* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Analysis of p75NTR-dependent apoptotic
Survey
Document related concepts
Cytokinesis wikipedia , lookup
Phosphorylation wikipedia , lookup
Cellular differentiation wikipedia , lookup
Hedgehog signaling pathway wikipedia , lookup
Protein phosphorylation wikipedia , lookup
Nerve growth factor wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
List of types of proteins wikipedia , lookup
Programmed cell death wikipedia , lookup
Signal transduction wikipedia , lookup
VLDL receptor wikipedia , lookup
Transcript
Analysis of p75NTR-dependent apoptotic pathways and of p75NTR gene products
Christine E. Paul
Neurology and Neurosurgery
McGill University
Montreal, Quebec
July, 2003
A Thesis submitted to the McGill University Faculty of Graduate Studies and Research in
partial fulfilment of the requirements of the degree of Master in Science
©Christine Paul 2003
11
Table of contents
Abstract
iv
Resume
vi
Acknowledgements
vii
List of Abbreviations
viii
Contribution of authors
x
Chapter 1: Introductory Literature Review
1
Developmental Apoptosis
The neurotrophins
p75NTR
The pan neurotrophin receptor
p75NTR is structurally similar to other TNFR superfamily members
Other p75NTR ligands
p75NTR Functions
p75NTR as a co-receptor for Trk
p75NTR as a co-receptor for NogoR
p75NTR autonomous signaling (death and survival)
The p75NTR knockouts
p75NTRExonm-/-mice
p75NTRExonIV-/-mice
Statements of projects/hypotheses
2
2
4
5
7
8
9
10
12
17
17
22
23
Preface to Chapter 2
25
Chapter 2: p75NTR does not transcriptionally activate BH3-only proteins
26
I.
II.
III.
IV.
V.
VI.
VII.
Literature Review: the BH3-domain-only proteins
Statement of project/hypothesis
Materials and Methods
Results
Closing Remarks
Figure Legends
Figures
i. Figure 1
27
30
31
33
34
37
38
Ill
Preface to Chapter 3
39
Chapter 3: A Pro-apoptotic Fragment of the p75 Neurotrophin Receptor is
expressed in p75NTRExonlv null mice
.40
I.
II.
III.
IV.
V.
VI.
VII.
VIII.
IX.
Title Page
Acknowledgements
Abstract.
Introduction
Materials and Methods
Results
Discussion
Figure Legends
Figures
i. Figure 2
ii. Figure 3
iii. Figure 4
41
42
43
44
46
51
56
58
62
63
64
Closing Remarks
65
Bibliography
70
Appendix
88
Asha L. Bhakar, Jenny L. Howell, Christine E. Paul. Amir H. Salehi, Esther B. E. Becker,
Farid Said, Azad Bonni and Philip A. Barker. Apoptosis Induced by p75NTR
Requires Jun Kinase-dependent Phosphorylation of Bad. Submitted to EMBO J, July
2003.
89
Research Compliance Certificate: McGill University Animal Use Protocol
124
IV
Title: Analysis of p75NTR-dependent apoptotic pathways and of p75NTR gene
products
Abstract: The p75 neurotrophin receptor (p75NTR) binds members of the neurotrophin
family and plays important roles in the regulation of neuronal survival, apoptosis and
growth during development and after nervous system injury. Many in vitro and in vivo
studies have shown that p75NTR induces cell death, though the signaling events that link
p75NTR activation to apoptosis are not thoroughly understood.
p75NTR-dependent
apoptosis is associated with an increase in Rac and Jun kinase (JNK) activity, and recent
work from our laboratory has shown that the p75NTR interactor, NRAGE, activates a
mitochondrial death pathway involving JNK-dependent cytochrome C release and
activation of Caspase-9, Caspase-7 and Caspase-3. Despite this progress, several
important details of p75NTR apoptotic signaling remain unknown.
In particular, it is unclear what targets of p75NTR-dependent JNK activation
result in mitochondrial cytochrome C release and caspase activation. BH3-domain-only
proteins are members of the Bcl-2 family that induce the association of Bax and Bak
which in turn facilitate release of mitochondrial proteins such as cytochrome C into the
cytosol. Transcriptional activation of BH3-domain-only genes through c-Jun- or p53dependent pathways is implicated in apoptosis in neuronal and non-neuronal systems. In
the first part of this thesis, I examined whether p75NTR-induced apoptosis is correlated
with accumulation of BH3-domain-only gene products. U373 human glioma cells and
mouse cortical neurons were infected with adenovirus expressing p75NTR to
constitutively activate p75NTR-dependent pathways, and alterations in mRNA levels of
the BH3-domain-only family members Bim, Bmf, Hrk, Bik, Puma, and Noxa were
determined by RT-PCR. The results from these experiments showed that p75NTRmediated cell death did not result in BH3-domain-only gene transcription. Subsequent
studies in our laboratory established that the BH3-domain-only protein, Bad, is
phosphorylated on Serine 128 in a JNK-dependent manner, and that this phosphorylation
is a critical component of p75NTR-dependent apoptosis.
The generation of animals that lack p75NTR expression has been a critical
advance in understanding the in vivo role of this receptor. In the second part of this
thesis, I analyzed the recently created p75NTRExonlv-/- mice, which were shown to
produce a null mouse lacking all p75NTR gene products, in contrast to the previously
constructed p75NTRExon1"-/- mouse, which maintains expression of an alternatively
spliced form of p75NTR (s-p75NTR). Our studies revealed that p75NTRExonIV-/- mice
continue to express a p75NTR gene product that encodes a truncated protein containing
the p75NTR extracellular stalk region together with the entire transmembrane and
intracellular domains.
The gene product is initiated from a cryptic Kozak
consensus/initiator ATG sequence within a region of Exon IV located 3' to the pGK-Neo
insertion site, likely as a result of enhancer elements within pGK-Neo cassette.
Characterization of this protein product indicated that it localized to the plasma
membrane and overexpression of the p75NTRExonIV fragment in heterologous cells
resulted in activation of JNK and cleavage of Procaspase 3, indicating that it can mediate
pro-apoptotic effects in vivo. These results indicate that aspects of the p75NTRExonIV-/phenotype may reflect a gain-of-function mutation rather than a loss of p75NTR function.
VI
Titre: Analyses des proprietes de signalement apoptotiques du recepteur de
neurotrophines p75NTR et analyses de souris, p75NTRExonIV -/-
Resume:
II est difficile d'identifier clairement la fonction du recepteur de
neurotrophines p75NTR, cependant plusieurs etudes indiquent qu'il peut activer des
signaux apoptotiques. Durant la premiere partie de ma maitrise, j'ai etudie la famille de
proteines denommee "BH3-domain-only", membres de la famille Bcl-2. Ces dernieres
interagissent avee les proteines Bax et Bak causant ultimement l'activation des caspases.
Utilisant un systeme ou p75NTR est exprime par un adenovirus, j'ai determine que les
proteines "BH3-domain-only" ne sont pas activees au niveau de la transcription durant
l'apoptose induite par 1'expression de p75NTR.
Des travaux effectues dans notre
laboratoire ont reveles que les signaux apoptotiques de p75NTR mene a la
phosphorilation de la proteine Bad, membre de la famille "BH3-domain-only", au niveau
de la Serinel28. De plus, cette phosphorylation depend de l'activation de la cascade
enzymatique de JNK et elle est essentielle a l'induction de l'apoptose par p75NTR.
Durant la deuxieme partie de ma maitrise, j'ai etudie la souris p75NTRExonIV -/- qui
a ete creee recemment. It a ete determine, suite a mes travaux, que cette souris exprime
toujours une certaine portion du recepteur p75NTR. Cette portion comprendrait tout le
domaine transmembranaire and intracellulaire. La transcription de cette partie du
recepteur est causee par une region "enhancer" situee dans la cassette pGK-Neo, qui a ete
introduite dans le quatrieme exon de p75NTR durant la modification du gene de
p75NTR. L'expression de cette portion de p75NTR dans plusieurs types de cellules a
mene a l'activation de la cascade enzymatique de JNK and au clivage de la Procaspase-3,
indiquant que p75NTRExonIV mene a des evenements apoptotiques. En conclusion, les
resultats obtenus indiquent que les aspects de la mutation p75NTRExonIV reflectent un gain
et non pas une perte de fonction.
Vll
Acknowledgements
I would like to thank my supervisor, Phil Barker, for providing such a great
environment in which to practice science. A part of that environment includes the people
in my surroundings. As such, I would like to thank past and present members of the
Barker lab for their friendship, guidance and support. I would like in particular to say
thanks to Asha Bhakar, Philippe Roux and Amir Salehi for showing me the ropes when I
first arrived. I would also like to acknowledge Genevieve Dorval (who helped me
translate my abstract-thank you!) and Kathleen Dickson, who both helped me with
numerous technical aspects of my projects.
I would also like to thank my advisory committee, who comprise Dr. Ted Fon and
Dr. Peter McPherson, for helpful advice throughout my studies. Thanks also to my
academic mentor, Dr. Andrea Leblanc, and to Dr. Wayne Sossin for helpful discussions.
I would like to thank my family (West Coast and East Coast contingencies alike)
and my roomate, Carolyn Picco, for her everpresent support and friendship. Also, to
other "MNIers" (especially to Amy Corcoran, Natasha Hussain, Andrew Jarjour, Tamra
Werbowetsky, Anna Lee, Nic Tritsch and Simon Moore) without whom my Msc would
never have been this much fun. In the social department, I would also like to thank the
Lobotomizers, Ultimate team, FACE people, and fellow RVH-PCU volunteers, who
helped make life great in Montreal.
Finally, I would like to acknowledge Greg Walsh, for so many little things, the
sum of which is much greater than the parts! You da best.
Vlll
List of Abbreviations
AP-1
Activating Protein-1
APP
Amyloid Precursor Protein
BDNF
Brain-Derived Neurotrophin Factor
BH
Bcl-2 Homology
CGN
Cerebellar Granule Neurons
CNS
Central Nervous System
COS-7
CV-1 cells expressing Simian virus-40 antigen
CRD
Cysteine Rich Domain
CRIB
Cdc42/Rac-Interactive Binding
CRNF
Cysteine-Rich Neurotrophic Factor
DD
Death Domain
DISC
Death-Inducing Signaling Complex
DRG
Dorsal Root Ganglia
ECD
Extracellular Domain
FADD
Fas receptor Associated Death Domain
FL-p75NTR Full Length p75NTR
GCK
Germinal Center Kinase
GDP
Guanine Diphosphate
GFP
Green Fluorescent Protein
GPI
GlycosylPhosphatidyllnositol
Guanine Triphosphate
GTP
Human Embryonic Kidney
HEK
Inhibitor of Apoptosis
IAP
Intracellular Domain
ICD
JNK Interacting Protein
JIP
c-Jun N-terminal Kinase
JNK
Myelin-Associated Glycoprotein
MAG
Mitogen Activated Protein Kinase
MAPK
Mixed Lineage Kinase
MLK
p75NTR-associated cell death executor
NADE
Nerve Growth Factor
NGF
Neurotrophin Receptor Interacting Mage Homolog
NRAGE
Neurotrophin-3
NT-3
Neurotrophin-4/5
NT-4/5
Nogo Receptor
NgR
Oligodendrocyte-Myelin Glycoprotein
OMgP
p75
Neurotrophin Receptor
p75NTR
PhosphoGlycerate Kinase
pGK
Pheochromocytoma Cell line 12
PC12
Phosphatidylinositol 3-Kinase
PI-3K
Protein Kinase A
PKA
PhosphoLipase C
PLCy
Peripheral Nervous System
PNS
Pro-Nerve Growth Factor
ProNGF
Receptor Activator of NF-kappa (3
RANK
IX
Rho-GDI
ROCK
RTK
SDS-PAGE
s-p75NTR
TNF
TNFR
TRADD
TrkA
TrkB
TrkC
TUNEL
Rho GDP Dissociation Inhibitor
Rho-associated serine-threonine protein kinase
Receptor Tyrosine Kinase
Sodium Dodecyl Sulfate-PolyAcrylamide Gel Electrophoresis
Splice variant of p75NTR
Tumor Necrosis Factor
Tumor Necrosis Factor Receptor
TNF Receptor Associated Death Domain
Tropomyosin Related Kinase A
Tropomyosin Related Kinase B
Tropomyosin Related Kinase C
Terminal deoxynucleotidyl transferase biotin-dUTP Nick End Labeling
Contribution of authors
Chapter 2: p75NTR does not transcriptionally activate BH3-only proteins
I performed the design of primers, preparation of cDNA from infected cells, and
subsequent RT-PCRs. This work was apart of the following manuscript, that is included
in the appendix of this thesis:
Asha L. Bhakar, Jenny L. Howell, Christine E. Paul. Amir H. Salehi, Esther B. E. Becker,
Farid Said, Azad Bonni and Philip A. Barker. Apoptosis Induced by p75NTR
Requires Jun Kinase-dependent Phosphorylation of Bad. Submitted to EMBO J, July
2003.
Chapter 3: A Pro-apoptotic Fragment of the p75 Neurotrophin Receptor is expressed in
p75NTRExonlv -/- mice
I performed all the experiments and helped to write the manuscript:
Christine E. Paul and Philip A. Barker. A Pro-apoptotic Fragment of the p75
Neurotrophin Receptor is expressed in p75NTRExonIV -/- mice. Submitted to Nature
Neuroscience, July 2003.
Chapter 1: Introductory Literature Review
Developmental Apoptosis
During embryonic and early postnatal development, many neurons formed by
neurogenesis are killed in an active form of cell death known as apoptosis. This process
results in death of up to 50% of all neurons initially created, in order to create appropriate
connections between neurons and their target organs, and to allow for proper
morphogenesis such as neural tube closure (Jacobson et al., 1997; Oppenheim, 1991).
There is also increasing evidence that apoptosis contributes significantly to the pathology
of human neurodegenerative diseases (Pettmann and Henderson, 1998).
The
neurotrophins are a family of growth factors that act as critical mediators of the survival
or death of neurons in both the developing and the mature nervous system. While
initially identified as target-derived neuronal survival cues (Oppenheim, 1991; Purves et
al., 1988), the neurotrophins are now widely recognized to elicit a variety of responses
ranging from regulation of proliferation, synaptic plasticity, neuritic outgrowth, survival
and apoptosis.
The Neurotrophins
In mammals, there have been four neurotrophins identified, which include nerve
growth factor (NGF), brain-derived neurotrophic factor (BDNF), neurotrophin-3 (NT-3)
and neurotrophin-4/5 (NT-4/5). NGF, the prototypical neurotrophin, was discovered
more than fifty years (Huang and Reichardt, 2001; Levi-Montalcini, 1987) as a survival
factor that enabled dorsal root ganglia to survive during a "critical phase" in which they
normally underwent massive and rapid cell death. Initial discoveries by Rita LeviMontalcini, Stanley Cohen and Victor Hamburger led to the neurotrophic hypothesis,
which maintained that the number of neurons innervating a target is limited by the
amount of target-derived trophic factor made available to these neurons.
In addition to NGF, three other neurotrophins have been discovered in mammals.
BDNF was the second discovered neurotrophin, and sequence homology between NGF
and BDNF prompted the search for the other mammalian neurotrophins, NT-3 and NT4/5, which were discovered by cloning (Ernfors et al., 1990; Hohn et al., 1990) in the
early 1990's.
All four neurotrophins share similar primary, secondary, tertiary and quaternary
structure. They are initially synthesized as 31-35 kDa precursors that are then cleaved by
a variety of processing enzymes to give rise to the mature processed neurotrophins (13.215.9 kDa).
Once fully processed, neurotrophins exist as non-covalently linked
homodimers that are secreted through either the constitutive secretory pathway (in the
case of NGF and NT-3) or through a regulated activity-dependent pathway (in the case of
BDNF) (Ghosh et al., 1994; Mowla et al., 1999). Each neurotrophin promotes the
survival of specific populations of mammalian neurons.
Neurotrophins are unique in that they use two different receptors, the
tropomyosin-related kinase (Trk) receptor and the p75 neurotrophin receptor (p75NTR),
a member of the tumor necrosis factor receptor (TNFR) superfamily. The Trk receptors
are highly related transmembrane receptor tyrosine kinases (RTKs) that are expressed in
discrete neuronal populations and which are bound by specific neurotrophins. Trk A
binds preferentially to NGF, Trk B binds BDNF and NT-4/5, and Trk C binds NT-3
(Cordon-Cardo et al., 1991; Ip et al., 1993; Kaplan et al., 1991a; Kaplan et al., 1991b;
Klein et al., 1990; Klein et al., 1991a; Klein et al., 1991b; Lamballe et al., 1991; Soppet
et al., 1991; Squinto et al., 1991). Activation of Trk receptors by their preferred
neurotrophic ligand elicits the well-known survival effects associated with the
neurotrophins, as shown in a number of gain and loss of function studies. For instance,
ectopic expression of Trk receptors confers neurotrophin-dependent survival and
differentiation in many neurons (Allsopp et al., 1993; Barrett and Bartlett, 1994). The
physiological importance of the Trks in neuronal survival is most clearly demonstrated in
mice with Trk receptor gene deletion, where extensive loss of specific neuronal
populations are observed (Snider, 1994). Many studies have shown that Trk activation
results in phosphorylation on intracellular residues Tyrosine 490 and Tyrosine 785, which
serve as important docking sites for adaptor proteins that in turn activate the Ras, PI-3K
and PLCy pathways, leading to modulation of neuronal survival, growth and
differentiation (Kaplan and Miller, 2000; Roux and Barker, 2002).
While the mechanism of action of the Trks has been well characterized, the roles
of p75NTR appear complex and have been much more difficult to ascertain. The
remainder of this chapter will focus on the structural and functional aspects of this
receptor.
p75NTR
The pan neurotrophin receptor
Early NGF binding studies established that NGF-responsive cells had high
affinity and low affinity binding sites, with dissociation constants of ~10"n and ~10"9 M
respectively. p75NTR was the first discovered NGF receptor (Chao et al., 1986; Johnson
et al., 1986; Radeke et al., 1987), and transfection studies established that p75NTR bound
NGF at the lower of these two affinities, thereby earning the name "low affinity NGF
receptor". The discovery of the other neurotrophins led to the finding that p75NTR could
bind all the neurotrophins with approximately equal affinity in most cells (RodriguezTebar et al., 1990; Rodriguez-Tebar et al., 1992; Squinto et al., 1991). While the
equilibrium binding constants are very similar, binding of each of the neurotrophins to
p75NTR display markedly different kinetics, suggesting that the binding of individual
neurotrophins to p75NTR could serve particular functions in the nervous system (Barrett,
2000).
Although p75NTR was the first identified neurotrophin receptor, its physiological
function remains poorly understood. Many studies indicate that the effects of p75NTR
range from promotion of survival pathways, induction of apoptosis, modulation of cell
cycle and differentiation, and facilitation or inhibition of neuritic growth. p75NTR can,
under certain conditions, act as a co-receptor to Trk, thereby enhancing or dampening its
response to neurotrophins (reviewed by Kaplan and Miller, 2000; Roux and Barker,
2002). p75NTR has also recently been shown to act as a co-receptor for the Nogo
receptor, thereby modulating neurite growth inhibition in response to the myelin
inhibitory proteins, MAG, OMgP and Nogo (reviewed by Kaplan and Miller, 2003;
McKerracher and Winton, 2002). However, p75NTR can also signal autonomously,
through ill-defined pathways that regulate growth, survival and death.
p75NTR is structurally similar to other TNFR superfamily members
p75NTR was the first identified member of the TNF receptor superfamily that
now contains approximately 25 receptors, including Tumor Necrosis Factor Receptor 1
(TNFR1) and TNFR2, Fas, RANK and CD40 (Baker and Reddy, 1998). All members of
the TNFR superfamily share a common structural cysteine-rich module in their
extracellular domain, and do not have any catalytic activity, but sometimes contain a
death domain (DD) which can interact with adaptor proteins that link TNFRs to caspase
activation. p75NTR is an atypical member of this family for a number of reasons. Firstly
p75NTR binds dimeric ligands (the neurotrophins) that are structurally distinct from
other TNFR ligands, which are trimeric (Banfield et al., 2001; Robertson et al., 2001;
Wiesmann et al, 1999). Secondly, the p75NTR DD has been shown to be significantly
differently from the Fas TNFR in the orientation of the first of its six alpha-helices
(Huang et al., 1996; Liepinsh et al., 1997). Finally, p75NTR has not been shown to bind
the same adaptor proteins that link other pro-apoptotic TNFRs to Caspase-8 activation
(reviewed in Wallach et al., 1996).
The p75NTR protein is a single Type I transmembrane protein that contains a
single asparagine-linked carbohydrate close to its N-terminal and several O-linked
carbohydrates in the juxtamembrane domain. The p75NTR extracellular domain (ECD)
has four cysteine-rich domains (CRDs) (Baldwin et al., 1992; Yan and Chao, 1991), and
several studies indicate that CRD2 to 4 are important for neurotrophin binding (Baldwin
et al., 1992; Chapman and Kuntz, 1995; Shamovsky et al., 1999; Yan and Chao, 1991).
The first CRD in other TNFRs including Fas, TNFR1, and CD40 has been shown to
mediate ligand-independent receptor trimerization (Chan et al., 2000; Siegel et al., 2000),
but it is unclear whether CRD1 of p75NTR functions in an analagous manner. The
p75NTR intracellular domain (ICD) contains a palmitoylation site on Cysteine 279
(Barker et al., 1994), which may serve to properly distribute p75NTR throughout the cell
membrane, and can be phosphorylated on several serine and threonine residues (Grob et
al., 1985; Taniuchi et al., 1986). The function of these phosphorylation events has not
been well understood, but one recent study suggests that phosphorylation at Serine 304
(within the juxtamembrane linker region of p75NTR) by Protein Kinase A (PKA) is a
critical requirement for recruiting p75NTR to lipid rafts, which contain a concentrated
pool of downstream signaling molecules that can promote signal transduction (Higuchi et
al., 2003).
The p75NTR juxtamembrane domain is well conserved between species (Large et
al., 1989) and its DD has been shown to be important for some of the protein-protein
interactions that are required to transduce its effects. The p75NTR DD contains a ~80
amino-acid association module initially identified in related pro-apoptotic TNFRs, but
unlike those TNFRs, the p75NTR DD does not associate with the same adaptor proteins
that result in direct activation of caspases (Wallach et al., 1996). As stated above, the
structure of the p75NTR DD is significantly different from that of the Fas receptor
(Liepinsh et al., 1997), which may help to explain why signaling mediated by p75NTR
differs from that of Fas and other related TNFRs.
Other p75NTR ligands
Neutrophins bind p75NTR with dissociation constants of ~10"9 M, but a recent
study indicates that the NGF precursor, pro-NGF, binds p75NTR with much higher
affinity than mature NGF, indicating that pro-neurotrophins may be the preferred ligands
for p75NTR (Lee et al., 2001). Pro-NGF binding to p75NTR in this study induced
significantly higher levels of apoptosis in comparison with the mature, fully processed
NGF. These findings may be physiologically significant since neurotrophin precursors
are the predominant forms of BNDF and NGF in the brain, and have been shown to be
elevated in Alzheimer's disease (Fahnestock et al., 2001). Another recent study has
shown that pro-NGF is likely responsible for activating p75NTR-dependent apoptosis in
vivo during spinal cord injury, suggesting that the role of the proneurotrophin is to
eliminate damaged cells by activating p75NTR apoptotic cascades after injury (Beattie et
al., 2002).
p75NTR has also been shown to bind the invertebrate snail L. stagnalis ligand
cysteine-rich neurotrophic factor (CRNF) with nanomolar affinity (Fainzilber et al.,
1996), raising the possibility that CRNF is a member of an as-yet-identified family of
p75NTR ligands. In addition, p75NTR has been shown to bind non-neurotrophin
ligands, including the A(3-peptide of the amyloid precursor protein (APP) (Kuner et al.,
1998; Perini et al., 2002; Yaar, 1997) and the rabies virus, suggesting that p75NTR may
facilitate the entry of neurotoxic substances into cells.
p75NTR Functions
p75NTR is reported to mediate strikingly diverse biological effects which include
cell death, survival, neurite outgrowth, positive regulation of developmental myelination
(Cosgaya et al., 2002), Schwann cell migration (Anton et al., 1994; Bentley and Lee,
2000), modulation of synaptic transmission and functional regulation of sensory neurons
and calcium channels (Stucky and Koltzenburg, 1997). Some of these effects are
mediated in concert with other receptors, while others are elicited through p75NTR on its
own. I will focus here on the actions of p75NTR in combination with Trk or Nogo
receptors, and then focus on p75NTR autonomous signaling that results in survival and
apoptosis.
p75NTR as a co-receptor for Trk: mediation of Trk-dependent signals
Neurotrophins bind their receptors with two different affinities: low-affinity (Ka =
1.7 x 10"9M) and high affinity {KA= 2.3 x 10" M). p75NTR displays the lower affinity
when binding each neurotrophin, but in concert with the appropriate Trk, binds with the
higher affinity (Hempstead et al., 1991; Mahadeo et al., 1994; Rodriguez-Tebar et al.,
1992). Whether p75NTRis required for high affinity binding, or whether Trk alone is
sufficient has been somewhat controversial, but it appears that the presence of both are
important since co-expression of p75NTR increases the rate of NGF association with
TrkA by 25-fold (Mahadeo et al., 1994).
In response to low concentrations of neurotrophins, Trk activation is enhanced by
the co-expression of p75NTR (Hantzopoulos et al., 1994; Verdi et al., 1994) indicating
that p75NTR is important for increased survival responses in low neurotrophin
concentrations. p75NTR co-expression with TrkA may therefore be physiologically
important in cases where developing neurons must bind neurotrophins that are secreted
from target organs at subpicomolar concentrations (Barde, 1989). p75NTR has also been
shown to enhance TrkB and TrkC responsiveness at low concentrations of BDNF and
NT-3, respectively (Hantzopoulos et al., 1994; Ip et al., 1993).
