of respiratory secretions (eg. taking respiratory samples or suctioning) and scrupulous handwashing after removing gloves (see contact precautions below). WHO recommends that HCWs must wear protective eyewear or faceshields during procedures where there is potential for generation of aerosols or splashing, splattering or spraying of blood or other body substances (this recommendation is not included in original NZ guidelines). plus • Airborne Precautions placement of the patient in a negative pressure air-conditioned room and use of respiratory protection – particulate filter respirator masks (TB masks - Tecnol PCM 2000 mask) - by all staff and visitors, plus • Contact Precautions use of non-sterile gloves and disposable, impervious isolation gown by all staff and visitors for any contact with the patient, the patient’s body fluids, with patient care equipment or with used linen or with waste 2. Patient masking for transport (ambulance or internal trolley transport) and short visits to other clinical departments (eg. Radiology) • Patient movement outside negative pressure rooms should be avoided as much as possible. • A standard surgical mask (eg. Tecnol Procedure mask 6001 or any other droplet containing mask) must be placed over the patient’s mouth and nose for transport through corridors and public areas (eg. between clinical areas such as Emergency Department and Radiology / wards /units) or for investigation in departments such as Radiology. • Attending staff do not then require masks but should continue use of gloves and gown for direct contact with the patient, with blood or body fluids or with patient equipment. • Receiving departments should be advised of the patient’s isolation status when transfer or investigations are ordered and patients must be escorted during transport between negative pressure areas to ensure that appropriate precautions are maintained. • See Equipment and Cleaning details below for advice on necessary equipment and environmental surface cleaning and decontamination following procedures on suspected SARS patients. PPHSN. SARS Guidelines 28/03/2003 3. Emergency Dept. and Ward management details Isolation room details • The patient must be isolated in a negative pressure single room – eg. rooms A3 or C7 in the Emergency Dept. or a room (preferably with attached ensuite bathroom) within the airborne isolation area in Ward 17. • The isolation room door must be kept closed at all times except when required by entry or exit of personnel. • A laminated “STOP” isolation sign (as distributed by the Infection Control Officers) must be placed on the door or where it will be visible to all who enter the room • The patient should leave the room only when clinically necessary (see point 2. above). • Microshield 4 antiseptic handwash should be used for all handwashing within the room and at adjacent washbasins outside the room (eg. in the anteroom) immediately following removal of protective clothing on exiting the isolation room. Handwashing is the most important hygiene measure for preventing the spread of infection. Gloves are not a substitute for handwashing. Hands must be washed and gloves replaced before and after significant contact with the patient, after activities involving handling of contaminated items and after removal of gloves. • All staff and visitors must put on mask, gowns and gloves as specified before entering the isolation room. • On leaving the isolation room, staff and visitors should remove gown, gloves and mask (in that order, masks should be handled only strings); place them into a yellow Biohazard waste bin and immediately wash their hands. (In Ward 17 this is done in the ante-room to the isolation rooms; in the Emergency Dept. this must be done immediately outside the isolation room and then the nearest washbasin must be used for handwashing.). These are single use items - fresh gowns, gloves and masks must be put on for any subsequent entry into the isolation room. • Provide patients with ample supplies of disposable tissues and teach them to cover their mouth and nose when coughing or sneezing. Tissues must be handled and disposed of as Biohazard waste. • Patient notes, including medicine and observation charts (and clinical staff’s pens) must not be kept or taken into the room. • Patient samples/specimens should be handled in the usual manner (placed in a Biohazard specimen bag for transport to