How does p75NTR mediate the increased Trk responsiveness to NGF? Though
this question remains unclear, experiments from the Yancopoulos group (Hantzopoulos et
al., 1994) show that a mutation within the p75NTR ECD leads to a deficiency in
formation of high-affinity neurotrophin binding sites. Further, it has shown that the
10
transmembrane and ICD of p75NTR are required for generation of high-affinity
neurotrophin binding sites (Esposito et al., 2001). Additional experiments are needed to
address the precise mechanism by which p75NTR facilitates Trk activation.
p75NTR can also dampen Trk-mediated survival signals, and this has been shown
in several studies of cells co-expressing p75NTR and Trk, where the non-preferred
ligands are used to activate Trk. In the presence of p75NTR, NGF readily activates
TrkA, but activation by NT-3 and NT-4/5 is greatly reduced (Bibel et al., 1999; Ip et al.,
1993; Lee et al., 1994; Mischel et al., 2001). Similarly, BDNF-mediated TrkB activation
is not altered in the presence of p75NTR, but TrkB activation by NT-3 and NT-4/5 is
reduced by p75NTR co-expression (Bibel et al., 1999). The precise mechanism by which
p75NTR reduces Trk activation by non-preferred ligands is also uncertain, but may
involve p75NTR-dependent phosphorylation on Trk intracellular domains (Roux and
Barker, 2002).
p75NTR as a co-receptor for NogoR: mediation of neurite growth inhibition
It is well established that while axons in the peripheral nervous system (PNS) can
regenerate after injury, those in the central nervous system (CNS) do not (McKerracher
and Winton, 2002). In the mature nervous system, a diverse class of myelin-derived
neuritogenic inhibitors are largely responsible for the failure of injured CNS neurites to
regrow. Recent work has shown that the myelin-inhibitory molecules, which include
Nogo, myelin-associated glycoprotein (MAG), and oligodendrocyte myelin-associated
glycoprotein (OMgP), all of which bind the Nogo66 Receptor (NgR) (Domeniconi et al.,
2002; Fournier et al., 2001; Liu et al., 2002; Wang et al., 2002), require p75NTR to
transduce their growth inhibitory effects (Wang et al., 2002; Wong et al., 2002).
The precise mechanisms by which p75NTR transduces the growth inhibitory
11
effects of the myelin inhibitory proteins are still unclear, but appear to converge on
modulation of RhoA activity (Yamashita et al., 1999). RhoA is a small GTPase that
regulates the state of actin polymerization depending on its GTP-bound state: in its active
GTP-bound form, RhoA rigidities the actin cytoskeleton, thereby inhibiting growth cone
collapse (Davies, 2000; Schmidt and Hall, 2002). Early last year, it was shown that
RhoA was modulated in a p75NTR-dependent manner, in response to treatment with the
myelin inhibitory protein, MAG (Yamashita et al., 2002), although MAG does not bind
directly to p75NTR. A number of subsequent studies showed that the GPI-linked Nogo
receptor, NgR, also bound directly to MAG and OMgp with similar affinities
(Domeniconi et al., 2002; Liu et al., 2002). Finally, two separate groups showed that
p75NTR formed a complex with the NgR in response to treatment with MAG and Nogo,
which in turn stimulated RhoA activity (Wang et al., 2002; Wong et al., 2002).
There continues to be controversy over the manner in which RhoA is modulated
by p75NTR in this model. Previous work using a yeast two-hybrid system had shown
that a region within the p75NTR DD bound directly to RhoA (Yamashita et al., 1999).
However, another study published early this year has established that the Rho-GDP
dissociation inhibitor, Rho-GDI, directly interacts with p75NTR (Yamashita and
Tohyama, 2003). Rho-GDI inhibits nucleotide dissociation in addition to shuttling Rho
proteins between the cytoplasm and membrane, and can only bind Rho in its GDP-bound,
inactive state (Sasaki and Takai, 1998). The study published earlier this year (Yamashita
and Tohyama, 2003) showed that Rho-GDI bound directly to p75NTR, and that this
interaction was strengthened in the presence of MAG or Nogo. Strengthened interaction
between Rho-GDI and p75NTR resulted in displacement of GDP-bound RhoA from
Rho-GDI, therein allowing RhoA to become activated through exchange of GDP for
GTP. Activated RhoA has been shown to inhibit neuritic outgrowth through pathways
that involve suppression of Rho kinase (ROCK) and that ultimately act on the actin
cytoskeleton (McKerracher and Winton, 2002; Woolf and Bloechlinger, 2002).
12
In addition to its ability to inhibit neurite outgrowth through a NgR interaction,
p75NTR has also been shown to modulate RhoA activity on its own. This work has
shown that neurotrophin binding to p75NTR can inactivate RhoA, in turn stimulating
neurite outgrowth. The same group that established the direct interaction between RhoGDI and p75NTR also showed that, whereas MAG and Nogo stimulated this interaction,
no effect was observed upon neurotrophin stimulation (Yamashita and Tohyama, 2003).
Their data suggest that Rho-GDI constitutively sequesters GDP-bound, inactive RhoA in
the presence of neurotrophin, thereby stimulating neuritic outgrowth. This idea may be
physiologically relevant during development where p75NTR could inactivate RhoA to
favour axonal or dendritic outgrowth in areas directly surrounding the target organ
secreting neurotrophin. In support of this hypothesis, it has been shown that p75NTR
expression is upregulated in sympathetic neurons following arrival at the target organ
(Miller et al., 1994).
p75NTR autonomous signaling: activation of apoptotic and survival pathways
p75NTR is perhaps best known for its ability to induce cell death. Several
findings indicate that NGF binding to p75NTR can initiate an apoptotic pathway in
neuronal cell types that do not express TrkA. For example, NGF treatment of embryonic
retinal cells (Frade et al., 1996) or postnatal oligodendrocytes (Casaccia-Bonnefil et al.,
1996) that express p75NTR, but not TrkA, causes apoptosis. The ligand dependency for
p75NTR-dependent apoptosis remains controversial, since in some studies, ligand
binding was essential for cell death and in others, it was not (reviewed in Roux and
Barker, 2002). Nonetheless, the importance of p75NTR in developmental apoptosis is
underscored by several in vivo studies. Application of antibodies directed against either
NGF or the p75NTR ECD reduces apoptosis in the developing avian retina (Frade et al.,
13
1996). Reduction of p75NTR using antisense oligonucleotides on axotomized sensory
neurons also resulted in decreased levels of apoptosis (Cheema et al., 1996). In addition
p75NTR expression levels are increased after insult to the nervous system, and have been
tightly correlated with the induction of apoptosis (Bagum et al., 2001; Casha et al., 2001;
Dowling et al., 1999; Kokaia et al., 1998; Martinez-Murillo et al., 1998; Oh et al., 2000;
Roux et al., 1999; Syroid et al., 2000; Wang et al., 2000).
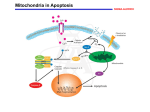
Activation of cellular apoptosis occurs through two main pathways. The first
such pathway is typified by the oligomerization of cell-surface pro-apoptotic TNFRs. In
this scheme, ligand activation of the death receptor induces assembly of a death-inducing
signaling complex (DISC) in which adaptor proteins TRADD or FADD bind directly to
the receptor's death domain (DD), thereby inducing aggregation and activation of
caspase-8, with subsequent activation of effector caspases (Strasser et al., 2000). In the
alternative "mitochondrial" pathway, pro-apoptotic BH3-domain-only proteins of the
Bcl-2 family accumulate at the mitochondria and induce pro-apoptotic Bcl-2 proteins to
reduce mitochondrial integrity and cause release of cytochrome C, IAF and Smac/Diablo
into the cytoplasm (Anderson and Tolkovsky, 1999; Gross et al., 1999; Harris and
Johnson, 2001). Cytochrome C and Smac/Diablo facilitate Caspase-9 activation and
thereby initiate the caspase cascade. The two pathways are not mutually exclusive, as
Caspase-8 activation can lead to cleavage of Bid, a BH3-domain-only protein, which in
turn leads to activation of the mitochondrial death pathway (Zha et al., 2000).
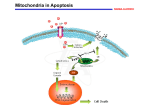
There is evidence that p75NTR-dependent apoptosis occurs via the mitochondrial
apoptotic pathway. It has been shown that Caspase-9, but not Caspase-8, is induced
during p75NTR-dependent apoptosis (Gu et al., 1999; Wang et al., 2001). Further, we
14
have also shown that p75NTR-dependent apoptosis results in mitochondrial cytochrome
C release (Bhakar et al., 2003, submitted).
Numerous studies of neuronal apoptosis, using models of trophic factor
deprivation, suggest that p75NTR-mediated death follows a mitochondrial pathway. In
NGF-deprived sympathetic neurons, loss of Trk-mediated survival signals promotes the
activation of molecules that result in loss of mitochondrial integrity, activation of
caspase-9 and cell death. Early studies from Eugene Johnson's group demonstrated that
apoptosis induced by NGF deprivation required both transcription and translation (Martin
et al., 1988). More recently, it has been shown that the c-Jun amino-terminal kinase
(JNK) cascade and c-Jun-mediated transcription play a crucial role in mitochondrial
cytochrome C release, caspase-9 activation and apoptosis in NGF-deprived sympathetic
neurons (Bruckner et al., 2001; Deshmukh and Johnson, 1998; Deshmukh et al., 1996;
Eilers et al., 2001; Harding et al., 2001; Martinou et al., 1999; Putcha et al., 1999). The
upstream signaling leading to JNK activation after neurotrophin withdrawal appear to
involve the small GTPases Cdc42 and Racl (Bazenet et al., 1998), which when activated,
bind the CRIB motif of MAPKKKs such as GCK, MEKK1-4 and MLK2-3 (Fan et al.,
1996; Hirai et al., 1996; Rana et al., 1996; Sakuma et al., 1997; Tibbies et al., 1996).
Activated MAPKKKs then phosphorylate and activate MKK4/7 (Xu et al., 2001),
MAPKK's that phosphorylate and activate specific JNK isoforms (Bruckner et al., 2001;
Sakuma et al., 1997). Once phosphorylated by activated JNK, c-Jun binds the promoters
of genes with appropriate AP-1 elements and induces transcription. The precise targets
of c-Jun necessary for induction of apoptosis have been the subject of intense interest,
and recently, Bim and Hrk/Dp5, both BH3-domain-only proteins, have been identified as
15
pro-apoptotic genes induced by c-Jun in both sympathetic neurons deprived of NGF and
in cerebellar granule neurons deprived of KC1 (Harris and Johnson, 2001; Putcha et al.,
2001; Whitfield et al., 2001).
There is reason to believe that p75NTR is involved in activation of the apoptotic
signaling induced by neurotrophin withdrawal. Reduced expression of p75NTR in
sensory neurons (Barrett and Bartlett, 1994) or in differentiated PC12 cells (Barrett and
Georgiou, 1996) is correlated with reduced apoptosis upon neurotrophin deprivation,
whereas increased p75NTR expression levels accelerates death following NGF
withdrawal (Barrett, 2000). Recently, it has also been shown that p75NTR facilitates
apoptosis in neonatal sympathetic neurons derived from TrkA-/-mice (Majdan et al.,
2001), suggesting that p75NTR signals autonomously during neurotrophin withdrawalmediated death.
There are several similarities that occur during p75NTR-mediated and trophicfactor-mediated apoptosis. Rac is induced in oligodendrocytes undergoing p75NTRmediated death and dominant-negative Rac blocks this death (Harrington et al., 2002).
p75NTR-mediated apoptosis correlates with activation of JNK or c-Jun in
oligodendrocytes (Casaccia-Bonnefil et al., 1996; Yoon et al., 1998), sympathetic
neurons (Aloyz et al., 1998; Bamji et al., 1998), hippocampal neurons (Friedman, 2000)
and PC 12 cells (Roux et al., 2001). Dominant-negative JNK or chemical inhibitors of
JNK can block this p75NTR-induced death (Friedman, 2000; Harrington et al., 2002).
And finally, as stated above, p75NTR activation results in mitochondrial cytochrome C
release and selective activation of Caspase-9 (Bhakar, et al., 2003, submitted; Gu et al.,
1999; Wang etal., 2001).
16
Recent work done in the Barker laboratory has examined the signaling pathways
activated by the p75NTR interactor, NRAGE (Salehi et al., 2002). This data shows that
NRAGE induces apoptosis through a mitochondrial death pathway involving cytochrome
C release and activation of Caspase-9, -3 and -7. Further, NRAGE-induced apoptosis
correlated with an MLK-independent activation JNK and its downstream target, c-Jun.
Blockers of JNK activity or of c-Jun mediated transcription strongly inhibited NRAGEmediated caspase activation and apoptosis. NRAGE is the first reported p75NTRinteracting protein to induce cell death through the activation of JNK. It remains to be
defined, however, what links p75NTR- and NRAGE- dependent c-Jun-mediated
transcription to mitochondrial cytochrome C release.
While p75NTR is well known to initiate cell death pathways, in some cases, it has
been shown to promote cell survival. Neurotrophin binding to p75NTR has been
reported to promote survival of developing neocortical subplate neurons (DeFreitas et al.,
2001), and NGF binding to p75NTR has been shown to promote survival of a
Schwannoma cell line (Gentry et al., 2000), sensory neurons (Hamanoue et al., 1999),
human breast cancer cells (Descamps et al., 2001) and cultured Schwann cells
(Khursigara et al., 2001). In addition, a mutant NGF which bound only p75NTR and not
Trk A has been shown to inhibit apoptosis in serum-deprived PC 12 cells (Hughes et al.,
2001). The mechanisms by which p75NTR-dependent survival occurs remain poorly
understood, but several studies indicate that p75NTR increases cell survival through PI3kinase and Akt activation (DeFreitas et al., 2001; Descamps et al., 2001; Roux et al.,
2001).
17
The p75NTR knockouts
In mice, the p75NTR gene contains six exons that span 18 kilobases of
Chromosome 11 (Radeke et al., 1987). The gene encodes a 3.4 kb mRNA with a 5'
untranslated region of about 300 nucleotides, and a ~2000 nucleotide 3' untranslated
region that contains a single consensus polyadenylation signal (Johnson et al., 1986).
The third exon, encoding CRDs 2 to 4 (the region responsible for neurotrophin binding),
and the fourth exon, encoding the stalk region of the extracellular domain and the
transmembrane domain, have both been targeted for deletion to create mice that are
deficient for p75NTR expression (Lee et al., 1992; von Schack et al., 2001)(see also
Figure 2). These mice have been useful for determining aspects of p75NTR function in
vivo.
p75NTRExonI11 -/- mice
In 1992, Lee and colleagues (Lee et al., 1992) constructed a p75NTR knockout
mouse in which the third exon of the p75NTR locus was targeted for deletion
(p75NTRExonIU -/-). In order to generate these mice, a replacement type targeting vector
consisting of a pGK-Neo cassette (a hybrid gene consisting of the phosphoglycerate
kinase I promoter driving the neomycin phosphotransferase gene) was inserted into Exon
III, and recombined such that the targeted p75NTR gene contained the pGK-Neo cassette
in the same direction as the p75NTR gene, and a deleted portion of Exon III. The mutant
animals displayed a complex phenotype with deficits in cutaneous innervation and heat
sensitivity, that lead to development of ulcers in the distal extremities. Early studies
showed that neonatal sympathetic and embryonic sensory neurons derived from these
18
animals had reduced sensitivity to NGF and deficits in developmental and injury-induced
apoptosis (Davies et al., 1993; Lee et al., 1994). Despite the neuronal phenotype, recent
studies indicate that p75NTRExonI"-/- mice are not fully deficient for p75NTR expression
(Dechant and Barde, 1997; von Schack et al., 2001). These studies show that the
p75NTR locus produces an alternatively spliced isoform of p75NTR (s-p75NTR) that
lacks Exon III.
Alternative splicing of Exon III occurs in both wild-type and
p75NTRExonln-/- mice, and produces a protein product that lacks the portion of the
extracellular domain responsible for neurotrophin binding.
Given this recent data, interpretations from studies with the p75NTRExonl"-/- mice
should be viewed with caution, in light of the potential signaling capabilities of sp75NTR. Alternatively, there is little data showing that s-p75NTR signals in either wildtype or p75NTRExonI11-/- mice, and the expression of s-p75NTR in a number of neuronal
and non-neuronal tissues remains the subject of intense controversy. Only one group has
shown the presence of s-p75NTR, and its presence has only been detected in whole brain
lysates and in Schwann cell extracts (Naumann et al., 2002; von Schack et al., 2001).
However, a multitude of studies have described neuronal and non-neuronal deficits in
p75NTRExonI11-/- mice, suggesting that at the very least, neurotrophins binding to p75NTR,
or CRDs 2-4, are a critical requirement for its function. I will summarize the data
pertaining to p75NTR's interaction with TrkA and NogoR, and its apoptotic potential in
the p75NTRExon1"-/- mice in the following paragraphs.
One function ascribed to p75NTR is enhancement of NGF binding to TrkA, and
several in vitro studies have shown that co-expression of p75NTR with TrkA produces
high-affinity NGF binding sites and enhanced TrkA activation (Roux and Barker, 2002).
This enhancement of TrkA activation is also observed at low concentrations of NGF
(Hantzopoulos et al., 1994; Verdi et al., 1994). This function is purported to be important
during development, when subsaturating levels of neurotrophin are required to support
19
survival of neurons are released from target organs. In keeping with this hypothesis,
p75NTRExonI11-/- mice have deficits in sensory and sympathetic innervation (Lee et al.,
1994; Lee et al., 1992) that correlate with reduced neurotrophin responsiveness to Trk.
However, other data indicates that TrkA activation is not substantially altered in
sympathetic neurons from either wild-type of p75NTRExonI11-/- mice (Majdan et al, 2001).
Since these sympathetic neurons did not appear to express s-p75NTR, the conclusion
from these experiments is that p75NTR is not required for optimal TrkA activation.
Thus, given the disparity between these results, it is still unclear whether p75NTR is
required in vivo to create high-affinity NGF-TrkA binding, and concomitant TrkA
activation.
Recent reports have shown that p75NTR mediates the growth inhibitory effects of
the myelin inhibitory proteins MAG and Nogo through an interaction with the Nogo
receptor. Many of these studies have used the p75NTRExonI0-/- mice to show that they do
not respond in the same manner as wild-type mice to the myelin inhibitors (Wang et al.,
2002; Yamashita et al., 2002; Yamashita and Tohyama, 2003). In the wild-type mice,
neurite growth from dissociated postnatal dorsal root ganglia (DRGs) and cerebellar
neurons (CGNs) was inhibited upon addition of myelin inhibitors. In contrast, DRGs and
CGNs from p75NTRExonI11-/- mice showed no growth inhibition, indicating that FLp75NTR is required for MAG-induced growth inhibition. If these neurons express sp75NTR, the results from these studies also show that CRDs 2-4 are required for the
interaction between NgR and p75NTR that mediates the growth inhibition. Would
pretreatment with neurotrophins (which also bind p75NTR at CRDs2-4) ablate the
interaction with NgR, and subsequent growth inhibition in wild-type mice? Wang et al
performed experiments in which high concentrations of NGF and BDNF was added to
wild-type DRGs prior to addition of myelin inhibitors (Wang et al., 2002), but there was
no effect on growth inhibition in these circumstances. In vitro co-immunoprecipitations
of s-p75NTR and NgR may help to resolve this outstanding issue.
20
Nonetheless, the fact that p75NTRexonI"-/- mice showed aberrant neurite outgrowth
in response to the myelin inhibitors was in keeping with several groups' findings showing
that they also exhibit perturbed developmental axon growth (Lee et al., 1994; Walsh et
al., 1999a; Walsh et al., 1999b). One notable study using two lines of transgenic mice
overexpressing NGF in CNS astrocytes in the presence and absence of the p75NTRexon111
mutation observed enhanced sympathetic axon growth in the myelinated portions of the
cerebellum where NGF was highly expressed only in the p75NTRexonI11 mice (Walsh et al.,
1999a). The reasons for this aberrant outgrowth were not immediately apparent, though
the authors proposed that p75NTR may directly activate intracellular signaling pathways
leading to outgrowth. In support of this hypothesis, Riopelle's group had published
seemingly incongruous results demonstrating that injection of a peptide identical to a
portion of the p75NTR cytoplasmic domain into embryonic sensory neurons enhanced
neurite outgrowth (Dostaler et al., 1996). This peptide was identical to the amphiphilic
peptide, mastoparan, a component of wasp venom that was known to activate guanine
nucleotide-binding regulatory proteins, including Rho (Koch et al., 1991). This study
implicated a role for a highly conserved motif of p75NTR in neurite growth through Rho,
and a subsequent yeast-two hybrid study showed that p75NTR interacted with RhoA
(Yamashita et al., 1999), though recent data indicates that this is not a direct interaction,
as described above (Yamashita and Tohyama, 2003).
In addition to their aberrant neuritic growth, p75NTRExonI11-/- mice also display
reduced developmental and injury-induced apoptosis. Mice lacking ngf alleles or
bearing the p75NTRExonin deletion have reduced apoptosis in the developmental retina and
spinal cord, as assessed by TUNEL staining (Frade and Barde, 1999), indicating that a
significant proportion of the early cell death observed in these tissues are mediated by
NGF acting through p75NTR.
p75NTRExonin -/- mice also have supernumerary
sympathetic neurons during the neonatal period (Bamji et al., 1998), indicating that
21
p75NTR mediates cell death in vivo in these developing neurons. Further, it has been
shown that p75NTRExonin deletion leads to small but statistically significant increases in
the number of basal forebrain cholinergic neurons (Naumann et al., 2002), though the
effect is highly dependent on different mice strains, which appear to differentially express
s-p75NTR. p75NTR expression levels increase after nervous system injury in a manner
that is correlated with increased apoptosis (Roux and Barker, 2002), and p75NTRExonI11 -/mice have been found to have reduced apoptosis following injury in many experimental
models (Beattie et al., 2002; Ferri and Bisby, 1999; Ferri et al., 1998).
To summarize, p75NTRExonI"-/- mice are not fully deficient for p75NTR
expression, though they exhibit a number of aberrant characteristics with regards to
neurite outgrowth and developmental and injury-induced apoptosis. Determining the
temporal and spatial expression pattern of s-p75NTR in these mice remains a crucial
requirement for not only assigning a function to s-p75NTR but also for interpreting
results obtained using p75NTRExon111-/- mice. A mouse that is fully deficient for p75NTR
is a absolute prerequisite for understanding the functions of this receptor.
22
p75NTRExonIV -/- mice
To produce animals that were fully deficient for p75NTR expression, von Schack
and colleagues targeted the fourth exon of the p75NTR locus to generate p75NTRExonIV-/mice (von Schack et al., 2001). Disruption of the reading frame was achieved by
insertion of the pGK-Neo cassette in the reverse orientation to the p75NTR locus in an
AatH restriction site at the 5' end of Exon IV. These animals produce neither full-length
p75NTR (FL-p75NTR) nor s-p75NTR and exhibited more severe nervous system defects
than p75NTRExon1"-/- mice. These defects included a larger reduction in the number of
DRG neurons and Schwann cells compared with p75NTRExonI11-/- mice, though no
aberrant phenotype was described for the sympathetic nervous system. Targeting of the
p75NTR locus in Exon IV lead to reduced body size and hind limb ataxia (due to
decreased sensory innervation). About 40% of p75NTRExonIV-/- mice do not survive
beyond the perinatal period, and this was attributed to severe vascular defects, including
aortic wall ruptures. This was the first report suggesting that p75NTR was important for
the development of vascular tissues.
The same group that created the p75NTRExonIV-/- mice went on to study the
number of basal forebrain cholinergic neurons in comparison with p75NTRExonI11-/- mice.
This study found that the p75NTRExonIH mice, which could express s-p75NTR, lead to a
moderate increase in the number these neurons, while the p75NTRExonIV mutation, which
prevents expression of both FL p75NTR and s-p75NTR, resulted in a more substantial
increase in basal forebrain cholinergic neurons compared to wild-type mice (Naumann et
al., 2002).
The inference was that FL-p75NTR expression normally mediates
developmental apoptosis in this neuronal population and that loss of FL-p75NTR results
23
in less cell death with subsequent increases in the number of these neurons. This study
also suggested that s-p75NTR could partially compensate for loss of FL-p75NTR,
although the authors never showed that s-p75NTR was expressed in the basal forebrain
cholinergic neurons.
Statements of projects/hypothesis
I pursued two distinct projects in my M.Sc. research. Firstly, I examined the
transcriptional activation of genes downstream of c-Jun during p75NTR-dependent
apoptosis. Two pieces of evidence suggested that p75NTR-mediated death results in
transcriptional activation of BH3-domain-only genes. Firstly, the transcription factor cJun is activated during p75NTR-dependent cell death in many cell types (Aloyz et al.,
1998; Bamji et al., 1998; Casaccia-Bonnefil et al., 1996; Friedman, 2000; Roux et al.,
2001; Yoon et al., 1998) and secondly, it has been shown that c-Jun can mediate
transcription of BH3-domain-only protein Bim and Hrk in neurons undergoing apoptosis
induced by trophic factor deprivation (Harris and Johnson, 2001; Putcha et al., 2001;
Whitfield et al, 2001). Given these data, I hypothesized that distinct BH3-domain-only
genes were transcriptionally activated during p75NTR-mediated apoptosis. Using a gainof-function approach in which adenovirus expressing p75NTR was infected into different
neuronal cell types to constitutively activate p75NTR pathways, my specific aim was to
determine by RT-PCR the specific BH3-domain-only genes transcribed as a function of
p75NTR-dependent JNK activation.
My second project was to examine the recently created p75NTRExonIV-/- mice for
full deletion of all p75NTR gene products. It had previously been shown that targeting
24
Exon III of p75NTR for deletion resulted in a mouse that continued to express a p75NTR
gene product, through endogenous splicing of Exonlll (von Schack et al., 2001). In order
to verify that the p75NTRExonlv-/- mice were fully deficient for p75NTR expression, I
used immunoblotting and RT-PCR techniques to analyse whether portions of the
extracellular or intracellular domains of p75NTR were maintained in these newly created
p75NTRExonlv-/- mice.
25
Preface to Chapter 2
A multitude of in vitro and in vivo studies support a role for p75NTR in both
developmental and injury-induced apoptosis, though the signals involved in p75NTRmediated cell death are uncertain. p75NTR-dependent apoptosis is associated with an
increase in Rac and JNK activity and recent work from our laboratory has shown that the
p75NTR interactor, NRAGE, activates a mitochondrial death pathway involving JNKdependent cytochrome C release and activation of Caspase-9, Caspase-7 and Caspase-3.