the laboratory accompanied by a completed request form) Staff allocation • The primary focus must be appropriate, skilled response to the clinical needs of the patient • The nurse allocated to direct patient care on each shift is responsible for directing other health care workers and visitors on the nature of the precautions required • The number of staff allocated to the patient should be as small as possible to decrease the possibility of transmission to other patients within the ward or to other areas of the hospital. eg. Blood tests are to be taken by the Medical Officer ordering the test rather than being put out for Phlebotomy service staff • Whenever possible, care is provided by nurses/staff usually working in the ward • If care cannot be provided from within the ward, the Coordinator Central Nursing, her deputy or the after hours manager is contacted for assistance by the nurse in charge and consultation must include the Clinical Microbiologist/Infectious Diseases physician on call • A nurse employed by the Casual Resource may look after the patient only if s/he has the skills and knowledge necessary to care safely and confidently for the patient, and the Central Nursing Coordinator and the nurse agree to the assignment. Equipment and supplies • Disposable equipment should be used wherever possible in the treatment of patients with suspected SARS. • A range of sizes of non-sterile gloves must be available inside and at the entrance to the isolation room. Gloves must be changed as clinically indicated and removed and disposed of in the Biohazard waste each time personnel leave the isolation room. • Disposable, impermeable isolation gowns are recommended (eg. Baxter yellow disposable isolation gowns). Gowns are single use items and may not be kept for reuse – they must be removed and PPHSN. SARS Guidelines 28/03/2003 disposed of in the Biohazard waste when personnel leave the isolation room and a new gown must be worn for re-entry. • Masks: Particulate filter respirator (Tb) masks must be available (eg. Tecnol PCM 2000 mask), preferably the fluid repellent orange version (PCM 2000 reorder number 47707 – available from Supply Dept.) and in the fluid repellent orange version with attached eyeguard (PCM 2000 reorder number 47757 – available as a buy-in, 15/box). Standard surgical masks (eg. Tecnol Procedure mask 6001 – available from Supply Dept.) must be available for use by the patient to prevent dispersal of respiratory droplets if the patient must leave the isolation room. Masks are single use items and must be removed and disposed of in the Biohazard waste each time staff and visitors leave the isolation room and a new mask must be worn for re-entry. • Equipment and supplies necessary for patient treatment, safety and comfort must be available in the isolation room but storage of supplies and equipment within the room must be kept to a minimum and be replenished daily if necessary. • All non-disposable equipment (eg. blood pressure cuffs, stethoscopes, tympanic membrane thermometers and including mobile units such as X-ray machines, IV pumps etc.) that is taken into the room must be decontaminated immediately after removal from the isolation room and before it may be reused in the care of other patients. At a minimum, items will require cleaning by surface wiping with a disposable cloth, detergent and water (or specialist products if these are normally used) and usual disinfection and sterilisation processes should be used for items normally reprocessed by these methods. • It is recommended that some dedicated items of non-disposable equipment (eg. blood pressure cuffs) are left in the room for the duration of the patient’s isolation so that discharge/terminal cleaning only is likely to be required. • Bedpans and urinals should be emptied and reprocessed (immediately after use ie. Should not be left unprocessed on communal dirty benches in utility rooms) by the usual method in ward sanitisers. The isolation nurse may need assistance from other nursing staff outside the isolation room to facilitate this (these staff should use gloves, disposable aprons and facial protection when handling used toilet items and plan the transfer of items so that minimal contamination of environmental surfaces occurs). • Metal surgical instruments requiring sterilisation in the Sterile Production Centre can be placed in a clear plastic bag, the opening