Despite this progress, several important details of p75NTR apoptotic signaling remain
unknown. In particular, the targets of p75NTR-dependent JNK activation that result in
mitochondrial cytochrome C release remain uncertain. BH3-domain-only proteins are
members of the Bcl-2 family that induce the association of Bax and Bak which in turn
facilitate release of Cytochrome c into the cytosol. Transcriptional activation of BH3domain-only proteins through c-Jun or p53 dependent pathways mediates Cytochrome C
release in some neuronal models of apoptosis. In this chapter, I examined whether
p75NTR-induced apoptosis was correlated with accumulation of BH3-domain-only gene
products. U373 cells and cortical neurons were infected with LacZ or p75NTR
adenovirus and alterations in mRNA levels of BH3-domain-only family members Bim,
Bmf, Hrk, Bik, Puma, and Noxa were examined by RT-PCR. The results from these
experiments showed that p75NTR-dependent apoptosis did not result in BH3-domainonly gene transcription. Further studies in our laboratory have subsequently established
that the BH3-domain-only protein, Bad, is phosphorylated on Serine 128 in a JNKdependent manner, and that this phosphorylation is a critical component of p75NTRdependent apoptosis.
26
Chapter 2: p75NTR does not transcriptionally activate BH3-only proteins
27
Literature Review: the BH3-domain-only proteins
Studies over the last six years have identified the BH3-domain-only proteins as
key initiators of apoptosis (Huang and Strasser, 2000). These members of the Bcl-2
protein family are crucial regulators of caspase activation, which function by inducing the
release of cytochrome C and Smac/Diablo from the mitochondria, an event that preceeds
Caspase-9 activation. Bcl-2 family members are defined functionally as either proapoptotic or anti-apoptotic based on their ability to kill or protect cells in vivo or in
culture (Strasser et al., 2000). The anti-apoptotic Bcl-2 proteins include mammalian Bcl2, Bcl-w, Bcl-XL and CED-9, the Bcl-2 homolog in C. Elegans, and generally contain
four Bcl-2 Homology domains, BH1-4. Proapoptotic Bcl-2 proteins can be divided into
two groups: the multidomain Bcl-2 proteins and the BH3-domain-only proteins. The
multidomain pro-apoptotic proteins include mammalian Bax, Bak and Bod, and contain
BH1-3. To date, eight mammalian BH3-domain-only proteins have been identified,
mainly through protein interaction assays with Bcl-2 or Bcl-XL, and include Bik/NBK,
Bad, Bid, Hrk/Dp5, Bim, Bmf, PUMA, NOXA/bbc3 and EGL-1 in C. Elegans. Each
contain the third BH domain and most contain a mitochondrial targeting motif near the Cterminus . The amphipathic a-helix formed by the BH3 domain of both the BH3domain-only and the "Bax-like" multidomain Bcl-2 proteins can bind to an elongated
cleft formed by BH domains 1-3 of the anti-apoptotic Bcl-2 proteins. It is this interaction
between the BH3-domain-only proteins with anti-apoptotic Bcl-2 proteins that sequesters
these latter molecules, and then allows pro-apoptotic Bcl-2 proteins to multimerize and
facilitate cytochrome C release (Nicholson and Thornberry, 1997).
The pro-apoptotic activity of the BH3-domain-only proteins is regulated by both
28
transcriptional and post-translational mechanisms that work to prevent inappropriate cell
death.
Transcriptional control-Some of the BH3-domain-only proteins have been shown
to be trancriptionally upregulated during apoptosis.
A subset of these include
PUMA/bbc3 (Yu et al., 2001) and NOXA (Oda et al, 2000), which have been identified
as target genes for p53, a transcription factor that mediates apoptosis in response to
stimuli including DNA damage, and which has also been reported to be necessary for
p75NTR-dependent apoptosis in sympathetic neurons (Aloyz et al., 1998). The mRNA
for these genes has been shown to be upregulated when exposed to death stimuli known
to induce p53, and both genes have potential p53-recognition sequences within the
promoter region (Huang and Strasser, 2000).
Other BH3-domain-only proteins have been shown to be transcriptionally
upregulated specifically during neuronal apoptosis. For instance, Hrk/Dp5 is upregulated
in rat sympathetic neurons deprived of NGF (Imaizumi et al., 1997), and in rat cortical
neurons exposed to amyloid |3-protein (Imaizumi et al., 1999).
Bim is also
trancriptionally upregulated during neuronal apoptosis. Rodent sympathetic neurons
deprived of NGF (Eilers et al., 2001; Putcha et al., 2001) and cerebellar granule neurons
deprived of KC1 (Harris and Johnson, 2001) all display increased Bim mRNA and protein
levels. This process involves c-Jun and its upstream regulatory pathway, JNK, which can
act to induce transcription of Bim and Hrk/Dp5 genes in neurons undergoing apoptosis as
a result of growth factor deprivation (Harris and Johnson, 2001; Whitfield et al, 2001).
The regulation of BH3-domain-only proteins during neuronal apoptosis induced
by trophic factor deprivation appears complex, especially in light of a number of several
29
recent studies (Harris and Johnson, 2001; Putcha et al., 2001; Whitfield et al., 2001). As
stated, the BH3-domain-only proteins Bim and Hrk are transcriptionally upregulated in
neurons undergoing trophic factor deprivation and this event is partially mediated by the
JNK pathway. A handful of recent studies show that JNK may also regulate BH3-onlyproteins Bim, Bad and Bmf by phosphorylation during neuronal apoptosis (Biswas and
Greene, 2002; Donovan et al., 2002; Lei et al., 2002; Putcha et al., 2003), suggesting that
c-Jun may be only one of a number of JNK targets. Since p75NTR-mediated apoptosis
correlates with a number of the same molecules as those activated during trophic factor
deprivation (discussed in Chapter 1), it is possible that p75NTR regulates the BH3domain-only proteins in much the same way as that described above.
Post-translational control-Some BH3-domain-only proteins are maintained in an
inactive form, but become activated following apoptotic stimuli. For example, Bad is
maintained in a hyperphosphorylated form in healthy cells, where it is sequestered by the
scaffold protein, 14-3-3 (Zha et al., 1996). Prosurvival stimuli such as neurotrophins and
growth factors trigger phosphorylation of Bad at Serines 112 and 126, thereby inducing
sequestration of Bad with 14-3-3 (Blume-Jensen et al., 1998; Datta et al., 1997; del Peso
et al., 1997; Zha et al., 1997). However, upon apoptotic stimuli, Bad is phosphorylated at
an additional site, Serine 128, a crucial step in its dissociation from 14-3-3 and
subsequent translocation to the mitochondria (Konishi et al., 2002).
Both Bim and Bmf are controlled post-translationally. Both of these BH3-only
proteins are normally associated with the cytoskeleton, Bim interacting with the LC8
cytoplasmic dynein light chain (Puthalakath et al., 1999) and Bmf interacting with
myosin V (Puthalakath et al., 2001). Upon different apoptotic stimuli, these proteins
30
dissociate from their cytoskeletal partner and translocate to the mitochondria. Recent
work also shows that phosphorylation of Bim and Bmf potentiates their apoptotic ability
(Biswas and Greene, 2002; Lei and Davis, 2003; Putcha et al., 2003). Finally Bid, which
normally resides in the cytosol, is activated by Caspase-8 cleavage in a Fas-mediated
pathway (Li et al., 1998; Luo et al., 1998; Wang et al., 1996). This cleavage generates a
15kDa C-terminal fragment, termed tBid, which becomes N-myristolated and can then
translocate to the mitochondria (Zha et al, 2000). This latter pathway may amplify rather
than initiate the caspase cascade.
Statements of projects/hypothesis
Given the similarities between apoptotic signals induced by trophic factor
deprivation in neurons and p75NTR activation (discussed in Chapter 1), it is possible that
BH3-domain-only proteins are transcriptionally activated through the JNK pathway
during p75NTR-mediated apoptosis. I hypothesize that p75NTR apoptotic signaling
leads to increases in mRNA and proteins levels of specific BH3-domain only proteins.
Further, I hypothesize that transcription of these BH3-domain-only proteins requires JNK
activity and c-Jun mediated transcription. In order to test these hypotheses, an adenoviral
system was used in which p75NTR was reproducibly introduced into a variety of
heterologous cells to constitutively activate p75NTR apoptotic signaling pathways.
31
Materials and Methods
Cell culture and infection. The human glioma (U373) cell line was provided by Dr. R.
Del Maestro and maintained in 5% C0 2 at 37° C in either Dulbecco's modified Eagle's
medium (DMEM) or RPMI medium and supplemented with 10% fetal calf serum (FCS,
Clontech), 2 mM L-glutamine, 100 jig/ml penicillin/streptomycin.
The rat
pheochromocytoma cell line, PC 12, was maintained as previously described (Roux et al.,
2001) and the PC12rtTa cell line (PC 12) was purchased from Clontech and maintained in
10% C0 2 at 37° C in DMEM supplemented with 10% FCS, 5% horse serum, 2 mM Lglutamine, 100 pg/ml penicillin/streptomycin and 100 pg/ml G418. Cell lines were
plated 18 to 24 hours prior to infection and harvested 24 hours after infection. Primary
cortical cultures were prepared from E14-16 CD1 mouse telencephalon as described
previously (Bhakar et al., 2002). Neuronal cultures were infected prior to plating and
then maintained in vitro for 24 hours in Neurobasal media (Life Technologies)
supplemented with IX B27 supplement (Life Technologies), 2mM L-glutamine, and 100
u.g/ml penicillin/streptomycin.
Semiquantitative R T-PCR. 450,000 U373 cells or primary cortical neurons were infected
with virus and 24 hours later mRNA was isolated using the RNEasy Mini kit according to
the manufacturer's instructions (Qiagen). cDNA was generated using the Omniscript RT
kit (Qiagen) and random hexamers (Roche) as primers. PCR was performed for 30 cycles
using 300 nM of the following primers for U373 cells: (3-actin sense, 5'CACCACTTTCTACAATGAGC; antisense, 5'03Cn?CAC<jATCITCATGAGG; hBIMEL sense,
5'TGGCAAACCAAa^ITCTGATG; antisense, 5'AGTCGTAAGATAA(XATTU3TGGG;
32
hBMF sense, 5'(HTC<nxnOGCTGACCTGTTTG; antisense,
5'AAGCa}ATAGCCAGCATTGC; hHrk/Dp5 sense, 5'TCGGCAC<jCC<3AACrTGTAG;
antisense, 5'GCKJTATGTAAATAGCATTGGGGTG; hBIK sense,
5'AACCCCGAGATAGTGCTGGAAC; antisense, 5'GCTGGAAACCAACATTTTATTGAGC;
hPUMA sense, 5'ACTGTGAATCCTGTGCIXTGCC; antisense,
5'ACCCCCCAAATGAATGCCAG; hNOXA sense,
5'CCAAACTCTTCTGCTCAGGAACC; antisense, 5'COJrAATCITCGGCAAAAACAC
For mouse cortical neurons, PCR was performed using the same conditions as above
using the following primers for certain human sequences: mBimEL sense,
5'CCCCTACCTCCCTACAGACAGAA; antisense, 5'CCAGACGGAAGATAAAGCGTAACAG;
mBMF antisense, 5'GTTGCGTATGAAGCCGATGG; mHrk/Dp5 sense,
5'TGGAAACACAGACAGAGGAAGCC; antisense, 5'AAAGGAAAGGGACCACCACG;
mBIK sense, 5'TCACCAACCTCAGGGAAAACATC; antisense,
5'AGCAGGGGTCAAGAGAAGAAGG; mNOXA sense,
5'TGATGTGATGAGAGAAA(DGCTCG; antisense, 5'AAAC<:AATCCCAAACGACTGCC;
p75NTR sense, 5'TGAATTCTGGAACAGCTGCAAAC; antisense,
5'CCTTAAGT<^CACTGGGGATGTG; NRAGE sense,
5'GCACCCCCTAATGTGClXTrcA(XTAATC^
antisense,
5'GAACKnGCAGTCCAGTC. For PCR analysis, 5% of the cDNA prepared was used in a
25ul PCR reaction; all of the reaction was separated on a 10% polyacrylamide gel, and
PCR products were visualized under UV light.
33
Results
Transcriptional activation of BH3-domain-only genes through JNK- and p53dependent pathways is involved in apoptosis in neuronal and non-neuronal models and
we therefore tested whether p75NTR-induced apoptosis was correlated with
accumulation of BH3-domain-only gene products.
To induce p75NTR-dependent
apoptosis, a gain-of-function approach was used in which recombinant adenovirus
expressing p75NTR was introduced into a variety of cell lines. Primary mouse cortical
neurons, Pheochromocytoma PC 12 cells and U343 and U373 glioma cells all showed
reductions in viability as assessed by MTT survival assay when infected with p75NTR
adenovirus, whereas infection with control adenovirus expressing (3-galactosidase (LacZ)
had no significant effect (Figure I, Appendix).
Cortical neurons and U373 cells were infected with either p75NTR or control
adenovirus (LacZ or GFP) and alterations in mRNA levels of the BH3-domain-only
family members Bim, Bmf, Hrk, Bik, Puma, and Noxa were determined by RT-PCR.
mRNA corresponding to each of these family members were readily detected in both
U373 cells (Figure 1A) and in cortical neurons (Figure IB). However, p75NTRdependent alterations in mRNA levels of the BH3-domain-only family were not detected
(Figure 1A & B). These results indicated that p75NTR apoptotic signaling occurs in the
absence of BH3-domain-only transcriptional activation, and suggests that alternate
pathways lead to p75NTR-induced mitochondrial cytochrome C release and caspase
activation (Figure 2, Appendix).
34
Closing Remarks
BH3-domain-only proteins inhibit the action of anti-apoptotic Bcl-2 family
members such as Bcl-2 and Bcl-XL and facilitate the release of cytochrome C, a
prerequisite for caspase activation in the mitochondrial apoptotic cascade. The regulation
of these proteins is now recognized as a crucial step in the apoptotic pathway. In
sympathetic neurons and cerebellar granule neurons, JNK activation results in
phosphorylation of c-Jun which in turn results in transcription of the BH3-domain only
family members Bim and Hrk (Harris and Johnson, 2001; Putcha et al., 2001; Whitfield
et al., 2001), whereas in other systems, p53 activation results in transcription of BH3domain-only family members, Noxa and Puma (Oda et al., 2000; Yu et al., 2001). It was
hypothesized that p75NTR-dependent apoptosis, which results in JNK activation,
involved transcriptional regulation of the BH3-domain only family members. However,
although p75NTR induced JNK-dependent apoptosis in the cell types analyzed, we found
no evidence for transcriptional regulation of BH3-domain only family members.
Some BH3-domain-only proteins are contitutively expressed, but are inactive due
to sequestration by other proteins. For example, the BH3-domain-only protein Bad binds
to scaffold protein, 14-3-3, and Bim and Bmf are sequestered to components of the
cytoskeleton (Puthalakath et al., 1999; Puthalakath et al., 2001; Zha et al., 1996).
Antiapoptotic stimuli, such as growth factors and neurotrophins can render constitutively
expressed BH3-domain-only family members inactive by promoting these protein-protein
interactions (Datta et al., 1997; del Peso et al., 1997). Recent findings have also
demonstrated that the sequestration of Bim, Hrk and Bad can be negatively regulated by
JNK (Biswas and Greene, 2002; Donovan et al., 2002; Lei and Davis, 2003; Putcha et al.,
35
2003). JNK-dependent phosphorylation of Bad on Serine 128 allows it to dissociate from
14-3-3 and contribute to apoptosis (Donovan et al., 2002). Similarly, UV irradiation of
HEK293 cells results in JNK-dependent phosphorylation of Bmf and Bim releasing these
proteins from their cytoskeletal binding partners and allowing them to exert their function
at the mitochondria (Lei and Davis, 2003).
While BH3-domain-only proteins do not appear to be subject to transcriptional
regulation during apoptosis induced by p75NTR, subsequent work in our laboratory
established that p75NTR-mediated death correlated with the phosphorylation and
accumulation of a higher molecular weight species of the BH3-domain-only protein, Bad
(Figure 8, Appendix). This product was cross-reactive with Bad antibodies raised against
two distinct epitopes as well as the anti-pSerine 128 Bad and therefore likely represents
phospho-Serinel28-Bad within a stable oligomeric complex. Further work established
that JNK activity contributes to this p75NTR-dependent Bad phosphorylation, since
infection of PC 12 cells with adenovirus expressing both p75NTR and a dominantnegative JNK contruct (encoding the JNK-binding domain of JIP) prevented formation of
the Bad oligomeric complex (Figure 8, Appendix). Finally, PC 12 cells transfected with a
dominant negative Bad Serine 128 construct that was previously described (Konishi et
al., 2002), and then infected with p75NTR, were significantly protected from p75NTRinduced apoptosis (Figure 9, Appendix),
indicating that the JNK-Bad pathway is
important for p75NTR-induced death.
Our data show that p75NTR induces JNK-dependent phosphorylation and
oligomerization of Bad. Furthermore, loss-of-function experiments using dominant
negatives and RNA interference demonstrated a role for Bad in p75NTR-induced
36
apoptosis. These findings link cell surface receptor activation to the post-translational
regulation of BH3-domain-only family members and indicate how p75NTR-dependent
JNK activation can contribute to apoptosis.
37
Figure Legends
Figure 1. p75NTR does not transcriptionally regulate BH3-domain-only proteins.
A. Cortical neurons were infected with 0, 50, or 200 MOI of adenovirus encoding figalactosidase (LacZ) or p75NTR (p75) and 24 hours later mRNA was isolated as
described in Materials and Methods. cDNA was generated and then PCR was performed
using primers directed against Bim, Bmf, Hrk, Bik, Puma, Noxa, p75NTR and Actin as
indicated. The multiple bands in the Bim RT-PCR reflect different isoforms, BimEL,
BimL and Bims.
B. Human glioma U373 cells were infected with 0 or 100 MOI of adenovirus encoding
Green Fluorescent Protein (GFP) or p75NTR (p75) and analysed as described above.
p75NTR expression in these experiments was verified by immunoblot (data not shown).
38
LacZ
p75
B
GFP p75
0 100 100
0 50 200 50 200
Bim
Bmf
W^^^W • ^ • ' V
(PtawP
M*V
Hrk
kW Uw i,J LJ
Bik
**w kari Iw4 * w
Puma
Puma
Noxa
^;..|r^ ^ J
p75NTR
Actin
Figure 1
39
Preface to Chapter 3
p75NTR functions include enhancing Trk receptor signaling, interacting with the
Nogo receptor to mediate neuritic growth inhibition, and initiating autonomous signaling
cascades that regulate growth, survival and apoptosis. The generation of animals that
lack p75NTR expression has been a critical advance in understanding the in vivo role of
this receptor. Recent studies have indicated that disruption of Exon IV produces a null
mouse lacking all p75NTR gene products (p75NTRExonlv-/-) whereas mice containing a
disruption of Exon III (p75NTRExonI11-/-) show no full-length p75NTR expression yet
maintain expression of an alternatively spliced form of p75NTR (s-p75NTR)(von Schack
etal.,2001).
In this chapter, our initial characterization of the p75NTRExonIV-/- mice revealed
that they express a p75NTR gene product that encodes a truncated protein containing the
extracellular stalk region together with the entire transmembrane and intracellular
domains. The gene product is initiated from a cryptic Kozak consensus/initiator ATG
sequence within a region of Exon IV located 3' to the pGK-Neo insertion site, likely as a
result of enhancer elements within the inserted pGK-Neo cassette. Characterization of
the protein product indicated that it was localized to the plasma membrane.
Overexpression of this fragment in heterologous cells resulted in activation of JNK and
cleavage of Procaspase 3, indicating that it can mediate pro-apoptotic effects in vivo.
These results indicate that aspects of the p75NTRExonIV/"phenotype may reflect a gain-offunction
mutation
rather
than
a
loss
of
p75NTR
function.
40
Chapter 3: A pro-apoptotic fragment of the p75 neurotrophin receptor is expressed
in P75NTRExonJV null mice
41
A pro-apoptotic fragment of the pir5 neurotrophin receptor is
expressed in p75NTRExonIV null mice
Christine E. Paul and Philip A. Barker
Centre for Neuronal Survival, Montreal Neurological Institute, McGill University, 3801
University Ave, Montreal, Quebec, H3A 2B4
Running title: Aberrantp75NTR expression inp75NTRExmUV-/-mice
Number of text pages: 16
Number of words:
Abstract:
131
Introduction: 464
Discussion: 421
Total characters: 25,626
Number of figures: 3
Correspondence:
Philip A. Barker
Montreal Neurological Institute
McGill University
3801 University Ave
Montreal, Quebec, Canada, H3A 2B4
Phone:(514)-398-3064
Fax: (514)-398-5214
Email: [email protected]
42
Acknowledgements
We gratefully acknowledge the technical assistance of Genevieve Dorval,
Kathleen Dickson and Asha Barker in colony maintenance, genotyping, tissue dissection,
and subcloning, and thank Wayne Sossin for helpful comments on the manuscript. The
p75NTRExonlv-/- mice were a kind gift of Georg Dechant (University of Innsbruck). This
work was supported by grants from the Canadian Institutes of Health Research (PAB).
CEP is supported by a Natural Sciences and Engineering Research Council scholarship
and PAB is a Scientist of the Canadian Institutes of Health Research.
43
Abstract
The p75 neurotrophin receptor (p75NTR) binds members of the neurotrophin
family and regulates neuronal survival, apoptosis and growth during development and
after nervous system injury. Recent studies have indicated that disruption of p75NTR
Exon IV in mice results in a p75NTR null strain gene whereas mice lacking Exon III are
hypomorphs that express a p75NTR splice variant. We report that p75NTRExonlv -/- mice
produce a p75NTR gene product that encodes a truncated protein containing the
extracellular stalk region together with the entire transmembrane and intracellular
domains. Expression of this fragment activates pro-apoptotic signaling cascades that
result in cleavage of Procaspase 3, indicating that it can mediate pro-apoptotic effects in
vivo. These results indicate that aspects of the p75NTRExonIV -/- phenotype may reflect a
gain-of-function mutation rather than a loss of p75NTR function.
44
Introduction
p75NTR is a receptor for the neurotrophins, a family of growth factors that
includes nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF),
neurotrophin-3 (NT-3) and neurotrophin-4/5 (NT-4/5). Along with the Trk tyrosine
kinase receptors, p75NTR mediates the diverse functions of the neurotrophins. The Trk
receptors show neurotrophin binding specificity and are generally associated with
regulation of neuronal growth, differentiation and survival.
p75NTR binds all
neurotrophins with approximately equal affinity and its functions include enhancing Trk
receptor signaling and initiating autonomous signaling cascades that regulate growth,
survival and apoptosis (Barrett, 2000; Roux and Barker, 2002). Recent work also
indicates that p75NTR is a component of a receptor complex that mediates growth
inhibition in response to myelin inhibitory proteins, MAG and Nogo (Kaplan and Miller,
2003; McKerracher and Winton, 2002).
To address the in vivo role of p75NTR, Lee and colleagues (Lee et al., 1992)
constructed a p75NTR knockout mouse in which Exon III of the p75NTR locus was
targeted for deletion (p75NTRExonI11-/-). This mouse exhibited profound deficits in the
peripheral nervous system, and neonatal sympathetic and embryonic sensory neurons
derived from these animals showed reduced sensitivity to NGF and deficits in
developmental and injury-induced apoptosis (Davies et al., 1993; Lee et al., 1994).
However, recent studies indicate that the p75NTR locus produces an alternatively spliced
isoform of p75NTR (s-p75NTR) that lacks Exon III (von Schack et al., 2001). This
transcript, which is present in both wild type and p75NTRExonin-/- mice, produces a
protein product that lacks the portion of the extracellular domain responsible for
neurotrophin binding. To produce animals deficient in p75NTR expression, von Schack
and colleagues targeted the fourth exon of the p75NTR gene to generate p75NTRExonIV-/-
45
mice (von Schack et al., 2001). These animals produce neither full-length p75NTR nor sp75NTR and exhibit severe nervous system and vascular system defects. About 40% of
p75NTRExonlv-/- mice do not survive beyond the perinatal period.
Here we report that the p75NTRExonIV-/- mice express a p75NTR gene product.
This fragment has an apparent molecular weight of 26 kDa and is not observed in
wildtype animals, shows moderate expression in heterozygotes and is most highly
expressed in p75NTRExonIV-/- animals. RT-PCR analysis revealed that this protein
expression pattern correlates with the aberrant expression of a p75NTR mRNA that is
initiated 3' to the inserted pGK-neo cassette. The resulting protein encodes a portion of
the extracellular stalk region, and the entire transmembrane and intracellular domains.
This fragment is likely produced through utilization of an enhancer sequence within the
pGK-Neo cassette inserted into Exon IV. When overexpressed, the p75NTRExon IV gene
product activates p75NTR signaling cascades that result in JNK phosphorylation and
Procaspase 3 cleavage. The signaling properties of this p75NTR fragment suggest that
some of the phenotypes observed within the p75NTRExonIV-/- mouse may be due to gainof-function rather than loss-of-function mutations.
46
Materials and Methods
Materials-Cell culture reagents were purchased from BioWhittaker unless otherwise
indicated. The JNK antibody (C-17, cat #sc-474) was purchased from Santa Cruz
Biotechnology and the Bcl-2 antibody (cat #6620) was purchased from BD Biosciences.
Cleaved caspase-3 (Aspl75; cat #9661), phospho-Thrl83/Tyrl85 JNK (G9, cat #9255),
phospho-Thr69/71 ATF-2 (cat #9225S), ATF-2 (cat #9222), and IKB (cat #9242) specific
antibodies were purchased from Cell Signaling Technology. All other reagents were
from Sigma or ICN Biochemicals unless otherwise indicated.
Animal colonies and genotyping-The p75NTRExonI" mutation was generated as described
previously (Lee et al., 1992) and mice bearing this mutation were obtained from the
Jackson Laboratories and interbred on a C57BL/6 background via heterozygous matings.
Genotyping for the p75NTRExonin mice was done on tail genomic DNA by PCR with
primers within Intron II (p75-lntll, 5'-(XATGCrCCTATGGCTACTA), Intron III
(p75IntIII, 5'-(XTCCCATTCC<JCGTCAGCC) and the pGK-Neo cassette inserted in Exon
III (pGK, 5'-GGGAAClTCCTGACTAGGGG). PCR parameters were 94°C for 150 s; 94°C
followed by 30 cycles at for 1 min, 60°C for 1 min, and 72°C for 1 min. The p75NTRExonIV
mutation was generated as previously described (von Schack et al., 2001) and mice
bearing this mutation were obtained from the lab of Georg Dechant (University of
Innsbruck) and maintained on a C57BL/6 background. Genotyping for the p75NTRExonIV
mice was performed on tail genomic DNA by PCR using primers directed to Exon IV
and to the inserted neo cassette as described (von Schack et al., 2001).