secured and then returned in the usual container to SPC for routine decontamination (full body and facial protective equipment is worn routinely in the SPC decontamination area). • A designated sharps container must be available in the room plus phlebotomy equipment if required (tourniquet, vacutainer collection system, A.N.D. disposal unit for vacutainer needles, etc). • The wash bowl is kept in the room for the duration of the isolation. It is cleaned with hot soapy water after use, dried, then stored, inverted, off the floor. Bowls used to clean patients after incontinence episodes are to be emptied in the sluice room, rinsed and wiped over with 1% Chlorine solution then sanitised in the usual manner. Food service • Usual meal trays, plates and cutlery may be used. Menus and trays do not need special marking or bagging as isolation items. • After use, waste food should be disposed of within the isolation room and the trays and utensils should be returned directly (with assistance from staff working outside the isolation area) to the Food Services trolley for return to the kitchen for reprocessing. Staff in this area routinely wear protective clothing and the usual machine dishwashing process is adequate to decontaminate the trays and utensils. Linen and waste handling • A linen skip (with a cloth linen bag lined with a hot water soluble alginate liner) and a Biohazard yellow waste bag must be present in the isolation room. • All waste (except sharps which must go into the sharps container) produced in the room must go into the Biohazard waste bag. • Care should be taken not to shake or flap the bedlinen. Change the bedlinen completely each morning shift and carefully scoop and fold used linen to place it in the used linen container. • Linen and waste bags are to be replaced at least daily and when two thirds full. • Nursing staff are responsible for the closure and replacement of bags. Double bagging and labelling are not required. PPHSN. SARS Guidelines 28/03/2003 Visitors • Visitors other than close family members should be discouraged as the causative organism and infectivity of SARS is not yet clear and specific treatments have not yet been identified. • Request visitors not to visit other patients in hospital if they are visiting patients with suspected SARS. • All visitors must wear full protective clothing (gown, gloves and mask) to enter the isolation room. (See procedures detailed for staff). • Nursing staff are responsible for ensuring that visitors comply with isolation procedures and should explain and assist as necessary. Daily cleaning • The isolation room must be cleaned daily. • Under the direction of the Team Leader, Clinical Coordinator or deputy, the cleaner must be directed and shown how to use full protective clothing (gown, gloves and mask). • The room (and ensuite) must be cleaned last of all the rooms on the ward. Disposable cleaning cloths must be used. • The cleaner must dust to a height of 6 feet the door, bed frame, bed light, window sills and furniture, shelves and ledges, trolleys and equipment. Dust must not be shaken out of cleaning cloths but must be contained by folding inwards. • Locker tops, washbasins, taps and door handles must be cleaned with clean cloths, fresh hot water and detergent. • The floor must be wet mopped with clean hot water and detergent. • Isolation bathroom areas must be cleaned following the isolation room, using the same precautions. • Dispose of all cleaning cloths in the Biohazard bag in the isolation room. The mop head must be placed in an alginate liner bag before being sent to the Laundry for routine laundering – special labelling is not required. • The bucket is to be washed thoroughly with hot water and detergent and turned upside down to dry. • The cleaner must be shown how and where to take off protective clothing in a safe manner, dispose of it into a Biohazard container and instructed to wash their hands immediately. Terminal cleaning (on patient discharge) • Infection Control advice must be sought for the decontamination complex equipment. • Nursing staff are responsible for supervising the safe stripping and cleaning of the isolation room and bathroom including instructing and supervising the cleaner in putting on protective clothing. • Strip the bed of linen. Check for breaks in the impervious covering of the pillow and mattress. Only when breaks are detected - dispose of these items as Biohazard waste (pillows can be placed in a Biohazard waste bag; if mattresses are to be disposed of they must be contained within clean large plastic bags, clearly labelled as intended for Biohazard waste and Orderlies should be contacted to collect and place them directly into a large yellow Biohazard waste bin for transport to the Infectious Waste room.) • Dispose of disposable equipment (used or unused stock) and other waste into the Biohazard waste bag. • Leave the waste and linen bags to be tied off by the cleaner once cleaning is completed so that cleaning cloths and protective clothing can be discarded safely. • The cleaner must clean all the surfaces including isolation bathrooms and floors as listed in the daily cleaning requirements with clean disposable cloths, clean hot water and detergent. • All cleaning cloths must be disposed of into the Biohazard waste bag. Mop heads must go to the Laundry as detailed above in Daily cleaning and the bucket must be cleaned as above. • Curtains: In the Emergency Department, ask the cleaner to check the curtains for soiling; if present, ask for the curtains to be changed. In the ward or unit setting, ask the cleaner to change the curtains. (New curtains must not be put up until the room has been fully cleaned and aired for 1 hour following completion of cleaning). • The cleaner must be shown how and where to take off protective clothing in a safe manner, dispose of it into a Biohazard container and instructed to wash their hands immediately. • The rooms should be left with the door closed for 1 hour before the bed is remade, curtains are rehung and the room is re-occupied. This is to allow for sufficient air changes to occur to ensure removal all possibly contaminated air. PPHSN. SARS Guidelines 28/03/2003 This information is subject to change and updating as further information and direction comes to hand from WHO, CDC and the NZ Ministry of Health. For further infection control advice • Please contact an Infection Control Officer (Clo Taylor on ext/page 5925 or Viv McEnnis on ext/page 6514). References 1. Garner JS, Hospital Infection Control Practices Advisory Committee “Guidelines for Isolation Precautions in Hospitals” Infection Control & Hospital Epidemiology 1996; 17: 53-80. 2. US Centers for Disease Control and Prevention information – Severe Acute Respiratory Syndrome “Interim Information and Recommendations for Health Care Providers” 15 March 2003. 3. NZ Ministry of Health – information distributed electronically on 18 March 2003 and sourced from WHO and CDC. PPHSN. SARS Guidelines 28/03/2003 ANNEX 9 Assessment of Risk and Capacities to Respond to a Multi-country outbreak of Severe Acute Respiratory Syndrome The following information has been prepared to assist WHO WRs and CLOs when liasing with national health authorities (NHA) regarding public health response to the current outbreak of Severe Acute Respiratory Syndrome (SARS). PPHSN thinks that this information should also be obtained from countries without WHO offices so that arrangements can be made to enhance our preparedness. PPHSN has already circulated interim guidance about SARS which will be updated continuously and circulated, as knowledge about this disease increases. (1) WPRO has provisionally categorized member countries based on 3 levels of risk and 3 levels of capability to respond to the outbreak. Risk Categories R1 countries where cases have been reported R2 where traffic risks exist between these and R1 countries R3 all other WPR countries Capability to Respond Categories C1 countries needing the most assistance from WHO in terms of emergency supplies, enhanced surveillance and technical assistance C2 countries with limited national resources and requiring some emergency assistance C3 countries not requiring much assistance except for technical advice and international network coordination Category R1 C1 Vietnam C2 China C3 Singapore R2 Cambodia Lao PDR Northern Mariana Islands Malaysia Philippines Macao SAR Republic of Korea Brunei Darussalaam Guam Australia Japan New Zealand R3 PNG Mongolia All other PICs Fiji New Caledonia * This assessment is current as of 20 March 2003. Revisions may be necessary as the SARS epidemic evolves, and will be included as necessary in subsequent updates. A kit of supplies developed to address initial requirements for management of an outbreak of SARS in select countries will