47
Tissue processing and immunoblotting- PI brain, cerebellum and heart tissues were lysed
in Nonidet P-40 lysis buffer (lOmM Tris, pH 8.0, 150 mM NaCl, 10% glycerol, 1%
Nonidet P-40, 1 protease inhibitor cocktail tablet (Roche, Cat No 1836153) dissolved in
10 ml buffer), subjected to 30 seconds of high-speed homogenization, followed by
centrifugation at 15,000 rpm for 15 min at 4°C and removal of pelleted material. Protein
concentrations were determined using the BCA assay kit from Pierce. 25-50 ixg of
protein was solubilized in Laemmli sample buffer, separated by SDS-polyacrylamide gel
electrophoresis, and electroblotted onto nitrocellulose. p75NTR immunoreactivity was
detected using a rabbit polyclonal antibody (p75NTR-Pl) raised against amino acids 276425 of rat p75NTR-ICD, as previously described (Barker and Shooter, 1994) or an
antibody directed against human p75NTR ICD (Promega cat #G3231). Blocking and
antibody incubations of immunoblots were performed in Tris-buffered saline/Tween
(lOmM Tris, pH 7.4, 150 mM NaCl and 0.2% Tween) supplemented with 5% (w/v) dried
skim milk powder, except for those involving phospho-specific antibodies which were
performed in Tris-buffered saline/Tween supplemented with 2% (w/v) bovine serum
albumin.
Horseradish peroxidase conjugated
donkey anti-IgGs
(Jackson
Immunoresearch Laboratories, Inc) were used at a dilution of 1:5000. Immunoreactive
bands were detected using enhanced chemiluminescence (PerkinElmer Life Sciences)
according to the manufacturer's instructions.
Reverse transcription-PCR analysis- mRNA was isolated from PI brain using the Qiagen
RNEasy Mini kit according to the manufacturer's instructions. cDNA was generated
using the Qiagen Omniscript RT kit and random hexamers (Roche) as primers. PCR was
48
performed for 30 cycles (94°C for 1 min, 58°C for 1 min, 72°C for 1 min) using 300 nM
(final
concentration)
of
the
following
primers:
(3-actin
sense,
5'
CA(XACITTCTACAATGAGC; (3-actin antisense, 5'-03GTCAC<}ATCTTCATGAGG;
p75NTR-ICD sense, 5'-CCAGCAGACCCACACACAGACTG; p75NTR-ICD antisense, 5'CCCTACACAGAGATCOCGGTTC;
p75NTR-Exon
IV
(791-813)
sense,
5'-
CACCACCTCCAGAGCGAGACCTCATAG; p75NTR ExonVI (1467-1448) antisense, 5'GAACATCAGCGGTCGGAATG. PCR was performed using primers directed against Exon
I and Exon III for a total of 30 cycles (94°C for 1 min, 4 cycles of 68 °C for 1 min, 8
cycles of 68 °C for 1 min ramping down by 1 °C each cycle, 18 cycles of 60 °C for 1 min,
extension for each cycle was 72°C for 1 min) using the following primers:
p75NTR-Exon I sense, 5'-ACOiCCATCGGTCCGCAG; p75NTR Exon III antisense, 5'TCATCTGAGTATGTGOXTCKJG.
20 LII of the reaction products were separated on a 1%
agarose gel, and PCR products were visualized under UV light.
Construction of mammalian expression vector - To clone the p75NTRExonIV construct into
a mammalian expression vector, an RT-PCR reaction was prepared using primers
directed against mouse p75NTR Exon IV-IV (bp 791-1467 within mouse p75NTR cDNA
(gi 23468246)). The PCR reaction product was gel purified using a Qiaquick Gel
Extraction kit, ligated into pENTR/D-TOPO vector (Invitrogen) and sequenced (Bio
S&T, Montreal, QC). The cDNA was then ligated into the GATEWAY pDEST12.2
mammalian expression vector in a recombination reaction that made use of the attLl and
attL2 sites within both the pENTR/D-TOPO and the pDEST12.2 vectors according to the
manufacturer's instructions (Invitrogen).
49
Cell culture and transfection - HEK293T cells and COS-7 cells were maintained in 5%
C0 2 at 37°C in Dulbecco's modified Eagle's medium containing 10% bovine serum
(BCS), 2 mM L-glutamine and 100 ug/ml penicillin/streptomycin.
The rat
pheochromocytoma cell line PC 12 was maintained in 10% C0 2 in Dulbecco's modified
Eagle's medium with 5% BCS, 5% horse serum, 2 mM L-glutamine and 100 ug/ml
penicillin/streptomycin. 350,000 PC 12 or 293T cells plated onto a 6-well plate were
transfected using LipofectAMINE 2000 (Invitrogen) according to the manufacturer's
instructions and were subsequently lysed in Laemmli sample buffer and analyzed by
SDS-PAGE and immunoblotting.
Subcellular fractionation - All manipulations were performed at 4°C. PI brain tissues
were lysed in homogenization buffer (20 mM Hepes, pH 7.4, 1 mM EDTA, 255 mM
sucrose, 1 protease inhibitor cocktail tablet (Roche, Cat No 1836153)) and then
centrifuged at 600g for 5 min. The supernatant was centrifuged at 10,000g and the pellet
obtained was designated heavy membrane. The supernatant was centrifuged at 100,000g
and the pellet was designated light membrane and the resulting supernatant, designated
cytosol.
Immunocytochemistry- 25,000 COS-7 cells were plated onto coverslips in wells of a 24well plate, transfected as above, then fixed for 30 minutes in 2% PFA in PBS
supplemented with 10 mM MgCl2. Coverslips were incubated in 2% donkey and 0.2%
Triton X-100 in PBS for 60 min, followed by overnight incubation at 4°C with polyclonal
antibodies directed against p75NTR (p75NTR-Pl, 1:1000) and with monoclonal
50
antibodies directed against nucleolin (1:1000). Primary antibodies were diluted in PBS
containing 2% donkey serum. Cells were then rinsed three times with PBS, followed by
incubation for 60 min at room temperature in Cy3-labeled anti-rabbit IgG (1:1000;
Jackson Laboratories), FITC-labeled anti-mouse IgG (1:1000; Jackson Laboratories) and
Hoescht 33258 (1:10,000; Molecular Probes). Stained cells were analyzed on a Zeiss 510
confocal microscope.
51
Results
A fragment ofp75NTR is expressed in p75NTRExon,v-l- mice
Expression analysis of p75NTR in mice bearing mutations within Exon III or
Exon IV of the p75NTR locus was performed.
As expected, PI brain lysates of
p75NTRExonI11-/- mice contained no full-length p75NTR (Figure 2A, lane 3) and PI brain
lysates from p75NTRExonIV/-mice also lacked full-length p75NTR (Figure 2A, lane 6).
Surprisingly, we found that mice bearing either one or two copies of the p75NTRExonIV
mutation contained a 26 kDa protein that was immunoreactive with two distinct
antibodies directed against the p75NTR intracellular domain (Figure 2A, lanes 5 and 6).
This 26 kDa band was not present in wild-type mice, nor was it detected in mice with
p75NTRExonUI mutations (Figure 2A, lanes 1-4). The genotype-specific expression profile
of the 26 kDa band was also observed in lysates derived from cerebellum and heart of PI
p75NTRExonIV wild-type, heterozygous and null littermates (Figure 2B, lanes 2,3 and
lanes 5,6). In heart lysates from wild-type mice, full-length p75NTR was present at low
levels and the 26 kDa species was not detected. However, the 26 kDa protein was
relatively abundant in heterozygote and null mice in the same litter. To determine if the
increase in the 26kDa protein reflected an increase in transcription of p75NTR, RT-PCR
was performed on PI brain cDNAs derived from p75NTRExonIV wild-type, heterozygous
and null littermates, using primers directed to ExonV and ExonVI of the p75NTR locus
(which encode the portion of the intracellular domain indicated in Figure 2C). This
fragment of the p75NTR mRNA was transcribed at higher levels in mice bearing the
p75NTRExonIV mutant allele (Figure 2D) than in wild-type littermates whereas levels of
fj-actin mRNA were maintained across all genotypes. Collectively, these data indicated
52
that a truncated p75NTR gene product is produced as both mRNA and protein in
p75NTRExonIV-/- mice.
Transcription
of the transmembrane
and intracellular
domains of p75NTR in
p75NTRExonlv -I- mice is the result ofpGK-Neo cassette insertion
To generate the p75NTRExonIV-/- mice, von Schack and colleagues inserted a pGKNeo cassette into an Aatll restriction site within Exon IV of mouse p75NTR (von Schack
et al., 2001). Several studies indicate that the retention of this selectable marker (a hybrid
gene consisting of the phosphoglycerate kinase I promoter driving the neomycin
phosphotransferase gene) in targeted loci of "knockout" mice can cause unexpected
phenotypes due to effects on flanking genes (Olson et al., 1996; Pham et al., 1996) and
we reasoned that pGK-Neo may lead to aberrant expression of 3' gene products in the
p75NTR locus. To determine whether the inserted pGK-Neo cassette affected the
transcription of p75NTR in p75NTRExonIV-/- mice, we designed primers to amplify cDNA
produced upstream and downstream of the pGK-Neo cassette placed within the p75NTR
gene (shown schematically in Figure 3A), and performed RT-PCR on PI brain mRNA
from p75NTRExonIV wildtype, heterozygous and null littermates. Figure 3A shows that
levels of p75NTR mRNA derived from a portion of the p75NTR locus located 5' to the
pGK-Neo insertion site were significantly reduced in brains from PI mice bearing the
p75NTRExonIV mutation, likely due to mRNA instability due to absence of a polyA tail on
the 3' end of mRNAs driven off the endogenous p75NTR promoter. In contrast, levels of
p75NTR mRNA derived from a portion of the gene located 3' to the pGK-Neo insertion
site was significantly enhanced in these same brains, suggesting that enhancer sequences
53
within the pGK-Neo cassette are responsible for transcription of 3' fragments of the
p75NTR gene in p75NTRExonIV-/- mice.
We next tried to identify the fragment of p75NTR expressed in the p75NTRExonIV
-/- mice. Direct cloning by RACE-PCR was unsuccessful due to the presence of a strong
reverse transcription stop within the cDNA that invariably produced truncated products
(data not shown). We therefore scanned the sequence 3' to the pGK-neo insertion site to
identify in-frame ATG codons that were flanked by potential cryptic Kozak consensus
sequences. One candidate (nt 846-848) was identified as the ATG codon closest to the
pGK-Neo insertion site (nt 760), and RT-PCR was used to determine if cDNA containing
this putative start site could be directly cloned from PI brains of p75NTRExonIV-/- mice.
Figure 3B shows the sequence of the 676 bp cDNA containing nucleotides 791-1467 that
was cloned from the p75NTRExonIV-/- mice. The predicted protein encoded by this
fragment will produce a 187 amino-acid fragment of p75NTR containing a small portion
of the extracellular stalk together with the entire transmembrane and intracellular
domains of the receptor (shown schematically in Figure 3C). To confirm that this
fragment could mediate protein expression, the p75NTRExonIV-/- cDNA was subcloned
into a mammalian expression vector and transfected into HEK293 cells. Figure 3D shows
that the p75NTRExonIV-/- cDNA directed abundant expression of a p75NTR fragment that
perfectly co-migrates with the protein product identified within p75NTRExonIV-/- brain
lysates.
54
Recombinant p75NTRExonlvprotein expression induces Caspase 3 cleavage
The p75NTR fragment produced in the p75NTRExonIV-/- mouse lacks the signal
sequence necessary for membrane insertion, but retains the hydrophobic transmembrane
domain. To determine if the p75NTRExonIV product is membrane-associated, PI brains
from p75NTRExon
1V
heterozygote and null littermates were subjected to subcellular
fractionation. Figure 4A shows that the 26 kDa p75NTRExon
IV
protein is lacking in
cytosolic fractions, but is enriched in heavy and light membrane fractions, similar to the
distribution of full-length p75NTR. To further characterize the subcellular localization of
the p75NTRExon
IV
product, COS-7 cells were transiently transfected with the
p75NTRExonIV-/- cDNA and immunocytochemistry was performed using antibodies
directed against p75NTR and nucleolin, used a a nuclear marker. Figure 4B shows that
p75NTRExon
lv
protein is excluded from the nucleus but is detected at the plasma
membrane and within the cytosol, where it is presumably associated with intracellular
membranes.
Together these results indicate that the p75NTRExonIV fragment is a
membrane-associated protein.
We have previously shown that the p75NTR intracellular domain can initiate
p75NTR signaling events when produced as a truncated protein (Majdan et al., 1997;
Roux et al., 2001) and therefore we tested the possibility that the p75NTRExonIV protein
can similarly activate p75NTR signaling cascades. Figure 4C shows that expression of
the p75NTRExonIV protein in PC 12 cells induced Procaspase-3 cleavage that was reduced
by co-expression of Bcl-2, indicating that p75NTRExonIV protein can induce activation of
the intrinsic death pathway. Similar results were obtained in 293T cells that do not
express endogenous p75NTR (data not shown), indicating that p75NTRExonIV protein can
elicit these signals regardless of the presence of full-length p75NTR. Several reports
show that p75NTR apoptotic signals involves activation of the c-Jun kinase pathway
(Friedman, 2000; Harrington et al., 2002) and Figure 4D shows that expression of the
p75NTRExonIV protein in PC 12 cells induced JNK phosphorylation as well as
55
phosphorylation of ATF-2, a well characterized JNK target (Eilers et al., 2001; van Dam
et al., 1995). The phosphorylation of these molecules was increased somewhat by coexpression of Bcl-2, likely due to Bcl-2-dependent suppression of caspase-dependent
death in transfected cells.
Together, these data show that overexpression of the
p75NTRExonlv protein is capable of activating p75NTR signaling cascades.
56
Discussion
Our results show that p75NTRExonlv-/- mice express a fragment of the p75NTR
protein that contains a portion of the extracellular domain and the complete
transmembrane and intracellular domains. The genotype-specific expression profile of
this fragment is consistent with the hypothesis that the pGK-Neo cassette inserted into
Exon IV of the p75NTR gene is responsible for this expression pattern. When expressed
in heterologous cells, this p75NTR fragment is membrane-associated where it may
activate p75NTR signaling cascades that include JNK activation and Procaspase-3
cleavage.
The PGK-1 promoter and enhancer sequence functions in an orientation and a
position-independent manner (McBurney et al., 1991) and several groups have
demonstrated that retention of the pGK-Neo cassette in targeted loci can profoundly
influence the expression of surrounding genes upstream and downstream from the
cassette (Olson et al., 1996; Pham et al., 1996). Our results suggest that the pGK-Neo
cassette placed within Exon IV of p75NTR results in aberrant transcription of the
adjacent 3' portion of the gene which, due to the presence of a cryptic Kozak consensus
sequence in Exon IV, results in production of the p75NTRExon IV protein product.
Initial characterization of the p75NTRExonIV-/- mice revealed severe peripheral
neuron loss as well as dilation and ruptures within large blood vessel (von Schack et al.,
2001). We have demonstrated that the p75NTRExon IV protein was highly expressed in
heart lysates of PI p75NTRExonIV/"mice and have shown that the p75NTRExon IV protein
product can activate p75NTR signaling cascades that result in Procaspase-3 cleavage.
Our previous studies have shown that expression of the p75NTR intracellular domain
57
within neurons of transgenic mice can induce apoptosis of peripheral and central neurons
in vivo (Majdan et al., 1997) and together, these findings raise the possibility that some
characteristics of the p75NTRExon IV mouse are due to gain-of-function mutations rather
than loss of p75NTR function. At a minimum, the presence of the p75NTRExonlv protein
product in these animals indicate that the cellular basis for observed phenotypes should
be interpreted with caution.
Although p75NTR was the founding member of the TNF receptor superfamily
and was the first identified neurotrophin receptor, its physiological function has not been
completely elucidated. Recent findings indicate roles that range from promotion of
survival pathways, induction of apoptosis, effects on cell cycle and differentiation, and
facilitation or inhibition of growth (Barrett, 2000; Kaplan and Miller, 2003; McKerracher
and Winton, 2002; Roux and Barker, 2002). Generation of strains of mice that are
rendered null for p75NTR gene products remains an important requirement for the
elucidation of its biological activities.
58
Figure Legends
Figure 2. A portion of p75NTR is expressed and transcribed in p75NTRExonIV' mice.
A, PI brain lysates from p75NTRExonI" and p75NTRExonIV wild-type (+/+), heterozygous
(+/-) and null (-/-) littermates were separated on a 15% SDS-PAGE gel, followed by
immunoblotting with two antibodies specific to the p75NTR-ICD (see Materials and
Methods).
Arrowheads indicate full-length p75NTR and a ~26 kDa band present in
lysates from mice bearing the p75NTRExonIV mutant allele. Blots were reprobed for (3IIItubulin to verify equal loading. B, PI cerebellar and heart lysates from p75NTRExonIV
wild-type (+/+), heterozygous (+/-) and null (-/-) littermates were prepared and subjected
to SDS-PAGE and immunoblotting as described above. C, Schematic diagram showing
the genomic structure of the mouse p75NTR locus and corresponding protein domains.
D, RNA was isolated from PI brain lysates of p75NTRExonIV wild-type (+/+),
heterozygous (+/-) and null (-/-) littermates. cDNA was prepared in the presence and
absence of reverse transcriptase (+ and - RT), followed by RT-PCR using primers for
(3-actin and for p75NTR-ICD. Arrows in Panel C indicate location of primers used for
this analysis. Experiments in Panels A, B and D were repeated 3 times with identical
results.
59
Figure 3. Transcription of p75NTR fragment in p75NTRExonIV mice is the result of
pGK-Neo cassette insertion into the p75NTRExonIV targeting vector. A, cDNA from
PI brain lysates of p75NTRExonlv wild-type (+/+), heterozygous (+/-) and null (-/-)
littermates prepared as described in Fig 1, followed by RT-PCR using primers for |3-actin
or regions of p75NTR upstream or downstream of the pGK-Neo cassette insertion in the
p75NTRExonIV targeting vector. The schematic diagram shows the p75NTR open reading
frame with arrows indicating location of primers used for RT-PCR analysis. B,
Alignment of mouse p75NTR (p75) cDNA sequence with p75NTR transcript cloned
from p75NTRExonIV/" mice. The putative start codon is underlined and putative Kozak
sequence boxed. Vertical lines indicate Exon IV-V and Exon V-VI junctions and boxes
underneath the sequence denote protein domains. C, Schematic diagram showing the
protein domains of mouse p75NTR and p75NTRExonIV proteins. D, Recombinant
p75NTRExonIV protein co-migrates with ~26 kDa protein present in PI brain lysates from
p75NTRExonIV null (-/-) mice. PI brain lysates from p75NTRExonIV wild-type (+/+),
heterozygous (+/-) and null (-/-) littermates were separated on a 15% SDS-PAGE gel
alongside lysates from 293T cells transiently transfected with plasmids encoding
p75NTRExonlv (Exon IV) or the parental vector (Mock). Immunoblotting was performed
as described in Fig 1 using a-p75NTR-Pl. Arrowheads indicate FL-p75NTR (upper) and
p75NTRExonIV (lower). Experiments in Panels A and D were repeated 3 times with
identical results.
60
Figure 4. p75NTRExonIV is membrane-associated and induces caspase zymogen
cleavage and JNK phosphorylation. A, PI brains from p75NTRExonIV heterozygote
(ExonlV +/-) and null (Exon IV -/-) littermates were subjected to subcellular fractionation
and fractions were analyzed by immunoblot. C denotes cytosolic fractions, Ml denotes
heavy membrane fraction and M2 denotes light membrane fraction. B, COS-7 cells were
transiently transfected for twenty-four hours with plasmids encoding either p75NTRExonIV
or encoding the p75NTR intracellular domain tagged with a myristoylation sequence,
which is targeted to the plasma membrane (Roux et al., 2001).
The cells were
subsequently fixed and stained with a-p75NTR-Pl and ct-nucleolin, a nuclear marker.
Left-hand panels show p75NTR staining using a Cy3-labeled donkey-anti-rabbit
secondary antibody, middle panels show nucleolin staining using a FITC-labeled donkeyanti-mouse secondary antibody, and right-hand panels show merged images. C,
p75NTRExonIV protein expression induces caspase zymogen cleavage. PC 12 cells were
transfected with control plasmid (pDEST 12.2) or plasmid expressing p75NTRExonlv
(Exon IV), in the presence or absence of plasmid expressing Bcl-2. Cells were lysed
forty-eight hours after transfection and analyzed by immunoblot to detect p75NTR and
Bcl-2 and, using cleavage-specific antibodies, to detect active caspase-3. IKBCX levels
were analyzed by immunoblot to confirm equal loading between lanes. Staurosporine
treatment was used as a positive control for caspase cleavage. D, p75NTRExonIV protein
expression induces phosphorylation of Jun kinase and ATF-2. PC 12 cells were left
untransfected or were transfected with 2 ug of plasmids encoding a control plasmid
(pcDNA3), or a clone of p75NTRExonIV protein (Exon IV) in the presence and absence of 2
ug of plasmid encoding Bcl-2. Cells were lysed twenty-four hours after transfection and
61
analyzed for levels of phospho-Thrl83/Tyrl85-JNK and total JNK, or phosphoThr69/71-ATF-2 and total ATF-2. Panels A, B, and C were repeated 3 times with
identical results.
62
Exon
B
Exon IV
Cerebellum
+/+ +/- -/- +/+ +/- -/-
+/+ +/- -/-
Heart
+/+ +/- -/-
|
83 —
62 —
83 —
62 —
a-P1
33— I
a-P1
33 —
25 —
25 —
17 —
17 —
•
83 —
62 —
83 —
62 —
a-p75NTR
33 —
25 —
25 —
17 —
17 —
48 —
- - a-p75NTR
33 —
a - p i l l tubulin
48 —
a-p||| tubulin
452 bp
I - li H
V
V
- IV
rn
- VI
V
V — V
V
Cysteine-Rich Domains
1
•
2
TM-
•
ICD
Neurotrophin
binding
+ RT
+/+ +/- -/500 bp
300 bp
-RT
+/+ +/- -/-
•3HHHH
9^^E^H
ACTIN
p75NTR-ICD
Figure 2
63
+ RT
+/+
+/- •/•
+/+
+/- -/ACTIN
Exon IV-VI (676 bp)
Exon l-lll (540 bp)
B
p75
ExonlV
7 8 1 AGCCGGAGGCACCTCCAGAGCGAGACCTCATAGCCAGCACAGTGGCCGATACGGTGACCA
1
ACCTCCAGAGCGAGACCTCATAGCCAGCACAGTGGCCGATACGGTGACCA
840
50
p75
ExonlV
8 4 1 CT GTGATGG KAGCTCCCAGCCTGTAGTGACCCGAGGCACCGCTGACAACCTCATTCCTG
5 1 CliGTGATGGPCAGCTCCCAGCCTGTAGTGACCCGAGGCACCGCTGACAACCTCATTCCTG
I
Stalk
II
I
900
110
p75
ExonlV
9 0 1 TCTATTGCTCCATCTTGGCTGCTGTGGTTGTGGGCCTTGTGGCCTATATTGCTTTCAAGA
1 1 1 TCTATTGCTCCATCTTGGCTGCTGTGGTTGTGGGCCTTGTGGCCTATATTGCTTTCAAGA
Transmembrane domain
960
170
p75
ExonlV
961 GAlTGGAACAGCTGCAAGCAAAATAAACAAGGAGCCAACAGCCGGCCGGTGAACCAGACAC
171 GAlTGGAACAGCTGCAAGCAAAATAAACAAGGAGCCAACAGCCGGCCGGTGAACCAGACAC
1020
230
J
El
CvtoDlasrtuc domain
1021 CCCCACCAGAGGGAGAGAAACTGCACAGCGACAGCGGCATCTCTGTGGACAGCCAGAGCC
p75
ExonlV 231 CCCCACCAGAGGGAGAGAAACTGCACAGCGACAGCGGCATCTCTGTGGACAGCCAGAGCC
1080
290
1081 TGCACGACCAGCAGACCCACACACAGACTGCCTCAGECCAAGCCCTCAAGGGTGATGGCA
p75
ExonlV 291 TGCACGACCAGCAGACCCACACACAGACTGCCTCAGCCCAAGCCCTCAAGGGTGATGGCA
1140
350
1141 ACCTCTACAGTAGCCTGCCCCTGACCAAGCGTGAGGAGGTCGAGAAGCTGCTCAATG6TG
P75
ExonlV 351 ACCTCTACAGTAGCCTGCGCCTGACCAAGCGTGAGGAGGTCGAGAAGCTGCTCAATGGTG
1200
410
1201 ACACCTGGCGACATCTGGCAGGCGAGCTGGGCTACCAGCCGGAGCATATAGACTCCTTTA
p75
ExonlV 411 ACACCTGGCGACATCTGGCAGGCGAGCTGGGCTACCAGCCGGAGCATATAGACTCCTTTA
Death domain
1260
470
1261 CCCACGAGGCCTGCCCAGTCCGAGCCCTGCTGGCCAGCTGGGGTGCCCAGGACAGCGCGA
471 CCCACGAGGCCTGCCCAGTCCGAGCCCTGCTGGCCAGCTGGGGTGCCCAGGACAGCGCGA
1320
530
1321 CGCTCGATGCCCTTTTAGCCGCCCTGCGACGCATCCAGAGAGCTGACATTGTGGAGAGCC
p75
ExonlV 531 CGCTCGATGCCCTTTTAGCCGCCCTGCGACGCATCCAGAGAGCTGACATTGTGGAGAGCC
1380
590
1381 TGTGCAGCGAGTCCACTGCCACGTCCCCTGTGTGAGCTCACCGACTGGGAGCCCCTGTCC
p75
ExonlV 591 TGTGCAGCGAGTCCACTGCCACGTCCCCTGTGTGAGCTCACCGACTGGGAGCCCCTGTCC
1440
650
1441 TGTCCCACATTCCGACCGCTGATGTTCTAGCCCCCACAGAGCCGCCCCCCTCTCCCTTGG
p75
ExonlV 651 TGTCCCACATTCCGACCGCTGATGTTC
1500
677
P 75
ExonlV
Brain
+/-
Cysleine-Rich Domains
1
2
3
4
TM
ICD
p75NTR
ICD
p75NTR e
Neurotrophin
binding
Figure 3
293 T
-/-
ExonlV
Mock
a-P1
64
Exon IV +/C M1 M2
Exon IV -/C M1 M2
83 —
-FL
62 —
- ^ " ~
33 —
_ _ _
~
25 —
B
1
Cy3
=
p75NTR
- ExonlV
-
FITC
Merged
p75NTR-mlCD
p75NTR
a
8
2
Q
Q.
&
&
Cvi
O
X
UJ
o
X
UJ
25-
62-
Cleaved
Caspase-3
17-
48 •
&•
mm - p 4 6 | ^
62-
- p55
4883 •
62 •
p75NTR
—
25
—
83
—
-pATF-2
-ExonlV
17
.ATF-2
—
• Bcl-2
83 •
62 •
25
JNK
•FL
33
25
- p46
—
-FL
p75NTR
; . IKBO
3325 •
-ExonlV
17 2517-
Figure 4
• Bcl-2
65
Closing Remarks
p75NTR plays an important role in the regulation of neuronal survival, apoptosis
and growth during both development and after injury in the adult nervous system, and the
signal transduction mechanisms that regulate these events are beginning to emerge. In
the first part of this thesis, I addressed an aspect of p75NTR-dependent apoptotic
signaling, in which the transcriptional events following JNK phosphorylation induced by
p75NTR activation were analyzed. In neurons deprived of trophic factor, JNK activation
results in phosphorylation of c-Jun which in turn promotes transcription of the BH3domain-only family members Bim and Hrk, proteins that facilitate Cytochrome C release
from the mitochondria (Harris and Johnson, 2001; Putcha et al., 2001; Whitfield et al.,
2001).
Given these data, it was hypothesized that p75NTR-dependent apoptosis
induced transcription of distinct BH3-domain only proteins. However, although p75NTR
induced JNK-dependent apoptosis in the cell types analyzed, no evidence for
transcriptional regulation of BH3-domain only family members was observed.
While BH3-domain-only proteins do not appear to be subject to transcriptional
regulation during apoptosis induced by p75NTR, subsequent work in our laboratory
established that p75NTR-mediated death correlated with the phosphorylation on
Serine 128 of BH3-domain-only family member, Bad. This work established that JNK is
responsible for this p75NTR-dependent Bad phosphorylation, since JNK inhibition
reduced the phosphorylation of Bad. Further, use of a dominant negative Bad Serine 128
significantly protected cells infected with p75NTR from p75NTR-induced apoptosis,
indicating that the JNK-Bad pathway is important for p75NTR-induced death. Future
66
studies using cultured neurons from Bad -/- mice will lend in vivo weight to the argument
that Bad is an important component of p75NTR-dependent cell death signaling.
During the second part of my M.Sc, I showed that p75NTRExonIV-/- mice express a
fragment of the p75NTR protein that contains a portion of the extracellular domain and
the complete transmembrane and intracellular domains.
The genotype-specific
expression profile of this fragment is consistent with the hypothesis that the pGK-Neo
cassette inserted into Exon IV of the p75NTR gene is responsible for this expression
pattern. When expressed in heterologous cells, this p75NTR fragment is membraneassociated where it may activate p75NTR signaling cascades that include JNK activation
and Procaspase-3 cleavage.
These results reverse the conclusions made by the Dechant group (von Schack et
al., 2001) that the p75NTRExonIV-/- mice represent a "complete" knockout in comparison
with the earlier p75NTRExonI11-/- mice (Lee et al., 1992). Evidence for this is based on RTPCR results using primers directed against the p75NTR intracellular domain, where it
was shown that mRNAs derived from this portion of p75NTR was increased as a function
of p75NTRExonIV mutant alleles. Immunoblots using two different antibodies directed
against the p75NTR intracellular domain also showed the same genotype-specific profile.
While there is some concern that the 26 kDa protein observed in mice with p75NTRExonIV
mutations might reflect cross-reaction of p75NTR antibodies with proteins that bear
homology to p75NTR such as PLAIDD (Frankowski et al., 2002), the RT-PCR data
provides compelling evidence that the intracellular domain of p75NTR is aberrantly
produced in the p75NTRExonIV-/- mice.
67
Our results suggest that the pGK-Neo cassette placed within Exon IV of p75NTR
results in aberrant transcription of the adjacent 3' portion of the gene which, due to the
presence of a cryptic Kozak consensus sequence in Exon IV, results in production of the
p75NTRExon IV protein product. The PGK-1 promoter and enhancer sequence functions in
an orientation and a position-independent manner (McBurney et al., 1991) and several
groups have demonstrated that retention of the pGK-Neo cassette in targeted loci can
profoundly influence the expression of surrounding genes upstream and downstream
from the cassette (Olson et al., 1996; Pham et al., 1996). One study examined targeted
mutations in two multigene clusters, the granzyme B locus and the (3-like globin gene
cluster and found that the pGK-Neo insertion abrogated the expression of multiple genes
both upstream and downstream from the cassette (Pham et al., 1996). Other examples of
this phenomenon have also been suggested in mice with targeted mutations of the
myogenic basis helix-loop-helix gene MRF4, the Igk light chain intronic enhancer, and
individual genes in the Hox gene clusters (Horan et al., 1995; Horan et al., 1994;
Jeannotte et al., 1993; Kostic and Capecchi, 1994; Ramirez-Solis et al., 1993; Zhang et
al., 1995). Our results are novel in that they suggest that retention of pGK-Neo
effectively induces rather than abrogates transcription of an adjacent portion of p75NTR,
and we cannot rule out the possibility that nearby genes are also affected. Our findings,
along with others, underscore the unpredictable phenotypes that can result from retained
pGK-Neo cassettes.
Initial characterization of the p75NTRExonlv-/- mice revealed severe peripheral
neuron loss as well as dilation and ruptures within large blood vessel (von Schack et al,
2001). We have demonstrated that the p75NTRExon IV protein was highly expressed in
68
heart lysates of PI p75NTRExonlv/-mice and have shown that the p75NTRExon ,v protein
product can activate p75NTR signaling cascades that result in Procaspase-3 cleavage.
Our previous studies have shown that expression of the p75NTR intracellular domain
within neurons of transgenic mice can induce apoptosis of peripheral and central neurons
in vivo (Majdan et al., 1997) and together, these findings raise the possibility that some
characteristics of the p75NTRExon IV mouse are due to gain-of-function mutations rather
than loss of p75NTR function.
At a minimum, the presence of the p75NTRExonIV protein product in these animals
indicates that previously observed phenotypes should be interpreted with caution. In
particular, the increased number of basal forebrain cholinergic neurons in p75NTRExonIV/"
mice warrants re-interpretation (Naumann et al., 2002). The actions of p75NTR are
complex and reports from ourselves and several other groups have shown that p75NTR
can, in some circumstances, induce a survival response, through activation of Akt
(DeFreitas et al., 2001; Descamps et al., 2001; Roux et al., 2001). We have tested
whether the aberrant p75NTR product detected in the p75NTRExonIV-/- is capable of
activating survival pathways and observed increased phosphorylation of Akt in PC 12 and
HEK293 cells transfected with the p75NTRExonIV protein product (data not shown). Thus,
the aberrant p75NTR product is capable of activating antagonistic pathways similar to
those elicited by the intact receptor and this suggests that the effects of aberrant p75NTR
product will vary as a function of tissue-specific expression levels and with specific
cellular contexts. The Dechant group showed increased survival of basal forebrain
cholinergic neurons of p75NTRExonIV-/- mice (Naumann et al., 2002) and, when coupled
with our data, two possibilities emerge. First, these neurons may express little or no
69
truncated p75NTR and this phenotype may reflect a bona fide p75NTR loss of function
phenotype. Alternatively, increased survival of basal forebrain cholinergic neurons
observed in the p75NTRExonIV -/- mice may reflect a survival gain-of-function effect due
to the activation of Akt induced by expression of the aberrant p75NTR product. Further
analysis of the p75NTRExonlv expression in the basal forebrain cholinergic neurons of
p75NTRExonIV -/- mice would help to resolve this issue.
Although p75NTR was the first discovered member of the TNF receptor
superfamily and was the first identified neurotrophin receptor, its physiological function
has not been completely elucidated. Recent findings indicate roles that range from
promotion of survival pathways, induction of apoptosis, effects on cell cycle and
differentiation, and facilitation or inhibition of growth (Barrett, 2000; Kaplan and Miller,
2003; McKerracher and Winton, 2002; Roux and Barker, 2002). Generation of strains of
mice that are rendered null for p75NTR gene products remains an absolute requirement
for the elucidation of its biological activities.
70
Bibliography
Allsopp, T.E., Robinson, M., Wyatt, S. and Davies, A.M. (1993) Ectopic trkA expression
mediates a NGF survival response in NGF-independent sensory neurons but not
in parasympathetic neurons. J Cell Biol, 123, 1555-1566.
Aloyz, R.S., Bamji, S.X., Pozniak, CD., Toma, J.G., Atwal, J., Kaplan, D.R. and Miller,
F.D. (1998) p53 is essential for developmental neuron death as regulated by the
TrkA and p75 neurotrophin receptors. J Cell Biol, 143, 1691-1703.
Anderson, C.N. and Tolkovsky, A.M. (1999) A role for MAPK/ERK in sympathetic
neuron survival: protection against a p53-dependent, JNK-independent induction
of apoptosis by cytosine arabinoside. J Neurosci, 19, 664-673.
Anton, E.S., Weskamp, G., Reichardt, L.F. and Matthew, W.D. (1994) Nerve growth
factor and its low-affinity receptor promote Schwann cell migration. Proc Natl
Acad Sci US A, 91, 2795-2799.
Bagum, M.A., Miyamoto, O., Toyoshima, T., Masada, T., Nagahata, S. and Itano, T.
(2001) The contribution of low affinity NGF receptor (p75NGFR) to delayed
neuronal death after ischemia in the gerbil hippocampus. Acta Med Okayama, 55,
19-24.
Baker, S.J. and Reddy, E.P. (1998) Modulation of life and death by the TNF receptor
superfamily. Oncogene, 17, 3261-3270.
Baldwin, A.N., Bitler, CM., Welcher, A.A. and Shooter, E.M. (1992) Studies on the
structure and binding properties of the cysteine-rich domain of rat low affinity
nerve growth factor receptor (p75NGFR). J Biol Chem, 267, 8352-8359.
Bamji, S.X., Majdan, M., Pozniak, CD., Belliveau, D.J., Aloyz, R., Kohn, J., Causing,
C.G. and Miller, F.D. (1998) The p75 neurotrophin receptor mediates neuronal
apoptosis and is essential for naturally occurring sympathetic neuron death. J Cell
Biol, 140, 911-923.
Banfield, M.J., Naylor, R.L., Robertson, A.G., Allen, S.J., Dawbarn, D. and Brady, R.L.
(2001) Specificity in Trk receptor:neurotrophin interactions: the crystal structure
of TrkB-d5 in complex with neurotrophin-4/5. Structure (Camb), 9, 1191-1199.
Barde, Y.A. (1989) Trophic factors and neuronal survival. Neuron, 2, 1525-1534.
71
Barker, P.A., Barbee, G., Misko, T.P. and Shooter, E.M. (1994) The low affinity
neurotrophin receptor, p75LNTR, is palmitoylated by thioester formation through
cysteine 219. J Biol Chem, 269, 30645-30650.
Barker, P.A. and Shooter, E.M. (1994) Disruption of NGF binding to the low affinity
neurotrophin receptor p75LNTR reduces NGF binding to TrkA on PC 12 cells.
Neuron, 13, 203-215.
Barrett, G.L. (2000) The p75 neurotrophin receptor and neuronal apoptosis. Prog
Neurobiol, 61, 205-229.
Barrett, G.L. and Bartlett, P.F. (1994) The p75 nerve growth factor receptor mediates
survival or death depending on the stage of sensory neuron development. Proc
Natl Acad Sci US A, 91, 6501-6505.
Barrett, G.L. and Georgiou, A. (1996) The low-affinity nerve growth factor receptor
p75NGFR mediates death of PC 12 cells after nerve growth factor withdrawal. J
Neurosci Res, 45, 117-128.
Bazenet, C.E., Mota, M.A. and Rubin, L.L. (1998) The small GTP-binding protein Cdc42
is required for nerve growth factor withdrawal-induced neuronal death. Proc Natl
Acad Sci USA, 95, 3984-3989.
Beattie, M.S., Harrington, A.W., Lee, R., Kim, J.Y., Boyce, S.L., Longo, F.M.,
Bresnahan, J.C, Hempstead, B.L. and Yoon, S.O. (2002) ProNGF induces p75mediated death of oligodendrocytes following spinal cord injury. Neuron, 36,
375-386.
Bentley, C.A. and Lee, K.F. (2000) p75 is important for axon growth and Schwann cell
migration during development. J Neurosci, 20, 7706-7715.
Bhakar, A.L., Tannis, L.L., Zeindler, C, Russo, M.P., Jobin, C , Park, D.S., MacPherson,
S. and Barker, P.A. (2002) Constitutive nuclear factor-kappa B activity is required
for central neuron survival. J Neurosci, 22, 8466-8475.
Bibel, M., Hoppe, E. and Barde, Y.A. (1999) Biochemical and functional interactions
between the neurotrophin receptors trk and p75NTR. Embo J, 18, 616-622.
Biswas, S.C and Greene, L.A. (2002) Nerve growth factor (NGF) down-regulates the
Bcl-2 homology 3 (BH3) domain-only protein Bim and suppresses its
proapoptotic activity by phosphorylation. J Biol Chem, 277, 49511-49516.
72
Blume-Jensen, P., Janknecht, R. and Hunter, T. (1998) The kit receptor promotes cell
survival via activation of PI 3-kinase and subsequent Akt-mediated
phosphorylation of Bad on Serl36. Curr Biol, 8, 779-782.
Bruckner, S.R., Tammariello, S.P., Kuan, C.Y., Flavell, R.A., Rakic, P. and Estus, S.
(2001) JNK3 contributes to c-Jun activation and apoptosis but not oxidative stress
in nerve growth factor-deprived sympathetic neurons. / Neurochem, 78, 298-303.
Casaccia-Bonnefil, P., Carter, B.D., Dobrowsky, R.T. and Chao, M.V. (1996) Death of
oligodendrocytes mediated by the interaction of nerve growth factor with its
receptor p75. Nature, 383, 716-719.
Casha, S., Yu, W.R. and Fehlings, M.G. (2001) Oligodendroglial apoptosis occurs along
degenerating axons and is associated with FAS and p75 expression following
spinal cord injury in the rat. Neuroscience, 103, 203-218.
Chan, F.K., Chun, H.J., Zheng, L., Siegel, R.M., Bui, K.L. and Lenardo, M.J. (2000) A
domain in TNF receptors that mediates ligand-independent receptor assembly and
signaling. Science, 288, 2351-2354.
Chao, M.V., Bothwell, M.A., Ross, A.H., Koprowski, H., Lanahan, A.A., Buck, C.R. and
Sehgal, A. (1986) Gene transfer and molecular cloning of the human NGF
receptor. Science, 232, 518-521.
Chapman, B.S. and Kuntz, I.D. (1995) Modeled structure of the 75-kDa neurotrophin
receptor. Protein Sci, 4, 1696-1707.
Cheema, S.S., Barrett, G.L. and Bartlett, P.F. (1996) Reducing p75 nerve growth factor
receptor levels using antisense oligonucleotides prevents the loss of axotomized
sensory neurons in the dorsal root ganglia of newborn rats. J Neurosci Res, 46,
239-245.
Cordon-Cardo, C , Tapley, P., Jing, S.Q., Nanduri, V., O'Rourke, E., Lamballe, F.,
Kovary, K., Klein, R., Jones, K.R., Reichardt, L.F. and et al. (1991) The trk
tyrosine protein kinase mediates the mitogenic properties of nerve growth factor
and neurotrophin-3. Cell, 66, 173-183.
Cosgaya, J.M., Chan, J.R. and Shooter, E.M. (2002) The neurotrophin receptor p75NTR
as a positive modulator of myelination. Science, 298, 1245-1248.
73
Datta, S.R., Dudek, H., Tao, X., Masters, S., Fu, H., Gotoh, Y. and Greenberg, M.E.
(1997) Akt phosphorylation of BAD couples survival signals to the cell-intrinsic
death machinery. Cell, 91, 231-241.
Davies, A.M. (2000) Neurotrophins: neurotrophic modulation of neurite growth. Curr
Biol, 10, R198-200.
Davies, A.M., Lee, K.F. and Jaenisch, R. (1993) p75-deficient trigeminal sensory neurons
have an altered response to NGF but not to other neurotrophins. Neuron, 11, 565574.
Dechant, G. and Barde, Y.A. (1997) Signalling through the neurotrophin receptor
p75NTR. Curr Opin Neurobiol, 7, 413-418.
DeFreitas, M.F., McQuillen, P.S. and Shatz, C.J. (2001) A novel p75NTR signaling
pathway promotes survival, not death, of immunopurified neocortical subplate
neurons. J Neurosci, 21, 5121-5129.
del Peso, L., Gonzalez-Garcia, M., Page, C , Herrera, R. and Nunez, G. (1997)
Interleukin-3-induced phosphorylation of BAD through the protein kinase Akt.
Science, 278, 687-689.
Descamps, S., Toillon, R.A., Adriaenssens, E., Pawlowski, V., Cool, S.M., Nurcombe,
V., Le Bourhis, X., Boilly, B., Peyrat, J.P. and Hondermarck, H. (2001) Nerve
growth factor stimulates proliferation and survival of human breast cancer cells
through two distinct signaling pathways. J Biol Chem, 276, 17864-17870.
Deshmukh, M. and Johnson, E.M., Jr. (1998) Evidence of a novel event during neuronal
death: development of competence-to-die in response to cytoplasmic cytochrome
c. Neuron, 21, 695-705.
Deshmukh, M., Vasilakos, J., Deckwerth, T.L., Lampe, P.A., Shivers, B.D. and Johnson,
E.M., Jr. (1996) Genetic and metabolic status of NGF-deprived sympathetic
neurons saved by an inhibitor of ICE family proteases. J Cell Biol, 135, 13411354.
Domeniconi, M., Cao, Z., Spencer, T., Sivasankaran, R., Wang, K., Nikulina, E., Kimura,
N., Cai, H., Deng, K., Gao, Y., He, Z. and Filbin, M. (2002) Myelin-associated
glycoprotein interacts with the Nogo66 receptor to inhibit neurite outgrowth.
Neuron, 35, 283-290.
74
Donovan, N., Becker, E.B., Konishi, Y. and Bonni, A. (2002) JNK phosphorylation and
activation of BAD couples the stress-activated signaling pathway to the cell death
machinery. J Biol Chem, 277, 40944-40949.
Dostaler, S.M., Ross, G.M., Myers, S.M., Weaver, D.F., Ananthanarayanan, V. and
Riopelle, R.J. (1996) Characterization of a distinctive motif of the low molecular
weight neurotrophin receptor that modulates NGF-mediated neurite growth. Eur J
Neurosci, 8, 870-879.
Dowling, P., Ming, X., Raval, S., Husar, W., Casaccia-Bonnefil, P., Chao, M., Cook, S.
and Blumberg, B. (1999) Up-regulated p75NTR neurotrophin receptor on glial
cells in MS plaques. Neurology, 53, 1676-1682.
Eilers, A., Whitfield, J., Shah, B., Spadoni, C , Desmond, H. and Ham, J. (2001) Direct
inhibition of c-Jun N-terminal kinase in sympathetic neurones prevents c-jun
promoter activation and NGF withdrawal-induced death. J Neurochem, 76, 14391454.
Ernfors, P., Ibanez, C.F., Ebendal, T., Olson, L. and Persson, H. (1990) Molecular
cloning and neurotrophic activities of a protein with structural similarities to
nerve growth factor: developmental and topographical expression in the brain.
Proc Natl Acad Sci USA, 87, 5454-5458.
Esposito, D., Patel, P., Stephens, R.M., Perez, P., Chao, M.V., Kaplan, D.R. and
Hempstead, B.L. (2001) The cytoplasmic and transmembrane domains of the p75
and Trk A receptors regulate high affinity binding to nerve growth factor. J Biol
Chem, 276, 32687-32695.
Fahnestock, M., Michalski, B., Xu, B. and Coughlin, M.D. (2001) The precursor pronerve growth factor is the predominant form of nerve growth factor in brain and is
increased in Alzheimer's disease. Mol Cell Neurosci, 18, 210-220.
Fainzilber, M., Smit, A.B., Syed, N.I., Wildering, W.C., Hermann, van der Schors, R.C,
Jimenez, C , Li, K.W., van Minnen, J., Bulloch, A.G., Ibanez, CF. and Geraerts,
W.P. (1996) CRNF, a molluscan neurotrophic factor that interacts with the p75
neurotrophin receptor. Science, 274, 1540-1543.
75
Fan, G., Merritt, S.E., Kortenjann, M., Shaw, P.E. and Holzman, L.B. (1996) Dual
leucine zipper-bearing kinase (DLK) activates p46SAPK and p38mapk but not
ERK2. J Biol Chem, 111, 24788-24793.
Ferri, C.C. and Bisby, M.A. (1999) Improved survival of injured sciatic nerve Schwann
cells in mice lacking the p75 receptor. Neurosci Lett, 272, 191-194.
Ferri, C.C, Moore, F.A. and Bisby, M.A. (1998) Effects of facial nerve injury on mouse
motoneurons lacking the p75 low-affinity neurotrophin receptor. J Neurobiol, 34,
1-9.
Fournier, A.E., GrandPre, T. and Strittmatter, S.M. (2001) Identification of a receptor
mediating Nogo-66 inhibition of axonal regeneration. Nature, 409, 341-346.
Frade, J.M. and Barde, Y.A. (1999) Genetic evidence for cell death mediated by nerve
growth factor and the neurotrophin receptor p75 in the developing mouse retina
and spinal cord. Development, 126, 683-690.
Frade, J.M., Rodriguez-Tebar, A. and Barde, Y.A. (1996) Induction of cell death by
endogenous nerve growth factor through its p75 receptor. Nature, 383, 166-168.
Frankowski, H., Castro-Obregon, S., del Rio, G., Rao, R.V. and Bredesen, D.E. (2002)
PLAIDD, a type II death domain protein that interacts with p75 neurotrophin
receptor. Neuromolecular Med, 1, 153-170.
Friedman, W.J. (2000) Neurotrophins induce death of hippocampal neurons via the p75
receptor. J Neurosci, 20, 6340-6346.
Gentry, J.J., Casaccia-Bonnefil, P. and Carter, B.D. (2000) Nerve growth factor
activation of nuclear factor kappaB through its p75 receptor is an anti-apoptotic
signal in RN22 schwannoma cells. J Biol Chem, 275, 7558-7565.
Ghosh, A., Carnahan, J. and Greenberg, M.E. (1994) Requirement for BDNF in activitydependent survival of cortical neurons. Science, 263, 1618-1623.
Grob, P.M., Ross, A.H., Koprowski, H. and Bothwell, M. (1985) Characterization of the
human melanoma nerve growth factor receptor. J Biol Chem, 260, 8044-8049.
Gross, A., McDonnell, J.M. and Korsmeyer, S.J. (1999) BCL-2 family members and the
mitochondria in apoptosis. Genes Dev, 13, 1899-1911.
Gu, C , Casaccia-Bonnefil, P., Srinivasan, A. and Chao, M.V. (1999) Oligodendrocyte
apoptosis mediated by caspase activation. J Neurosci, 19, 3043-3049.
76
Hamanoue, M., Middleton, G., Wyatt, S., Jaffray, E., Hay, R.T. and Davies, A.M. (1999)
p75-mediated NF-kappaB activation enhances the survival response of developing
sensory neurons to nerve growth factor. Mol Cell Neurosci, 14, 28-40.
Hantzopoulos, P.A., Suri, C , Glass, D.J., Goldfarb, M.P. and Yancopoulos, G.D. (1994)
The low affinity NGF receptor, p75, can collaborate with each of the Trks to
potentiate functional responses to the neurotrophins. Neuron, 13, 187-201.
Harding, T.C., Xue, L., Bienemann, A., Haywood, D., Dickens, M., Tolkovsky, A.M. and
Uney, J.B. (2001) Inhibition of JNK by overexpression of the JNL binding
domain of JIP-1 prevents apoptosis in sympathetic neurons. J Biol Chem, 276,
4531-4534.
Harrington, A.W., Kim, J.Y. and Yoon, S.O. (2002) Activation of Rac GTPase by p75 is
necessary for c-jun N-terminal kinase-mediated apoptosis. J Neurosci, 22, 156166.
Harris, C.A. and Johnson, E.M., Jr. (2001) BH3-only Bcl-2 family members are
coordinately regulated by the JNK pathway and require Bax to induce apoptosis
in neurons. J Biol Chem, 276, 37754-37760.
Hempstead, B.L., Martin-Zanca, D., Kaplan, D.R., Parada, L.F. and Chao, M.V. (1991)
High-affinity NGF binding requires coexpression of the trk proto-oncogene and
the low-affinity NGF receptor. Nature, 350, 678-683.
Higuchi, H., Yamashita, T., Yoshikawa, H. and Tohyama, M. (2003) PKA
phosphorylates the p75 receptor and regulates its localization to lipid rafts. Embo
J, 22, 1790-1800.
Hirai, S., Izawa, M., Osada, S., Spyrou, G. and Ohno, S. (1996) Activation of the JNK
pathway by distantly related protein kinases, MEKK and MUK. Oncogene, 12,
641-650.
Hohn, A., Leibrock, J., Bailey, K. and Barde, Y.A. (1990) Identification and
characterization of a novel member of the nerve growth factor/brain-derived
neurotrophic factor family. Nature, 344, 339-341.
Horan, G.S., Kovacs, E.N., Behringer, R.R. and Featherstone, M.S. (1995) Mutations in
paralogous Hox genes result in overlapping homeotic transformations of the axial
skeleton: evidence for unique and redundant function. Dev Biol, 169, 359-372.
77
Horan, G.S., Wu, K., Wolgemuth, D.J. and Behringer, R.R. (1994) Homeotic
transformation of cervical vertebrae in Hoxa-4 mutant mice. Proc Natl Acad Sci
US A, 91, 12644-12648.
Huang, B., Eberstadt, M., Olejniczak, E.T., Meadows, R.P. and Fesik, S.W. (1996) NMR
structure and mutagenesis of the Fas (APO-1/CD95) death domain. Nature, 384,
638-641.
Huang, D.C and Strasser, A. (2000) BH3-Only proteins-essential initiators of apoptotic
cell death. Cell, 103, 839-842.
Huang, E.J. and Reichardt, L.F. (2001) Neurotrophins: roles in neuronal development and
function. Annu Rev Neurosci, 24, 677-736.