be dispatched this week to Laos, Mongolia, Cambodia, Papua New Guinea and Fiji (2 kits). A list of contents is attached. Attachments WPRO SARS Preparedness Kit contents list. PPHSN. SARS Guidelines 28/03/2003 Check list for National Health Authorities PPHSN requests that EpiNET focal points please liase with their health authorities to complete the following information about preparedness to deal with cases of SARS. WHO / National SARS Preparedness Checklist (attach additional pages if necessary) . Action. Details. Status / contact details (tel/fax –office and after hours), email WHO Country Focal Point National Focal Point Point of first contact for national and WPRO SARS MOH/DOH designated SARS focal point / coordinator Hospital with isolation (preferably intensive care) facilities designated by MOH /DOH for SARS admissions Laboratory with appropriate infection control capability designated by MOH /DOH for SARS specimen processing and/or trans-shipment. Consider P3 / Polio Reference Laboratory if available, or measles reference lab, in close proximity to isolation centre WR or CLO in countries where present Designated SARS Isolation Facility (s) Designated SARS laboratory Institute Pasteur Alain Berlioz-Arthaud, Laboratoire de Biologie Medicale, Institut PASTEUR de Nouvelle Caledonie, BP 61, 98845 Noumea, New Caledonia. Tel : +(687) 27.02.85 Fax : +(687) 27.33.90 Email : [email protected] WHO Collaborating Centre for Reference and Research on Influenza, Australia Ian Barr WHO Collaborating Centre for Reference and Research on Influenza 45 Poplar Road, Parkville. Victoria, Australia 3052 Tel +(61) 3 9389 1761 Fax + (61) 3 9389 1881 Email [email protected] National SARS preparedness stockpile Immediately identifiable requirements from WHO. Other national SARS preparedness activities. Readily available barrier nursing and pathology equipment secured by DOH/MOH, (available, and planned. See attached WPRO SARS Preparedness Kit list for guidance. Refer to attached WPRO SARS Preparedness Kit list. Action taken so far by national health authorities. PPHSN. SARS Guidelines 28/03/2003 Daily Updates Please advise whether you can access the WHO daily epidemiologic update via the [ ] internet (http://www.who.int/csr/sars/) or would you like a copy sent by: [ ] fax [ ] email as attachment After completion Please return completed form by email or fax to: PPHSN-CB Focal Point Dr Tom Kiedrzynski Epidemiologist (Ag) Secretariat of the Pacific Community, PPHSN-CB Focal Point SPC PO Box D5 98848 Noumea cedex New Caledonia Tel: +(687) 26 20 00 or 01 43 Fax: +(687) 26 38 18 Out of hours +(687) 259236 E-mail: [email protected] WHO South Pacific Dr Kevin Carroll MO/Epidemiologist WHO South Pacific PO Box 113 Suva, Fiji Tel +(679) 3304 600 Fax +(679) 3300 462 Out of hours +(679) 99202971 Email [email protected] PPHSN. SARS Guidelines 28/03/2003 ANNEX 10 WPRO SARS Preparedness Kit contents list. The list below is designed to provide essential equipment for barrier nursing and pathology collection for suspected SARS cases. It is based on estimated needs for maximum 50 health workers per day for 3-4 days, and high-level protection for 6-8 procedural / laboratory workers for a similar period. INFECTION CONTROL EQUIPMENT Coveralls Disposable Aprons (yellow) Shoe covers Hair cover (bouffant) Safety glasses Non-fog goggles UVEX goggles Hepa Mask N95 particulate mask Disposable gloves Surgical gloves Anti-microbial waterless hand-wash Anti-microbial waterless hand-wash refills Absorbent laboratory mat Aprons: impermeable Rubber boots 30 200 200 pair 200 30 50 10 30 200 500 pair 150 10 bottles 10 bottles 2 rolls 200 10 pairs 10 large, 10 medium, 10 small (includes hood) polypropylene Biohazard disposal bags 100 autoclavable Polypropylene 2-lens Can be worn with glasses/spectacles 100 large, 200 medium, 200 small > 60% alcohol, with emollient ('Sterillium 1000ml) dispensor (Sterillium 1000 ml) 3 x 50ft 2 L, 4 M, 4 S LABORATORY EQUIPMENT Sterile cotton swab sticks Vacutainer 5 ml EDTA Vacutainer 5ml plain Vacutainer needles Vacutainer adapters Syringe 5ml Butterfly needles Sterile screw-top plastic specimen containers 1 box 1 box 1 box 200 200 100 50 25 ml (1000) (50) (100) 100 SPECIMEN SHIPPING Bio-bottles 0.5L shipping containers Cryogenic vials 5ml Cooler, large capacity Ice packs Ziplock bags PPHSN. SARS Guidelines 28/03/2003 2 100 1 12 200 For transport of field specimens