Hughes, A.L., Messineo-Jones, D., Lad, S.P. and Neet, K.E. (2001) Distinction between
differentiation, cell cycle, and apoptosis signals in PC 12 cells by the nerve growth
factor mutant delta9/13, which is selective for the p75 neurotrophin receptor. J
Neurosci Res, 63, 10-19.
Imaizumi, K., Morihara, T., Mori, Y., Katayama, T., Tsuda, M., Furuyama, T., Wanaka,
A., Takeda, M. and Tohyama, M. (1999) The cell death-promoting gene DP5,
which interacts with the BCL2 family, is induced during neuronal apoptosis
following exposure to amyloid beta protein. J Biol Chem, 274, 7975-7981.
Imaizumi, K., Tsuda, M., Imai, Y., Wanaka, A., Takagi, T. and Tohyama, M. (1997)
Molecular cloning of a novel polypeptide, DP5, induced during programmed
neuronal death. J Biol Chem, 272, 18842-18848.
Ip, N.Y., Stitt, T.N., Tapley, P., Klein, R., Glass, D.J., Fandl, J., Greene, L.A., Barbacid,
M. and Yancopoulos, G.D. (1993) Similarities and differences in the way
neurotrophins interact with the Trk receptors in neuronal and nonneuronal cells.
Neuron, 10, 137-149.
Jacobson, M.D., Weil, M. and Raff, M.C (1997) Programmed cell death in animal
development. Cell, 88, 347-354.
Jeannotte, L., Lemieux, M., Charron, J., Poirier, F. and Robertson, E.J. (1993)
Specification of axial identity in the mouse: role of the Hoxa-5 (Hoxl.3) gene.
Genes Dev, 7, 2085-2096.
78
Johnson, D., Lanahan, A., Buck, C.R., Sehgal, A., Morgan, C , Mercer, E., Bothwell, M.
and Chao, M. (1986) Expression and structure of the human NGF receptor. Cell,
47, 545-554.
Kaplan, D.R., Hempstead, B.L., Martin-Zanca, D., Chao, M.V. and Parada, L.F. (1991a)
The trk proto-oncogene product: a signal transducing receptor for nerve growth
factor. Science, 252, 554-558.
Kaplan, D.R., Martin-Zanca, D. and Parada, L.F. (1991b) Tyrosine phosphorylation and
tyrosine kinase activity of the trk proto-oncogene product induced by NGF.
Nature, 350, 158-160.
Kaplan, D.R. and Miller, F.D. (2000) Neurotrophin signal transduction in the nervous
system. Curr Opin Neurobiol, 10, 381-391.
Kaplan, D.R. and Miller, F.D. (2003) Axon growth inhibition: signals from the p75
neurotrophin receptor. Nat Neurosci, 6, 435-436.
Khursigara, G., Bertin, J., Yano, H., Moffett, H., DiStefano, P.S. and Chao, M.V. (2001)
A prosurvival function for the p75 receptor death domain mediated via the
caspase recruitment domain receptor-interacting protein 2. J Neurosci, 21, 58545863.
Klein, R., Conway, D., Parada, L.F. and Barbacid, M. (1990) The trkB tyrosine protein
kinase gene codes for a second neurogenic receptor that lacks the catalytic kinase
domain. Cell, 61, 647-656.
Klein, R., Jing, S.Q., Nanduri, V., O'Rourke, E. and Barbacid, M. (1991a) The trk protooncogene encodes a receptor for nerve growth factor. Cell, 65, 189-197.
Klein, R., Nanduri, V., Jing, S.A., Lamballe, F., Tapley, P., Bryant, S., Cordon-Cardo, C ,
Jones, K.R., Reichardt, L.F. and Barbacid, M. (1991b) The trkB tyrosine protein
kinase is a receptor for brain-derived neurotrophic factor and neurotrophin-3.
Cell, 66, 395-403.
Koch, G., Haberman, B., Mohr, C , Just, I. and Aktories, K. (1991) Interaction of
mastoparan with the low molecular mass GTP-binding proteins rho/rac. FEBS
Lett, 291, 336-340.
79
Kokaia, Z., Andsberg, G., Martinez-Serrano, A. and Lindvall, O. (1998) Focal cerebral
ischemia in rats induces expression of P75 neurotrophin receptor in resistant
striatal cholinergic neurons. Neuroscience, 84, 1113-1125.
Konishi, Y., Lehtinen, M., Donovan, N. and Bonni, A. (2002) Cdc2 phosphorylation of
BAD links the cell cycle to the cell death machinery. Mol Cell, 9, 1005-1016.
Kostic, D. and Capecchi, M.R. (1994) Targeted disruptions of the murine Hoxa-4 and
Hoxa-6 genes result in homeotic transformations of components of the vertebral
column. Mech Dev, 46, 231-247.
Kuner, P., Schubenel, R. and Hertel, C (1998) Beta-amyloid binds to p57NTR and
activates NFkappaB in human neuroblastoma cells. J Neurosci Res, 54, 798-804.
Lamballe, F., Klein, R. and Barbacid, M. (1991) trkC, a new member of the trk family of
tyrosine protein kinases, is a receptor for neurotrophin-3. Cell, 66, 967-979.
Large, T.H., Weskamp, G., Helder, J.C., Radeke, M.J., Misko, T.P., Shooter, E.M. and
Reichardt, L.F. (1989) Structure and developmental expression of the nerve
growth factor receptor in the chicken central nervous system. Neuron, 2, 11231134.
Lee, K.F., Davies, A.M. and Jaenisch, R. (1994) p75-deficient embryonic dorsal root
sensory and neonatal sympathetic neurons display a decreased sensitivity to NGF.
Development, 120, 1027-1033.
Lee, K.F., Li, E., Huber, L.J., Landis, S.C, Sharpe, A.H., Chao, M.V. and Jaenisch, R.
(1992) Targeted mutation of the gene encoding the low affinity NGF receptor p75
leads to deficits in the peripheral sensory nervous system. Cell, 69, 737-749.
Lee, R., Kermani, P., Teng, K.K. and Hempstead, B.L. (2001) Regulation of cell survival
by secreted proneurotrophins. Science, 294, 1945-1948.
Lei, K. and Davis, R.J. (2003) JNK phosphorylation of Bim-related members of the Bcl2
family induces Bax-dependent apoptosis. Proc Natl Acad Sci USA, 100, 24322437.
Lei, K., Nimnual, A., Zong, W.X., Kennedy, N.J., Flavell, R.A., Thompson, C.B., BarSagi, D. and Davis, R.J. (2002) The Bax subfamily of Bcl2-related proteins is
essential for apoptotic signal transduction by c-Jun NH(2)-terminal kinase. Mol
Cell Biol, 22, 4929-4942.
80
Levi-Montalcini, R. (1987) The nerve growth factor: thirty-five years later. Biosci Rep, 1,
681-699.
Li, H., Zhu, H., Xu, C.J. and Yuan, J. (1998) Cleavage of BID by caspase 8 mediates the
mitochondrial damage in the Fas pathway of apoptosis. Cell, 94, 491-501.
Liepinsh, E., Hag, L.L., Otting, G. and Ibanez, CF. (1997) NMR structure of the death
domain of the p75 neurotrophin receptor. Embo J, 16, 4999-5005.
Liu, B.P., Fournier, A., GrandPre, T. and Strittmatter, S.M. (2002) Myelin-associated
glycoprotein as a functional ligand for the Nogo-66 receptor. Science, 297, 11901193.
Luo, X., Budihardjo, I., Zou, H., Slaughter, C and Wang, X. (1998) Bid, a Bcl2
interacting protein, mediates cytochrome c release from mitochondria in response
to activation of cell surface death receptors. Cell, 94, 481-490.
Mahadeo, D., Kaplan, L., Chao, M.V. and Hempstead, B.L. (1994) High affinity nerve
growth factor binding displays a faster rate of association than pl40trk binding.
Implications for multi-subunit polypeptide receptors. J Biol Chem, 269, 68846891.
Majdan, M., Lachance, C , Gloster, A., Aloyz, R., Zeindler, C , Bamji, S., Bhakar, A.,
Belliveau, D., Fawcett, J., Miller, F.D. and Barker, P.A. (1997) Transgenic mice
expressing the intracellular domain of the p75 neurotrophin receptor undergo
neuronal apoptosis. J Neurosci, 17, 6988-6998.
Majdan, M., Walsh, G.S., Aloyz, R. and Miller, F.D. (2001) TrkA mediates
developmental sympathetic neuron survival in vivo by silencing an ongoing
p75NTR-mediated death signal. J Cell Biol, 155, 1275-1285.
Martin, D.P., Schmidt, R.E., DiStefano, P.S., Lowry, O.H., Carter, J.G. and Johnson,
E.M., Jr. (1988) Inhibitors of protein synthesis and RNA synthesis prevent
neuronal death caused by nerve growth factor deprivation. J Cell Biol, 106, 829844.
Martinez-Murillo, R., Fernandez, A.P., Bentura, M.L. and Rodrigo, J. (1998) Subcellular
localization of low-affinity nerve growth factor receptor-immunoreactive protein
in adult rat purkinje cells following traumatic injury. Exp Brain Res, 119, 47-57.
81
Martinou, I., Desagher, S., Eskes, R., Antonsson, B., Andre, E., Fakan, S. and Martinou,
J.C (1999) The release of cytochrome c from mitochondria during apoptosis of
NGF- deprived sympathetic neurons is a reversible event. J Cell Biol, 144, 883889.
McBurney, M.W., Sutherland, L.C, Adra, C.N., Leclair, B., Rudnicki, M.A. and Jardine,
K. (1991) The mouse Pgk-1 gene promoter contains an upstream activator
sequence. Nucleic Acids Res, 19, 5755-5761.
McKerracher, L. and Winton, M.J. (2002) Nogo on the go. Neuron, 36, 345-348.
Miller, F.D., Speelman, A., Mathew, T.C, Fabian, J., Chang, E., Pozniak, C. and Toma,
J.G. (1994) Nerve growth factor derived from terminals selectively increases the
ratio of p75 to trkA NGF receptors on mature sympathetic neurons. Dev Biol, 161,
206-217.
Mischel, P.S., Smith, S.G., Vining, E.R., Valletta, J.S., Mobley, W.C and Reichardt, L.F.
(2001) The extracellular domain of p75NTR is necessary to inhibit neurotrophin-3
signaling through TrkA. / Biol Chem, 276, 11294-11301.
Mowla, S.J., Pareek, S., Farhadi, H.F., Petrecca, K., Fawcett, J.P., Seidah, N.G., Morris,
S.J., Sossin, W.S. and Murphy, R.A. (1999) Differential sorting of nerve growth
factor and brain-derived neurotrophic factor in hippocampal neurons. J Neurosci,
19, 2069-2080.
Naumann, T., Casademunt, E., Hollerbach, E., Hofmann, J., Dechant, G., Frotscher, M.
and Barde, Y.A. (2002) Complete deletion of the neurotrophin receptor p75NTR
leads to long-lasting increases in the number of basal forebrain cholinergic
neurons. J Neurosci, 22, 2409-2418.
Nicholson, D.W. and Thornberry, N.A. (1997) Caspases: killer proteases. Trends
Biochem Sci, 22, 299-306.
Oda, E., Ohki, R., Murasawa, H., Nemoto, J., Shibue, T., Yamashita, T., Tokino, T.,
Taniguchi, T. and Tanaka, N. (2000) Noxa, a BH3-only member of the Bcl-2
family and candidate mediator of p53-induced apoptosis. Science, 288, 10531058.
82
Oh, J.D., Chartisathian, K., Chase, T.N. and Butcher, L.L. (2000) Overexpression of
neurotrophin receptor p75 contributes to the excitotoxin-induced cholinergic
neuronal death in rat basal forebrain. Brain Res, 853, 174-185.
Olson, E.N., Arnold, H.H., Rigby, P.W. and Wold, B.J. (1996) Know your neighbors:
three phenotypes in null mutants of the myogenic bHLH gene MRF4. Cell, 85, 14.
Oppenheim, R.W. (1991) Cell death during development of the nervous system. Annu
Rev Neurosci, 14, 453-501.
Perini, G., Della-Bianca, V., Politi, V., Delia Valle, G., Dal-Pra, I., Rossi, F. and Armato,
U. (2002) Role of p75 neurotrophin receptor in the neurotoxicity by beta-amyloid
peptides and synergistic effect of inflammatory cytokines. J Exp Med, 195, 907918.
Pettmann, B. and Henderson, CE. (1998) Neuronal cell death. Neuron, 20, 633-647.
Pham, C.T., Maclvor, D.M., Hug, B.A., Heusel, J.W. and Ley, T.J. (1996) Long-range
disruption of gene expression by a selectable marker cassette. Proc Natl Acad Sci
US A, 93, 13090-13095.
Purves, D., Snider, W.D. and Voyvodic, J.T. (1988) Trophic regulation of nerve cell
morphology and innervation in the autonomic nervous system. Nature, 336, 123128.
Putcha, G.V., Deshmukh, M. and Johnson, E.M., Jr. (1999) BAX translocation is a
critical event in neuronal apoptosis: regulation by neuroprotectants, BCL-2, and
caspases. J Neurosci, 19, 7476-7485.
Putcha, G.V., Le, S., Frank, S., Besirli, C.G., Clark, K., Chu, B., Alix, S., Youle, R.J.,
LaMarche, A., Maroney, A.C. and Johnson, E.M. (2003) JNK-Mediated BIM
Phosphorylation Potentiates BAX-Dependent Apoptosis. Neuron, 38, 899-914.
Putcha, G.V., Moulder, K.L., Golden, J.P., Bouillet, P., Adams, J.A., Strasser, A. and
Johnson, E.M. (2001) Induction of BIM, a proapoptotic BH3-only BCL-2 family
member, is critical for neuronal apoptosis. Neuron, 29, 615-628.
Puthalakath, H., Huang, D.C, O'Reilly, L.A., King, S.M. and Strasser, A. (1999) The
proapoptotic activity of the Bcl-2 family member Bim is regulated by interaction
with the dynein motor complex. Mol Cell, 3, 287-296.
83
Puthalakath, H., Villunger, A., O'Reilly, L.A., Beaumont, J.G., Coultas, L., Cheney, R.E.,
Huang, D.C and Strasser, A. (2001) Bmf: a proapoptotic BH3-only protein
regulated by interaction with the myosin V actin motor complex, activated by
anoikis. Science, 293, 1829-1832.
Radeke, M.J., Misko, T.P., Hsu, C , Herzenberg, L.A. and Shooter, E.M. (1987) Gene
transfer and molecular cloning of the rat nerve growth factor receptor. Nature,
325, 593-597.
Ramirez-Solis, R., Zheng, H., Whiting, J., Krumlauf, R. and Bradley, A. (1993) Hoxb-4
(Hox-2.6) mutant mice show homeotic transformation of a cervical vertebra and
defects in the closure of the sternal rudiments. Cell, 73, 279-294.
Rana, A., Gallo, K., Godowski, P., Hirai, S., Ohno, S., Zon, L., Kyriakis, J.M. and
Avruch, J. (1996) The mixed lineage kinase SPRK phosphorylates and activates
the stress-activated protein kinase activator, SEK-1. J Biol Chem, 271, 1902519028.
Robertson, A.G., Banfield, M.J., Allen, S.J., Dando, J.A., Mason, G.G., Tyler, S.J.,
Bennett, G.S., Brain, S.D., Clarke, A.R., Naylor, R.L., Wilcock, G.K., Brady,
R.L. and Dawbarn, D. (2001) Identification and structure of the nerve growth
factor binding site on TrkA. Biochem Biophys Res Commun, 282, 131-141.
Rodriguez-Tebar, A., Dechant, G. and Barde, Y.A. (1990) Binding of brain-derived
neurotrophic factor to the nerve growth factor receptor. Neuron, 4, 487-492.
Rodriguez-Tebar, A., Dechant, G., Gotz, R. and Barde, Y.A. (1992) Binding of
neurotrophin-3 to its neuronal receptors and interactions with nerve growth factor
and brain-derived neurotrophic factor. Embo J, 11, 917-922.
Roux, P.P. and Barker, P.A. (2002) Neurotrophin signaling through the p75 neurotrophin
receptor. Prog Neurobiol, 67, 203-233.
Roux, P.P., Bhakar, A.L., Kennedy, T.E. and Barker, P.A. (2001) The p75 neurotrophin
receptor activates Akt (protein kinase B) through a phosphatidylinositol 3-kinasedependent pathway. J Biol Chem, 276, 23097-23104.
Roux, P.P., Colicos, M.A., Barker, P.A. and Kennedy, T.E. (1999) p75 neurotrophin
receptor expression is induced in apoptotic neurons after seizure. / Neurosci, 19,
6887-6896.
84
Sakuma, H., Ikeda, A., Oka, S., Kozutsumi, Y., Zanetta, J.P. and Kawasaki, T. (1997)
Molecular cloning and functional expression of a cDNA encoding a new member
of mixed lineage protein kinase from human brain. J Biol Chem, 272, 2862228629.
Salehi, A.H., Xanthoudakis, S. and Barker, P.A. (2002) NRAGE, a p75 neurotrophin
receptor-interacting protein, induces caspase activation and cell death through a
JNK-dependent mitochondrial pathway. J Biol Chem, 277, 48043-48050.
Sasaki, T. and Takai, Y. (1998) The Rho small G protein family-Rho GDI system as a
temporal and spatial determinant for cytoskeletal control. Biochem Biophys Res
Commun, 245, 641-645.
Schmidt, A. and Hall, A. (2002) Guanine nucleotide exchange factors for Rho GTPases:
turning on the switch. Genes Dev, 16, 1587-1609.
Shamovsky, I.L., Ross, G.M., Riopelle, R.J. and Weaver, D.F. (1999) The interaction of
neurotrophins with the p75NTR common neurotrophin receptor: a comprehensive
molecular modeling study. Protein Sci, 8, 2223-2233.
Siegel, R.M., Frederiksen, J.K., Zacharias, D.A., Chan, F.K., Johnson, M., Lynch, D.,
Tsien, R.Y. and Lenardo, M.J. (2000) Fas preassociation required for apoptosis
signaling and dominant inhibition by pathogenic mutations. Science, 288, 23542357.
Snider, W.D. (1994) Functions of the neurotrophins during nervous system development:
what the knockouts are teaching us. Cell, 11, 627-638.
Soppet, D., Escandon, E., Maragos, J., Middlemas, D.S., Reid, S.W., Blair, J., Burton,
L.E., Stanton, B.R., Kaplan, D.R., Hunter, T. and et al. (1991) The neurotrophic
factors brain-derived neurotrophic factor and neurotrophin-3 are ligands for the
trkB tyrosine kinase receptor. Cell, 65, 895-903.
Squinto, S.P., Stitt, T.N., Aldrich, T.H., Davis, S., Bianco, S.M., Radziejewski, C , Glass,
D.J., Masiakowski, P., Furth, M.E., Valenzuela, D.M. and et al. (1991) trkB
encodes a functional receptor for brain-derived neurotrophic factor and
neurotrophin-3 but not nerve growth factor. Cell, 65, 885-893.
Strasser, A., O'Connor, L. and Dixit, V.M. (2000) Apoptosis signaling. Annu Rev
Biochem, 69, 217-245.
85
Stucky, CL. and Koltzenburg, M. (1997) The low-affinity neurotrophin receptor p75
regulates the function but not the selective survival of specific subpopulations of
sensory neurons. J Neurosci, 17, 4398-4405.
Syroid, D.E., Maycox, P.J., Soilu-Hanninen, M., Petratos, S., Bucci, T., Burrola, P.,
Murray, S., Cheema, S., Lee, K.F., Lemke, G. and Kilpatrick, T.J. (2000)
Induction of postnatal Schwann cell death by the low-affinity neurotrophin
receptor in vitro and after axotomy. J Neurosci, 20, 5741-5747.
Taniuchi, M., Johnson, E.M., Jr., Roach, P.J. and Lawrence, J.C, Jr. (1986)
Phosphorylation of nerve growth factor receptor proteins in sympathetic neurons
and PC 12 cells. In vitro phosphorylation by the cAMP-independent protein kinase
FA/GSK-3. J Biol Chem, 261, 13342-13349.
Tibbies, L.A., Ing, Y.L., Kiefer, F., Chan, J., Iscove, N., Woodgett, J.R. and Lassam, N.J.
(1996) MLK-3 activates the SAPK/JNK and p38/RK pathways via SEK1 and
MKK3/6. Embo J, 15, 7026-7035.
van Dam, H., Wilhelm, D., Herr, I., Steffen, A., Herrlich, P. and Angel, P. (1995) ATF-2
is preferentially activated by stress-activated protein kinases to mediate c-jun
induction in response to genotoxic agents. Embo J, 14, 1798-1811.
Verdi, J.M., Birren, S.J., Ibanez, C.F., Persson, H., Kaplan, D.R., Benedetti, M., Chao,
M.V. and Anderson, D.J. (1994) p75LNGFR regulates Trk signal transduction
and NGF-induced neuronal differentiation in MAH cells. Neuron, 12, 733-745.
von Schack, D., Casademunt, E., Schweigreiter, R., Meyer, M., Bibel, M. and Dechant,
G. (2001) Complete ablation of the neurotrophin receptor p75NTR causes defects
both in the nervous and the vascular system. Nat Neurosci, 4, 977-978.
Wallach, D., Boldin, M., Goncharov, T., Goltsev, Y., Mett, I., Malinin, N., Adar, R.,
Kovalenko, A. and Varfolomeev, E. (1996) Exploring cell death mechanisms by
analyzing signaling cascades of the TNF/NGF receptor family. Behring Inst Mitt,
144-155.
Walsh, G.S., Krol, K.M., Crutcher, K.A. and Kawaja, M.D. (1999a) Enhanced
neurotrophin-induced axon growth in myelinated portions of the CNS in mice
lacking the p75 neurotrophin receptor. J Neurosci, 19, 4155-4168.
86
Walsh, G.S., Krol, K.M. and Kawaja, M.D. (1999b) Absence of the p75 neurotrophin
receptor alters the pattern of sympathosensory sprouting in the trigeminal ganglia
of mice overexpressing nerve growth factor. J Neurosci, 19, 258-273.
Wang, K., Yin, X.M., Chao, D.T., Milliman, CL. and Korsmeyer, S.J. (1996) BID: a
novel BH3 domain-only death agonist. Genes Dev, 10, 2859-2869.
Wang, K.C, Kim, J.A., Sivasankaran, R., Segal, R. and He, Z. (2002) P75 interacts with
the Nogo receptor as a co-receptor for Nogo, MAG and OMgp. Nature, 420, 7478.
Wang, S., Bray, P., McCaffrey, T., March, K., Hempstead, B.L. and Kraemer, R. (2000)
p75(NTR) mediates neurotrophin-induced apoptosis of vascular smooth muscle
cells. Am J Pathol, 157, 1247-1258.
Wang, X., Bauer, J.H., Li, Y., Shao, Z., Zetoune, F.S., Cattaneo, E. and Vincenz, C.
(2001) Characterization of a p75(NTR) apoptotic signaling pathway using a novel
cellular model. J Biol Chem, 276, 33812-33820.
Whitfield, J., Neame, S.J., Paquet, L., Bernard, O. and Ham, J. (2001) Dominant-negative
c-Jun promotes neuronal survival by reducing BIM expression and inhibiting
mitochondrial cytochrome c release. Neuron, 29, 629-643.
Wiesmann, C , Ultsch, M.H., Bass, S.H. and de Vos, A.M. (1999) Crystal structure of
nerve growth factor in complex with the ligand-binding domain of the TrkA
receptor. Nature, 401, 184-188.
Wong, ST., Henley, J.R., Kanning, K.C, Huang, K.H., Bothwell, M. and Poo, M.M.
(2002) A p75(NTR) and Nogo receptor complex mediates repulsive signaling by
myelin-associated glycoprotein. Nat Neurosci, 5, 1302-1308.
Woolf, C.J. and Bloechlinger, S. (2002) Neuroscience. It takes more than two to Nogo.
Science, 297, 1132-1134.
Xu, Z., Maroney, A.C., Dobrzanski, P., Kukekov, N.V. and Greene, L.A. (2001) The
MLK family mediates c-Jun N-terminal kinase activation in neuronal apoptosis.
Mol Cell Biol, 21, 4713-4724.
Yaar, I. (1997) Computing normative ranges without recruiting normal subjects. Muscle
Nerve, 20, 1510-1514.
87
Yamashita, T., Higuchi, H. and Tohyama, M. (2002) The p75 receptor transduces the
signal from myelin-associated glycoprotein to Rho. J Cell Biol, 157, 565-570.
Yamashita, T. and Tohyama, M. (2003) The p75 receptor acts as a displacement factor
that releases Rho from Rho-GDI. Nat Neurosci, 6, 461-467.
Yamashita, T., Tucker, K.L. and Barde, Y.A. (1999) Neurotrophin binding to the p75
receptor modulates Rho activity and axonal outgrowth. Neuron, 24, 585-593.
Yan, H. and Chao, M.V. (1991) Disruption of cysteine-rich repeats of the p75 nerve
growth factor receptor leads to loss of ligand binding. J Biol Chem, 266, 1209912104.
Yoon, S.O., Casaccia-Bonnefil, P., Carter, B. and Chao, M.V. (1998) Competitive
signaling between TrkA and p75 nerve growth factor receptors determines cell
survival. J Neurosci, 18, 3273-3281.
Yu, J., Zhang, L., Hwang, P.M., Kinzler, K.W. and Vogelstein, B. (2001) PUMA induces
the rapid apoptosis of colorectal cancer cells. Mol Cell, 1, 673-682.
Zha, J., Harada, H., Osipov, K., Jockel, J., Waksman, G. and Korsmeyer, S.J. (1997) BH3
domain of BAD is required for heterodimerization with BCL-XL and proapoptotic activity. J Biol Chem, 272, 24101-24104.
Zha, J., Harada, H., Yang, E., Jockel, J. and Korsmeyer, S.J. (1996) Serine
phosphorylation of death agonist BAD in response to survival factor results in
binding to 14-3-3 not BCL-X(L). Cell, 87, 619-628.
Zha, J., Weiler, S., Oh, K.J., Wei, M.C. and Korsmeyer, S.J. (2000) Posttranslational Nmyristoylation of BID as a molecular switch for targeting mitochondria and
apoptosis. Science, 290, 1761-1765.
Zhang, W., Behringer, R.R. and Olson, E.N. (1995) Inactivation of the myogenic bHLH
gene MRF4 results in up-regulation of myogenin and rib anomalies. Genes Dev,
9, 1388-1399.
88
Appendix
89
Section Editor: Gary Westbrook
Senior Editor: David Ginty
Apoptosis Induced by p75NTR Requires Jun Kinase-dependent Phosphorylation of
Bad.
Asha L. Bhakar1, Jenny L. Howell1, Christine E. Paul1, Amir H. Salehi1, Esther B. E.
Becker2, Farid Said3, Azad Bonni2 and Philip A. Barker1.
'Centre for Neuronal Survival, Montreal Neurological Institute, McGill University, 3801
University Avenue, Montreal, Quebec, Canada, H3A 2B4.
department of Pathology, Harvard Medical School, Boston, Massachusetts, USA. 02115
3
Aegera Therapeutics Inc., 810 Golf Street, Montreal, Quebec Canada, H3E 1A8
Running title: p75NTR activates Bad to induce apoptosis
Number of text pages:
28
Number of words:
Abstract:
Introduction:
Discussion:
Number of figures:
163
498
1097
9
Correspondence:
Philip A. Barker
Montreal Neurological Institute
McGill University
3801 University Avenue
Montreal, Quebec, Canada, H3A 2B4
Phone: (514) 398-3064, Fax: (514) 398-5214
Email: [email protected]
ACKNOWLEDGEMENTS: We are grateful to David Kaplan and Mathieu Boudreau for providing the
MLK3 adenovirus, to Aviva Tolkovsky for providing the truncated-JIP1 adenovirus, to Ze'ev Ronai for
providing phospho-specific antibodies to Threonine 81 in p53, to Mohanish Deshmukh for advice on
cleaved Caspase-3 immunostaining and to Genevieve Dorval for technical assistance in titrating
adenovirus. This work was supported by grant MOP37850 from the Canadian Institute of Health Research
(CIHR; to PAB) and by NIH grant RO1-NS41021-01 (to AB). ALB was supported by a studentship from
the CIHR, JLH is supported by a Jean Timmons Costello Foundation studentship, CEP is supported by a
Natural Science and Engineering Research Centre studentship, AHS is supported by a National Cancer
Institute of Canada studentship, and EB is supported by the Boehringer Ingelheim Fonds. PAB is Scientist
of the Canadian Institute of Health Research.
Keywords: neurotrophin, BH3-domain only, JNK, trk, cell death
90
ABSTRACT
The p75 neurotrophin receptor (p75NTR), a member of the TNF receptor superfamily,
facilitates apoptosis during development and following injury to the central nervous
system. The signaling cascades activated by p75NTR that result in apoptosis remain
poorly understood. In this study, we show that activation of p75NTR in primary cortical
neurons, in PC 12 cells and in glioma cells results in activation of Jun kinase (JNK),
accumulation of Cytochrome C within the cytosol, and activation of Caspases 9, 6 and 3.
To link p75NTR-dependent JNK activation to mitochondrial Cytochrome C release,
regulation of BH3-domain-only family members was examined. Transcription of BH3domain-only family members was not induced by p75NTR but p75NTR-dependent JNK
activation resulted in phosphorylation and oligomerization of the BH3-domain-only
family member, Bad. Loss of function experiments using Bad dominant negatives or
RNA interference demonstrated a requirement for Bad in p75NTR-induced apoptosis.
Together, these studies provide the first data linking apoptosis induced by cell surface
receptor activation to the post-translational regulation of BH3-domain-only family
members.
91
INTRODUCTION
The four mammalian neurotrophins comprise a family of related growth factors required
for differentiation, survival, development, and death of specific populations of neurons
and non-neuronal cells. The effects of the neurotrophins are mediated by binding to cell
surface TrkA, TrkB and TrkC tyrosine-kinase receptors and to the p75 neurotrophin
receptor (p75NTR). Roles for Trk receptors in neurotrophin action in neuronal survival,
growth and synaptic modulation are now well established (Kaplan and Miller, 2000;
Patapoutian and Reichardt, 2001). The functions of the p75NTR receptor are complex
and have been more difficult to ascertain (Dechant and Barde, 2002; Roux and Barker,
2002). It is clear that p75NTR functions as a Trk co-receptor that increases neurotrophin
binding affinity (Barker and Shooter, 1994; Ryden et al., 1997; Esposito et al., 2001) and
recent studies suggest that it may be a critical element in a receptor complex that
responds to myelin-based growth inhibitory signals (Wang et al., 2002; Wong et al.,
2002) and regulates myelination (Cosgaya et al., 2002). p75NTR also has autonomous
signaling roles, particularly in facilitating apoptosis. In vitro analyses have shown that
p75NTR induces cell death in primary trigeminal (Davies et al., 1993), hippocampal
(Friedman, 2000; Brann et al., 2002), and sympathetic neurons (Lee et al., 1994; Bamji et
al., 1998), as well as retinal precursor (Frade et al., 1996; Frade and Barde, 1998),
Schwann (Soilu-Hanninen et al., 1999; Syroid et al., 2000; Petratos et al., 2003),
oligodendrocyte (Casaccia-Bonnefil et al., 1996; Yoon et al., 1998) and neuroblastoma
cells (Bunone et al., 1997). In vivo, p75NTR plays a prominent role in apoptosis that
occurs in glia and neurons following traumatic injury to the spinal cord (Casha et al.,
2001; Beattie et al., 2002) or brain (Roux et al., 1999; Troy et al., 2002) and has been
implicated in developmental apoptosis in somites, (Cotrina et al., 2000) retina and spinal
cord (Frade and Barde, 1999) and in the peripheral nervous system (Bamji et al., 1998).
The signaling events that link p75NTR activation to apoptosis are beginning to emerge
and p75NTR-dependent apoptosis is associated with an increase in Rac and Jun kinase
(JNK) activity and Caspase activation (Tournier et al., 2000; Harrington et al., 2002). The
precise ligand requirements for p75NTR apoptotic signaling are not clear but recent
studies have shown that unprocessed NGF (proNGF) is a more efficacious p75NTR
92
ligand than mature NGF (Lee et al., 2001; Beattie et al., 2002). A plethora of p75NTR
interacting proteins have been identified (Roux and Barker, 2002) and some of these,
including NRAGE (Salehi et al., 2000), NRIF (Casademunt et al., 1999) and NADE
(Mukai et al., 2000), facilitate p75NTR-dependent apoptosis. We have recently shown
that NRAGE activates a mitochondrial death pathway involving JNK-dependent
Cytochrome C release and the activation of Caspases (Salehi et al., 2002) but establishing
the precise roles of each of the cytosolic interactors of p75NTR remains a significant
challenge.
Despite this progress, several important questions remain unresolved. The proximal
elements that connect p75NTR to apoptotic pathways remain uncertain and it is not clear
whether JNK activation is a prerequisite for p75NTR-induced apoptosis in all responsive
cells. Further, the mechanisms employed by p75NTR to induce mitochondrial
Cytochrome C release and Caspase activation are unknown. In this report, we addressed
the mechanism of p75NTR-induced apoptosis in primary mouse cortical neurons and in
pheochromacytoma, glioma, neuroblastoma and medulloblastoma cells. Our findings
reveal that activated p75NTR invariably causes JNK activation, mitochondrial
Cytochrome C release and Caspase 9, 6 and 3 activation. We show that JNK activation is
necessary for p75NTR-dependent Caspase cleavage in all responsive cell types. To link
p75NTR-induced JNK activation to mitochondrial dysfunction, we examined the ability
of p75NTR to increase expression of BH3-domain-only proteins but found that p75NTR
did not activate transcription of BH3-domain-only genes. Instead, we demonstrate that
p75NTR activation results in JNK-dependent phosphorylation of the BH3-domain-only
protein Bad and show that Bad is required for p75NTR-induced apoptosis.
93
MATERIALS AND METHODS
Materials. Cell culture reagents were purchased from BioWhittaker, unless otherwise
indicated. The p75NTR antibody aPl and the phospho-Serl28 antibody have been
previously described (Roux et al., 1999). The phospho-Thr83 p53 antibody was a kind
gift of Ze'ev Ronai. The JNK1 antibody (C-17, cat# sc-474), the two Bad antibodies (C20, cat# sc-943 and N-19 cat# sc-6542) and the actin antibody (C-2, cat#sc-8432) were
purchased from Santa Cruz Biotechnology. Anti-Flag antibody (M2, cat# F-3165) was
obtained from Sigma, Cytochrome C antibody was purchased from Pharmingen (cat#
556433), b-galactosidase (LacZ) antibody was purchased from Promega (cat# 23781) and
anti-HA antibody (12CA5, cat# 1583816) was purchased from Roche. PhosphoThr183/Tyr185 JNK (G9, cat# 9255), phospho-Ser63 c-Jun (cat# 9261), phospho-Ser73 c-Jun
(cat# 9164S), c-Jun (cat# 9162), Caspase-9 (cat# 9502), cleaved Caspase-3 (Aspl75, cat#
9661), cleaved Caspase-6 (Aspl98; cat# 9761S), and cleaved PARP (Asp214; cat# 9541)
specific antibodies were obtained from Cell Signaling Technology. Horseradish
peroxidase-conjugated
secondary antibodies were purchased from
Jackson
ImmunoResearch Laboratories. Immunoreactive bands were detected using enhanced
chemiluminescence purchased from Perkin Elmer Life Sciences. All other reagents were
from Sigma, Calbiochem, or ICN Biochemicals, unless otherwise indicated.
Plasmids and recombinant adenovirus. Preparation of recombinant adenovirus
expressing enhanced green fluorescence protein (AdGFP), b-galactosidase (AdLacZ),
full-length p75NTR (Adp75NTR) , the Flag-tagged JNK-binding domain of JIP1
(AdJBD) and HA-epitope tagged MLK-3 (adMLK3) have been previously described
(Roux et al., 2002). All adenoviruses were amplified in 293A cells and purified on a
sucrose gradient, as previously described (Roux et al., 2002). Viruses were titered by
optical density and using the tissue culture infectious dose 50 (TCID) assay in 293A
cells. Titers are expressed in term of plaque forming units. The Bad dominant negative
plasmid consisting of GFP fused to a Bad nonapeptide in which Serl28 was substituted
by Ala and the parental GFP vector have both been previously described (Konishi et al.,
2002). The Bad RNAi construct was generated as previously described (Gaudilliere et al.,
2002).
94
Cell culture, infection and transfection. Human glioma (U343, U373, U87, and U251)
and medulloblastoma (UW228-1, UW228-3, UW228-3, and Daoy) cell lines were
provided by Dr. Roland Del Maestro (McGill University) and maintained in 5% C0 2 at
37° C in either Dulbecco's modified Eagle's medium (DMEM) or RPMI medium and
supplemented with 10% fetal calf serum (FCS, Clontech), 2 mM L-glutamine, 100 ug/ml
penicillin/streptomycin. Neuroblastoma cell lines (SY5Y, SKNAS, 15N, and NGP) were
provided by Dr. David Kaplan and maintained as above. The rat pheochromocytoma cell
line, PC 12, was maintained as previously described (Roux et al., 2001) and the PC12rtTA
cell line (PC 12) was purchased from Clontech and maintained in 10% C0 2 at 37° C in
DMEM supplemented with 10% FCS, 5% horse serum, 2 mM L-glutamine, 100 ug/ml
penicillin/streptomycin and 100 ug/ml G418. Cell lines were plated 18 to 24 hours prior
to transfection and typically harvested 24 to 48 hours after infection. Primary cortical
cultures were prepared from El4-16 CD1 mouse telencephalon as described previously
(Bhakar et al., 2002). Neuronal cultures were infected prior to plating and then
maintained in vitro for 2 days in Neurobasal media (Life Technologies) supplemented
with IX B27 supplement (Life Technologies), 2mM L-glutamine, and 100 ug/ml
penicillin/streptomycin. PC 12 cells were plated on poly-L-lysine coated plates and
transfected using Lipofectamine2000 as directed by the manufacturer (Invitrogen). Cells
lines were infected with adenovirus 24 hours after plating.
Cytochrome C release assay. Cytosol enriched subcellular fractions were prepared as
described in (Salehi et al., 2002). In brief, five million cells were harvested, washed once
in Tris-buffered saline (10 mM Tris (pH 8.0), 150 mM NaCl), once in Buffer A (100 mM
sucrose, 1 mM EGTA, 20 mM MOPS (pH 7.4)), and then resuspended in 500 ul Buffer B
(Buffer A plus 5% Percoll, 0.01% digitonin, 1 ug/ml aprotinin, 1 ug/ml leupeptin, 1
ug/ml pepstatin, 1 mM sodium orthovanadate, 1 mM phenylmethylsulfonyl fluoride). A
sample of this suspension was retained as total cell lysate. The remainder was incubated
on ice for 15 minutes and then centrifuged at 2500 g for 10 minutes to remove intact cells
and nuclei. The supernatant was then centrifuged at 15 000 g for 15 min to pellet
mitochondria. The final supernatant was designated cytosol.
95
Immunoblotting. Cells were lysed in RIPA buffer (10 mM Tris (pH 8.0), 150 mM NaCl,
1% Nonidet P-40, 0.5% deoxycholate, 0.1% SDS, 1 ug/ml Aprotinin, 1 ug/ml leupeptin,
1 ug/ml pepstatin, 1 mM sodium orthovanadate, 1 mM phenylmethylsulfonyl fluoride)
and analyzed for protein content using the BCA assay (Pierce). Samples were normalized
for protein content, suspended in Laemmli sample buffer, separated by SDSpolyacrylamide gel electrophoresis, and electroblotted onto nitrocellulose. Blocking and
secondary antibody incubations of immunoblots were performed in Tris-buffered
saline/Tween (10 mM Tris (pH 8.0), 150 mM NaCl, 0.2% Tween 20) supplemented with
5% (w/v) dried skim milk powder or 5% (w/v) bovine serum albumin (BSA) (Pierce). All
primary antibody incubations were performed in the blocking solution, except for those
involving phospho-specific antibodies which were performed in Tris-buffered
saline/Tween supplemented with 5% BSA. Immunoreactive bands were detected by
chemiluminescence (Perkin Elmer Life Sciences), according to the manufacturer's
instructions.
Survival assay. Analysis of cell survival was performed by MTT assay using 3-(4,5dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), which was added at a
final concentration of 1 mg/ml for the last four hours of a 48 hour infection. The reaction
was ended by the addition of one volume of solubilization buffer (20% SDS, 10%
dimethylformamide, and 20% acetic acid). After overnight solubilization, specific and
non-specific absorbencies were read at 570 and 690 nm, respectively. Each data point
was performed in triplicate or quadruplicate, and experimental results were analyzed by
multiple analyses of variance with statistical probabilities assigned using the Tukey test
for multiple comparisons. Each experiment was performed independently at least three
times.
RT-PCR. 450,000 U373 cells or primary cortical neurons were infected with virus and
24 hours later mRNA was isolated using the RNEasy Mini kit according to the
manufacturer's instructions (Qiagen). cDNA was generated using the Omniscript RT kit
96
(Qiagen) and random hexamers (Roche) as primers. PCR was performed for 30 cycles
using 300 nM of the following primers for U373 cells:
actin
hBIMEL
hBMF
hHrk/Dp5
hBIK
hPUMA
hNOXA
sense, 5'
CACCACTTTCTACAATGAGC
antisense,
5'
sense, 5'
TGGCAAAGCAACCTTCTGATG
antisense,
5'
sense, 5'
CTTGCTCTCTGCTGACCTGTTTG
antisense,
5'
sense, 5'
TCGGCAGGCGGAACTTGTAG
antisense,
5'
sense, 5'
AACCCCGAGATAGTGCTGGAAC
antisense,
5'
sense, 5'
ACTGTGAATCCTGTGCTCTGCC
antisense,
5'
sense, 5'
CCAAACTCTTCTGCTCAGGAACC
antisense,
5'
CGGTCAGGATCTTCATGAGG
AGTCGTAAGATAACCATTCGTGGG
AAGCCGATAGCCAGCATTGC
GCTGTATGTAAATAGCATTGGGGTG
GCTGGAAACCAACATTTTATTGAGC
ACCCCCCAAATGAATGCCAG
CGGTAATCTTCGGCAAAAACAC
For mouse cortical neurons, PCR was performed using the same conditions as above
using the following primers:
mBimEL
mBMF
mHrk/Dp5
mBIK
mNOXA
p75NTR
sense, 5'
CCCCTACCTCCCTACAGACAGAA
antisense,
5*
sense, 5'
CTTGCTCTCTGCTGACCTCTTTG
antisense,
5'
sense, 5'
TGGAAACACAGACAGAGGAAGCC
antisense,
5'
sense, 5'
TCACCAACCTCAGGGAAAACATC
antisense,
5'
sense, 5'
TGATGTGATGAGAGAAACGCTCG
antisense,
5'
sense, 5'
TGAATTCTGGAACAGCTGCAAAC
antisense,
5'
CCAGACGGAAGATAAAGCGTAACAG
GTTGCGTATGAAGCCGATGG
AAAGGAAAGGGACCACCACG
AGCAGGGGTCAAGAGAAGAAGG
AAAGCAATCCCAAACGACTGCC
CCTTAAGTCACACTGGGGATGTG
97
5% of the cDNA prepared was used in a 25ul PCR reaction and the reaction product was
separated on an 8% polyacrylamide gel, stained with ethidium bromide and visualized
under UV light.
Single Cell Caspase-3 Activation Assay. PC 12 cells were transfected with plasmids
encoding either GFP alone, GFP fused to a dominant interfering Bad nonapeptide, or
GFP plasmid and pU6/BS-Bad RNA interference plasmid at a 1:2 ratio. Cells were
infected with either adLacZ or with adp75NTR 48 hours after transfection and fixed 24
hours later using 4% paraformaldehyde (PFA) in PBS. Cells were blocked in TBS
supplemented with 5% donkey serum and 0.3% Triton-xlOO for 30 minutes and then
incubated for 18 hours at 4°C with control rabbit sera or with antibodies directed against
cleaved Caspase 3. Secondary antibodies (donkey anti-rabbit conjugated Cy3) and
Hoescht 33248 were applied for 2 hours at 4° C. GFP-positive cells were scored for the
presence of activated Caspase 3 by a blinded observer, with 300 cells counted per
condition. This experiment was repeated three times and the composite data was analyzed
for statistical significance by ANOVA (Tukey HDS multiple comparison).
98
RESULTS
The physiological conditions that result in activation of p75NTR apoptotic pathways are
complex and likely regulated by multiple ligands and co-receptors. We have previously
shown that recombinant adenovirus expressing full-length p75NTR or the p75NTR
intracellular domain efficiently induces apoptosis in the absence of added ligand (Roux et
al., 2001) and this approach was used to define apoptotic signaling pathways activated by
p75NTR. We began by testing cell lines and primary cell types for susceptibility to
p75NTR-induced death. Figure 1 shows that primary mouse cortical neurons, PC 12
pheochromacytoma cells and U343 and U373 glioma lines all showed reduced viability
when infected with adenovirus expressing p75NTR whereas infection with control
adenovirus expressing b-galactosidase (LacZ) had no significant effect. Other lines
tested, including other glioma lines (U251 and U87), various medulloblastoma lines
(Daoy, UW288-1, UW288-2, and UW288-3), and neuroblastoma lines (SY5Y, 15N,
NGP, and SKNAS) were resistant to p75NTR-induced apoptosis in this assay (data not
shown). For the remainder of this study, we focused our attention on p75NTR-dependent
apoptosis in primary mouse cortical neurons, rat PC 12 cells and human U343 and U373
glioma lines.
Activation of the extrinsic apoptotic pathway by death receptors that are structurally
related to p75NTR results in autocleavage and activation of Caspase 8. Activation of the
intrinsic apoptotic pathway results in release of mitochondrial contents and activation of
Caspase 9. We therefore determined the activation status of apical Caspases 8 and 9 and
effector Caspases 3 and 6 during p75NTR-induced apoptosis. Expression of p75NTR
resulted in a reduction in levels of full-length Caspase 9, a corresponding increase in
activated Caspase 9, Caspase 3, and Caspase 6 and accumulation of the cleaved form of
PARP, a Caspase 3 substrate (Figure 2A-B). In contrast, p75NTR-dependent Caspase 8
cleavage was not observed in any of the cell types examined (data not shown). p75NTRdependent Caspase activation was not due to adenoviral toxicity since cells infected with
comparable quantities of LacZ adenovirus did not exhibit Caspase activation. These data
indicate that p75NTR-induced apoptosis occurs primarily through an intrinsic death
pathway that involves release of mitochondrial contents and activation of Caspase 9.
99
Caspase 9 activation requires formation of an apoptosome complex consisting of
Caspase 9, Apaf-1 and cytosolic Cytochrome C. Release of Cytochrome C from
mitochondria into the cytosol is a key regulatory step in this process. To determine if
Cytochrome C is released during p75NTR-induced apoptosis, cells were left uninfected
or were infected with p75NTR or a control adenovirus, then lysed, subjected to
subcellular fractionation and cytosolic fractions were analyzed for Cytochrome C levels
by immunoblot. Figure 2C shows that Cytochrome C was not detected in the cytosol of
uninfected cells or in cells infected with control adenovirus whereas cytosolic
Cytochrome C was readily detected in the cytosol of cells expressing p75NTR. Thus,
p75NTR induces Cytochrome C release from mitochondria of multiple cell types.
Activation of the JNK pathway is an important regulator of apoptotic events in several
neuronal death paradigms and JNK can be activated by p75NTR in several cell types.
Consistent with this, we found that p75NTR expression in primary mouse cortical
neurons and in PC 12 and U373 cells consistently resulted in phosphorylation of JNK
(Figure 3A-B) or induced a dose-responsive increase in the phosphorylation of c-Jun, a
JNK target, (Figure 3C). These results indicate that 75NTR-induced JNK activation is a
consistent feature of a variety of p75NTR-responsive cell types.
To begin to address the role of the JNK pathway in p75NTR-induced apoptosis, we tested
the effect of CEP1347, a MAP3K inhibitor that exhibits anti-apoptotic effects in several
neuronal and non-neuronal systems (Saporito et al., 2002). We first tested the ability of
CEP 1347 to block c-Jun phosphorylation in PC 12 cells overexpressing MLK3, a MAP3K
identified as a target of CEP1347. Figure 4A shows that the compound almost completely
blocked the robust c-Jun phosphorylation induced by this kinase. We next examined
whether CEP 1347 reduced c-Jun phosphorylation or Caspase 3 activation which was
induced by p75NTR. CEP 1347 did indeed reduce p75NTR-dependent c-Jun
phosphorylation and Caspase 3 activation but only at high concentrations (500-1000 nM;
Figure 4B-C; data not shown). These findings indicate that reductions in MAP3K and
100
JNK signaling attenuates apoptosis induced by p75NTR yet suggest that blockade of a
non-preferred target of CEP 1347 is required for this effect.
To directly assess the role of JNK activity in p75NTR-induced death, an adenovirus
expressing the JNK binding domain of the JIP scaffolding molecule (AdJBD) was used to
inhibit JNK activity in vivo. This JIP fragment is believed to sequester JNK and thus acts
as an effective dominant inhibitor of JNK signaling (Harding et al., 2001). We first
confirmed that AdJBD is capable of blocking JNK-dependent target phosphorylation by
demonstrating that it blocked c-Jun phosphorylation induced by TNFa, a well
characterized JNK pathway inducer (Figure 5A). Subsequent studies established that
AdJBD was equally effective in blocking c-Jun phosphorylation induced by p75NTR
expression (Figure 5B). To determine if JNK inhibition blocked apoptotic signaling
induced by p75NTR, cells were infected with p75NTR in the absence or presence of
AdJBD and assessed for Caspase 3 activation. Expression of AdJBD effectively blocked
Caspase 3 activation in all responsive cell types, indicating a crucial role for JNK
activation in p75NTR-induced apoptosis (Figure 5C and data not shown).
These data demonstrate that JNK activation is a prerequisite for p75NTR-induced
apoptosis but substrates of JNK that play a role in p75NTR-induced apoptosis are
unknown. To begin to characterize targets of JNK involved in p75NTR-induced death,
we first compared c-Jun phosphorylation induced by p75NTR or MLK3, a potent inducer
of JNK activity (see above). Figure 6A shows that p75NTR and MLK3 induced robust
phosphorylation of JNK. However, there was considerable discordance between the JNK
activation, c-Jun phosphorylation and Caspase-3 activation induced by p75NTR versus
MLK3. p75NTR and MLK3 induced comparable JNK phosphorylation but only MLK3
produced a substantial increase in c-Jun phosphorylation whereas only p75NTR induced
substantial cleavage of Caspase 3. To determine if our experimental design may have
missed an early peak in p75NTR-induced c-Jun phosphorylation, JNK activation and cJun phosphorylation were examined at 12, 18, 24 and 30 hours after adenovirus infection.
Figure 6B shows that phosphorylated JNK was first detected 18 hours after p75NTR
infection and increased further by 24 and 30 hours. Cleaved Caspase 3 was detectable 24
101
hours after infection but c-Jun phosphorylation showed a significant lag, and an elevation
in phospho-Jun levels were detected only after 30 hours infection. These data show that
JNK activation correlates with p75NTR-induced death and suggests that c-Jun is not a
preferred substrate of the JNK complex which is activated by p75NTR.
BH3-domain-only proteins directly and indirectly induce the association of Bax and Bak,
which in turn facilitates release of mitochondrial proteins such as Cytochrome C into the
cytosol. Transcriptional activation of BH3-domain-only genes through c-Jun or p53
dependent pathways is important in apoptosis in several neuronal and non-neuronal
settings. We therefore examined whether p75NTR-induced apoptosis correlated with
accumulation of BH3-domain-only gene products. PC 12 and U373 cells and cortical
neurons were infected with LacZ or p75NTR adenovirus and alterations in mRNA levels
of the BH3-domain-only family members Bim, Bmf, Hrk, Bik, Puma, and Noxa were
determined by RT-PCR. mRNA corresponding to each of these family members were
readily detected in both cell types examined but p75NTR-dependent increases in their
levels were not detected (Figure 7 and data not shown). This indicates that JNK
activation induced by p75NTR does not induce transcription of BH3-domain-only genes
and suggests that alternate pathways are responsible for p75NTR-induced Cytochrome C
release and Caspase 3 activation.
BH3-domain-only proteins can, in some instances, be regulated by post-translational
mechanisms. Akt-dependent phosphorylation of Bad on Serl 12 and Serl36 allows it to
associate with 14-3-3 proteins and thereby suppresses its pro-apoptotic activity.
Apoptotic kinases including JNK directly activate the cell death machinery by
phosphorylating Bad at Serine 128 (Donovan et al., 2002). The phosphorylation of Bad at
this residue disrupts the interaction of Bad with 14-3-3 proteins thus allowing Bad to
induce apoptosis (Konishi et al., 2002). We therefore determined if p75NTR activation
resulted in phosphorylation of Bad on Serl28. PC 12 and U373 cells were infected with
LacZ or p75NTR adenovirus and alterations in Bad phosphostatus was examined by
immunoblot. Figures 8A and 8B show that p75NTR expression had little effect on the
levels or phosphostatus of monomeric Bad (~25 kD) but rather induced the accumulation
102
of a higher molecular weight species (~75 kD). This product was detected by two
antibodies directed against distinct epitopes in Bad (N19, C20) as well as by a phosphospecific antibody directed against the JNK phosphorylation site within Bad. The 75 kDa
product therefore appears to represent a stable oligomeric complex containing Bad
phosphorylated on Serine 128. To determine if JNK activity contributes to p75NTRdependent Bad phosphorylation and oligomerization, PC 12 cells were infected with
adenovirus expressing p75NTR in the absence or presence of AdJBD, lysed and
examined by Bad immunoblot. Figure 8C shows that inhibiting JNK activity with AdJBD
prevented formation of the Bad complex, indicating that JNK activity is required for
p75NTR-dependent Bad phosphorylation and oligomerization.
To determine if phosphorylation of Serine 128 within Bad is necessary for p75NTRinduced apoptosis, PC 12 cells were transfected with a dominant negative Bad serine 128
mutant allele (Konishi et al., 2002) and then infected with p75NTR or control virus. The
ability of the dominant negative Bad construct to inhibit p75NTR-dependent Caspase 3
activation was assessed after twenty-four hours of virus infection by scoring transfected
cells for the presence of cleaved Caspase 3. Figure 9 shows that expression of the
dominant negative Bad serine 128 mutant allele confers significant protection from
p75NTR-induced apoptosis, indicating that Bad phosphorylation is necessary for
p75NTR-induced apoptosis. To confirm that Caspase 3 cleavage induced by p75NTR
requires Bad, p75NTR-induced apoptosis was assessed in cells in which the endogenous
level of Bad were reduced using RNA interference. The ability of the RNAi construct to
reduce Bad levels was first validated in 293 cells (Supplementary Figure 1) and then used
to reduce Bad levels in PC 12 cells. PC 12 cells were transfected with GFP alone or with
GFP together with the Bad-RNAi plasmid and, 48 hours later, were infected with either
p75NTR or LacZ adenovirus for 24 hours. Figure 9 shows that PC 12 cells transfected
with Bad-RNAi are highly resistant to p75NTR-induced apoptosis, indicating a crucial
role for Bad in the p75NTR apoptotic pathway.
103
DISCUSSION
The mechanisms utilized by p75NTR to induce apoptosis are unique and bear little
similarity to cell death signaling pathways employed by other pro-apoptotic members of
the TNF receptor superfamily. In this report, we show that p75NTR-induced death
correlates with cytosolic accumulation of Cytochrome C and activation of Caspase 9 and
Caspase 3. Using the JNK binding domain of JIP as a dominant suppressor of JNK
activity, we show that JNK is required for p75NTR-induced Caspase 3 activation. Under
conditions in which p75NTR induces JNK phosphorylation and death, p75NTR does not
increase mRNA levels of BH3-domain-only family members that are transcriptionally
regulated by c-Jun or p53. Instead, we demonstrate that p75NTR specifically increases
phosphorylation and oligomerization of Bad and show that Bad plays a crucial role in
p75NTR-induced death.
Ligand binding to cell surface apoptotic receptors such as Fas and DR3 induces cell death
by initiating formation of a DISC complex that facilitates FADD-dependent Caspase 8
aggregation and activation. Other death stimuli induce apoptosis primarily via
Cytochrome C-dependent activation of Caspase 9 (Shi, 2002). Activation of Caspase 8
versus Caspase 9 is therefore a distinguishing regulatory event that provides insight into
the precise apoptotic pathways invoked by an extracellular stimulus. We have found that
in glioma cells, PC 12 cells and primary cortical neurons, p75NTR-induced apoptosis is
invariably accompanied by the activation of Caspase 9, Caspase 6, and Caspase 3.
p75NTR-dependent Caspase 8 activation was never observed. This suggests that
activation of the intrinsic death pathway is crucial for p75NTR-induced apoptosis and
indicates that cytosolic mitochondrial Cytochrome C accumulation is an important
regulatory step in p75NTR-induced death. These findings are in substantial agreement
with other studies examining p75NTR-dependent Caspase activation and are consistent
with a recent study showing that blockade of Caspase 9 activity significantly attenuates
p75NTR-induced apoptosis (Gu et al., 1999; Wang et al., 2001; Troy et al., 2002).
Together, these results show that p75NTR induces apoptosis through an intrinsic death
pathway that results in mitochondrial Cytochrome C release and Caspase 9 activation.
104
The JNK signaling cascade plays a crucial role in apoptosis induced by a variety of
stimuli (Kuranaga and Miura, 2002). We examined the role of JNK in p75NTR-induced
apoptotic signaling by expressing a fragment of the JIP scaffolding molecule that directly
binds to JNK and thus acts as a dominant JNK suppressor. This approach revealed that
JNK signaling is a critical prerequisite for p75NTR-dependent Caspase activation in all
cell types examined. We also report that CEP1347 reduces p75NTR-dependent death but
only at high concentrations, suggesting that inhibition of p75NTR-induced death by
CEP 1347 likely results from blockade of a non-preferred target distinct from MLK3.
Together with other recent studies (Friedman, 2000; Harrington et al., 2002), these data
therefore indicate a crucial role for JNK activation in p75NTR-induced apoptosis in all
cell types examined to date and raises the possibility that enzymes in the JNK pathway
may provide feasible targets for inhibiting p75NTR-induced apoptosis following
traumatic CNS injury.
BH3-domain-only family members inhibit the action of anti-apoptotic Bcl-2 family
members such as Bcl-2 and Bcl-xL and facilitate the action of Bax and Bak at the
mitochondria (Letai et al., 2002). The regulation of BH3-domain-only proteins is a key
step linking proximal signaling events to the induction of cell death (Huang and Strasser,
2000). In sympathetic neurons, JNK activation results in phosphorylation of c-Jun which
in turn results in transcription of the BH3-domain-only family members Bim and Hrk
(Harris and Johnson, 2001; Putcha et al., 2001; Whitfield et al., 2001). In other systems,
p53 activation results in transcription of Noxa and Puma, also pro-apoptotic BH3domain-only family members (Wu and Deng, 2002). We therefore hypothesized that
p75NTR-dependent apoptosis was associated with transcription of known BH3-domainonly family members. However, p75NTR does not appear to enhance transcription of
BH3-domain-only family members, suggesting that alternative pathways are responsible.
BH3-domain-only family members are present in normal cells in the absence of apoptotic
stimuli and must be rendered inactive to prevent apoptosis. One mechanism that
accomplishes this is their sequestration through protein-protein interactions. For example,
the BH3-domain-only protein Bad is bound to 14-3-3 (Zha et al., 1996; Datta et al., 2000)
105
and Bim and Bmf can be sequestered in the cytosol by binding to dynein light chain or
myosin V (Puthalakath et al., 1999; Puthalakath et al., 2001). Significantly, recent
findings have revealed that the sequestration of these three BH3-domain-only proteins
can be negatively regulated by JNK. UV irradiation of HEK293 cells results in JNKdependent phosphorylation of Bmf and Bim, releasing these proteins from their
sequestration and allowing them to contribute to the apoptotic cascade (Lei and Davis,
2003). The Serine 128 phosphorylation of BAD activates BAD specifically by inhibiting
the interaction of Serine 136-phosphorylated BAD with 14-3-3 proteins (Konishi et al.
2002). Serine 136 is a target of survival factor-induced kinases including Akt in neurons.
That p75NTR induces cell death in part by inducing the phosphorylation of BAD at
Serine 128 suggests that p75NTR promotes apoptosis by opposing survival factor signals
that suppress the cell death machinery. Further, p75NTR activation results in the
oligomerization of Bad through a JNK-dependent pathway. Aside from Bad itself, the
components of this stable oligomeric complex remain unknown but may include antiapoptotic proteins such as Bcl-2 and Bcl-Xl (Letai et al., 2002). These findings provide
the first data linking cell surface receptor activation to the post-translational regulation of
BH3-domain-only family members and indicate that p75NTR regulates apoptosis through
a JNK pathway that is independent of transcription.
Palmada et al (2002) have recently found that c-Jun is not required for p75NTR-induced
cell death. Consistent with this, our data show that levels of c-Jun phosphorylation
induced by p75NTR are modest and do not induce transcription of c-Jun targets that
include Bim and Hrk. Thus, although c-Jun phosphorylation is a useful surrogate to
assess JNK activation, it does not appear to play a significant role in p75NTR-induced
apoptosis. However, alternative JNK-dependent pathways may contribute to p75NTRdependent apoptosis. One candidate pathway involves p53, which can be activated by
direct JNK phosphorylation and has been implicated in p75NTR-induced apoptosis in
one study (Aloyz et al., 1998). However, and p75NTR readily induces apoptosis in cells
lacking functional p53 (eg. U373 cells, Figure 1) and phosphospecific antibodies directed
against Thr 81, a JNK target residue in p53 (Buschmann et al., 2001), or against Serl5 or
Ser20 (Dumaz et al., 2001) did not reveal significant p75NTR-dependent phosphorylation
106
of p53 (data not shown). Nonetheless, we cannot rule out the possibility that p53 or
related family members may play a role in p75NTR-induced apoptosis in specific
circumstances. Further examination of transcriptional pathways in p75NTR action is
warranted.
p75NTR plays a prominent role in nervous system apoptosis, particularly following
trauma, and a detailed picture of the pro-apoptotic signal transduction mechanisms
activated by the receptor is required. In this study, we show that p75NTR-dependent JNK
activation is invariably required for Caspase activation and find that p75NTR-dependent
JNK activation induces phosphorylation and activation of the BH3-domain-only protein
Bad and that Bad is required for p75NTR-induced apoptosis.
107
REFERENCES
Aloyz RS, Bamji SX, Pozniak CD, Toma JG, Atwal J, Kaplan DR, Miller FD (1998) p53
is essential for developmental neuron death as regulated by the TrkA and p75
neurotrophin receptors. J Cell Biol 143:1691-1703.
Bamji SX, Majdan M, Pozniak CD, Belliveau DJ, Aloyz R, Kohn J, Causing CG, Miller
FD (1998) The p75 neurotrophin receptor mediates neuronal apoptosis and is
essential for naturally occurring sympathetic neuron death. J Cell Biol 140:911923.
Barker PA, Shooter EM (1994) Disruption of NGF binding to the low affinity
neurotrophin receptor p75LNTR reduces NGF binding to trkA on PC 12 cells.
Neuron 13:203-215.
Beattie MS, Harrington AW, Lee R, Kim JY, Boyce SL, Longo FM, Bresnahan JC,
Hempstead BL, Yoon SO (2002) ProNGF induces p75-mediated death of
oligodendrocytes following spinal cord injury. Neuron 36:375-386.
Bhakar AL, Tannis LL, Zeindler C, Russo MP, Jobin C, Park DS, MacPherson S, Barker
PA (2002) Constitutive nuclear factor-kappa B activity is required for central
neuron survival. J Neurosci 22:8466-8475.
Brann AB, Tcherpakov M, Williams IM, Futerman AH, Fainzilber M (2002) Nerve
growth factor-induced p75-mediated death of cultured hippocampal neurons is
age-dependent and transduced through ceramide generated by neutral
sphingomyelinase. J Biol Chem 277:9812-9818.
Bunone G, Mariotti A, Compagni A, Morandi E, Delia VG (1997) Induction of apoptosis
by p75 neurotrophin receptor in human neuroblastoma cells. Oncogene 14:14631470.
Buschmann T, Potapova O, Bar-Shira A, Ivanov VN, Fuchs SY, Henderson S, Fried VA,
Minamoto T, Alarcon-Vargas D, Pincus MR, Gaarde WA, Holbrook NJ, Shiloh
Y, Ronai Z (2001) Jun NH2-terminal kinase phosphorylation of p53 on Thr-81 is
important for p53 stabilization and transcriptional activities in response to stress.
Mol Cell Biol 21:2743-2754.
Casaccia-Bonnefil P, Carter BD, Dobrowsky RT, Chao MV (1996) Death of
oligodendrocytes mediated by the interaction of nerve growth factor with its
receptor p75. Nature 383:716-719.
Casademunt E, Carter BD, Benzel I, Frade JM, Dechant G, Barde YA (1999) The zinc
finger protein NRIF interacts with the neurotrophin receptor p75(NTR) and
participates in programmed cell death. Embo J 18:6050-6061.
Casha S, Yu WR, Fehlings MG (2001) Oligodendroglial apoptosis occurs along
degenerating axons and is associated with FAS and p75 expression following
spinal cord injury in the rat. Neuroscience 103:203-218.
Cosgaya JM, Chan JR, Shooter EM (2002) The neurotrophin receptor p75NTR as a
positive modulator of myelination. Science 298:1245-1248.
Cotrina ML, Gonzalez-Hoyuela M, Barbas J A, Rodriguez-Tebar A (2000) Programmed
cell death in the developing somites is promoted by nerve growth factor via its
p75(NTR) receptor. Dev Biol 228:326-336.
Datta SR, Katsov A, Hu L, Petros A, Fesik SW, Yaffe MB, Greenberg ME (2000) 14-3-3
proteins and survival kinases cooperate to inactivate BAD by BH3 domain
phosphorylation. Mol Cell 6:41-51.
108
Davies AM, Lee KF, Jaenisch R (1993) p75-deficient trigeminal sensory neurons have an
altered response to NGF but not to other neurotrophins. Neuron 11:565-574.
Dechant G, Barde YA (2002) The neurotrophin receptor p75(NTR): novel functions and
implications for diseases of the nervous system. Nat Neurosci 5:1131-1136.
Donovan N, Becker EB, Konishi Y, Bonni A (2002) JNK phosphorylation and activation
of BAD couples the stress-activated signaling pathway to the cell death
machinery. J Biol Chem 277:40944-40949.
Dumaz N, Milne DM, Jardine LJ, Meek DW (2001) Critical roles for the serine 20, but
not the serine 15, phosphorylation site and for the polyproline domain in
regulating p53 turnover. Biochem J 359:459-464.
Esposito D, Patel P, Stephens RM, Perez P, Chao MV, Kaplan DR, Hempstead BL
(2001) The cytoplasmic and transmembrane domains of the p75 and Trk A
receptors regulate high affinity binding to nerve growth factor. J Biol Chem
276:32687-32695.
Frade JM, Barde YA (1998) Microglia-derived nerve growth factor causes cell death in
the developing retina. Neuron 20:35-41.
Frade JM, Barde YA (1999) Genetic evidence for cell death mediated by nerve growth
factor and the neurotrophin receptor p75 in the developing mouse retina and
spinal cord. Development 126:683-690.
Frade JM, Rodriguez-Tebar A, Barde YA (1996) Induction of cell death by endogenous
nerve growth factor through its p75 receptor. Nature 383:166-168.
Friedman WJ (2000) Neurotrophins induce death of hippocampal neurons via the p75
receptor. J Neurosci 20:6340-6346.
Gaudilliere B, Shi Y, Bonni A (2002) RNA interference reveals a requirement for
myocyte enhancer factor 2A in activity-dependent neuronal survival. J Biol Chem
277:46442-46446.
Gu C, Casaccia-Bonnefil P, Srinivasan A, Chao MV (1999) Oligodendrocyte apoptosis
mediated by caspase activation. J Neurosci 19:3043-3049.
Harding TC, Xue L, Bienemann A, Haywood D, Dickens M, Tolkovsky AM, Uney JB
(2001) Inhibition of JNK by overexpression of the JNL binding domain of JIP-1
prevents apoptosis in sympathetic neurons. J Biol Chem 276:4531-4534.
Harrington AW, Kim JY, Yoon SO (2002) Activation of Rac GTPase by p75 is necessary
for c-jun N-terminal kinase-mediated apoptosis. J Neurosci 22:156-166.
Harris CA, Johnson EM, Jr. (2001) BH3-only Bcl-2 family members are coordinately
regulated by the JNK pathway and require Bax to induce apoptosis in neurons. J
Biol Chem 276:37754-37760.
Huang DC, Strasser A (2000) BH3-Only proteins-essential initiators of apoptotic cell
death. Cell 103:839-842.
Kaplan DR, Miller FD (2000) Neurotrophin signal transduction in the nervous system.
Curr Opin Neurobiol 10:381-391.
Konishi Y, Lehtinen M, Donovan N, Bonni A (2002) Cdc2 phosphorylation of BAD
links the cell cycle to the cell death machinery. Mol Cell 9:1005-1016.
Kuranaga E, Miura M (2002) Molecular genetic control of caspases and JNK-mediated
neural cell death. Arch Histol Cytol 65:291-300.
109
Lee K-F, Davies AM, Jaenisch R (1994) p75-deficient embryonic dorsal root sensory and
neonatal sympathetic neurons display a decreased sensitivity to NGF.
Development 120:1027-1033.
Lee R, Kermani P, Teng KK, Hempstead BL (2001) Regulation of cell survival by
secreted proneurotrophins. Science 294:1945-1948.
Lei K, Davis RJ (2003) JNK phosphorylation of Bim-related members of the Bcl2 family
induces Bax-dependent apoptosis. Proc Natl Acad Sci U S A 100:2432-2437.
Letai A, Bassik MC, Walensky LD, Sorcinelli MD, Weiler S, Korsmeyer SJ (2002)
Distinct BH3 domains either sensitize or activate mitochondrial apoptosis, serving
as prototype cancer therapeutics. Cancer Cell 2:183-192.
Mukai J, Hachiya T, Shoji-Hoshino S, Kimura MT, Nadano D, Suvanto P, Hanaoka T, Li
Y, Irie S, Greene LA, Sato TA (2000) NADE, a p75NTR-associated cell death
executor, is involved in signal transduction mediated by the common
neurotrophin receptor p75NTR. J Biol Chem 275:17566-17570.
Patapoutian A, Reichardt LF (2001) Trk receptors: mediators of neurotrophin action.
Curr Opin Neurobiol 11:272-280.
Petratos S, Butzkueven H, Shipham K, Cooper H, Bucci T, Reid K, Lopes E, Emery B,
Cheema SS, Kilpatrick TJ (2003) Schwann cell apoptosis in the postnatal
axotomized sciatic nerve is mediated via NGF through the low-affinity
neurotrophin receptor. J Neuropathol Exp Neurol 62:398-411.
Putcha GV, Moulder KL, Golden JP, Bouillet P, Adams JA, Strasser A, Johnson EM
(2001) Induction of BIM, a proapoptotic BH3-only BCL-2 family member, is
critical for neuronal apoptosis. Neuron 29:615-628.
Puthalakath H, Huang DC, O'Reilly LA, King SM, Strasser A (1999) The proapoptotic
activity of the Bcl-2 family member Bim is regulated by interaction with the
dynein motor complex. Mol Cell 3:287-296.
Puthalakath H, Villunger A, O'Reilly LA, Beaumont JG, Coultas L, Cheney RE, Huang
DC, Strasser A (2001) Bmf: a proapoptotic BH3-only protein regulated by
interaction with the myosin V actin motor complex, activated by anoikis. Science
293:1829-1832.
Roux PP, Barker PA (2002) Neurotrophin signaling through the p75 neurotrophin
receptor. Prog Neurobiol 67:203-233.
Roux PP, Colicos MA, Barker PA, Kennedy TE (1999) p75 neurotrophin receptor
expression is induced in apoptotic neurons after seizure. J Neurosci 19:68876896.
Roux PP, Bhakar AL, Kennedy TE, Barker PA (2001) The p75 neurotrophin receptor
activates Akt (protein kinase B) through a phosphatidylinositol 3-kinasedependent pathway. J Biol Chem 276:23097-23104.
Roux PP, Dorval G, Boudreau M, Angers-Loustau A, Morris SJ, Makkerh J, Barker PA
(2002) K252a and CEP1347 are neuroprotective compounds that inhibit mixedlineage kinase-3 and induce activation of Akt and ERK. J Biol Chem 277:4947349480.
Ryden M, Hempstead B, Ibanez CF (1997) Differential modulation of neuron survival
during development by nerve growth factor binding to the p75 neurotrophin
receptor. J Biol Chem 272:16322-16328.
110
Salehi AH, Xanthoudakis S, Barker PA (2002) NRAGE, a p75 neurotrophin receptorinteracting protein, induces caspase activation and cell death through a JNKdependent mitochondrial pathway. J Biol Chem 277:48043-48050.
Salehi AH, Roux PP, Kubu CJ, Zeindler C, Bhakar A, Tannis LL, Verdi JM, Barker PA
(2000) NRAGE, a novel MAGE protein, interacts with the p75 neurotrophin
receptor and facilitates nerve growth factor-dependent apoptosis. Neuron:279288.
Saporito MS, Hudkins RL, Maroney AC (2002) Discovery of CEP-1347/KT-7515, an
inhibitor of the JNK/SAPK pathway for the treatment of neurodegenerative
diseases. Prog Med Chem 40:23-62.
Shi Y (2002) Mechanisms of caspase activation and inhibition during apoptosis. Mol Cell
9:459-470.
Soilu-Hanninen M, Ekert P, Bucci T, Syroid D, Bartlett PF, Kilpatrick TJ (1999) Nerve
growth factor signaling through p75 induces apoptosis in Schwann cells via a Bcl2-independent pathway. J Neurosci 19:4828-4838.
Syroid DE, Maycox PJ, Soilu-Hanninen M, Petratos S, Bucci T, Burrola P, Murray S,
Cheema S, Lee KF, Lemke G, Kilpatrick TJ (2000) Induction of postnatal
Schwann cell death by the low-affinity neurotrophin receptor in vitro and after
axotomy. J Neurosci 20:5741-5747.
Tournier C, Hess P, Yang DD, Xu J, Turner TK, Nimnual A, Bar-Sagi D, Jones SN,
Flavell RA, Davis RJ (2000) Requirement of JNK for stress-induced activation of
the cytochrome c- mediated death pathway. Science 288:870-874.
Troy CM, Friedman JE, Friedman WJ (2002) Mechanisms of p75-mediated death of
Hippocampal neurons: Role of caspases. J Biol Chem 3:3.
Wang KC, Kim JA, Sivasankaran R, Segal R, He Z (2002) P75 interacts with the Nogo
receptor as a co-receptor for Nogo, MAG and OMgp. Nature 420:74-78.
Wang X, Bauer JH, Li Y, Shao Z, Zetoune FS, Cattaneo E, Vincenz C (2001)
Characterization of a p75NTR apoptotic signaling pathway using a novel cellular
model. J Biol Chem 12:12.
Whitfield J, Neame SJ, Paquet L, Bernard O, Ham J (2001) Dominant-negative c-Jun
promotes neuronal survival by reducing BIM expression and inhibiting
mitochondrial cytochrome c release. Neuron 29:629-643.
Wong ST, Henley JR, Kanning KC, Huang KH, Bothwell M, Poo MM (2002) A
p75(NTR) and Nogo receptor complex mediates repulsive signaling by myelinassociated glycoprotein. Nat Neurosci 5:1302-1308.
Wu X, Deng Y (2002) Bax and BH3-domain-only proteins in p53-mediated apoptosis.
Front Biosci7:dl51-156.
Yoon SO, Casaccia-Bonnefil P, Carter B, Chao MV (1998) Competitive signaling
between TrkA and p75 nerve growth factor receptors determines cell survival. J
Neurosci 18:3273-3281.
Zha J, Harada H, Yang E, Jockel J, Korsmeyer SJ (1996) Serine phosphorylation of death
agonist BAD in response to survival factor results in binding to 14-3-3 not BCLX(L). Cell 87:619-628.
Ill
FIGURE LEGENDS
Figure 1. Overexpression of p75NTR induces cell death in a variety of cell types. (A)
cortical neurons, (B) PC 12, (C) U343 (wild type p53), and (D) U373 (mutant p53) cells
were infected with increasing multiplicities of infection (MOI) of LacZ or p75NTR
recombinant adenovirus and then analyzed for survival by the MTT assay (see Materials
and Methods). Error bars indicate SD. Results were analyzed for statistical significance
by ANOVA (Tukey HSD multiple comparison). Statistically significant differences of
p<0.001 are indicated by an asterisk.
Figure 2. p75NTR activates Caspases and induces accumulation of cytosolic
Cytochrome C. (A) Cortical neurons infected with 10, 50, or 100 MOI of LacZ or
p75NTR recombinant adenovirus were lysed and analyzed by immunoblot for levels of
LacZ, p75NTR and full-length Caspase 9 protein or, using cleavage-specific antibodies,
for levels of cleaved Caspases 3 and 6 and cleaved Poly(ADP-ribose) polymerase
(PARP). (B) U373 cells were infected with 50 or 100 MOI of either LacZ or p75NTR
adenovirus for 48 hours, or treated with etoposide 50 uM (+), and then lysed and
analyzed for increases in cleaved Caspase 9. (C) E15 cortical neurons, U373, and PC12
cells were left uninfected (0) or were infected with 100 MOI of LacZ (Lz), or p75NTR
(p75) recombinant adenovirus. 30 hours later cells were fractionated for cytosolic
components as described in "Materials and Methods". Cytosolic fractions normalized for
protein content were analyzed by immunoblotting with an antibody directed against
Cytochrome C.
Figure 3. p75NTR activates the JNK pathway. (A) U373 cells were infected with 0,
50, 100, or 200 MOI of control AdLacZ or with Adp75NTR, (B) PC12 cells were
injected with 0 or 50 MOI of AdLacZ or Adp75NTR, and (C) cortical neurons were
infected with 10, 50, or 150 MOI of AdLacZ or Adp75NTR. Lysates were prepared 3048 hours after infection and examined by immunoblot for LacZ, p75NTR,
phosphorylated JNK (pJNK), total JNK, phosphorylated c-Jun (pJun) and total c-Jun as
indicated.
112
Figure 4. Inhibition of MAP3K signaling attenuates apoptosis induced by p75NTR.
(A) U373 cells infected with 100 MOI of MLK3 adenovirus or left uninfected (0), were
treated 47 hours later with DMSO or CEP 1347 at 200 nM for 1 hour. Cells were
harvested and lysates subjected to immunoblot analysis for phospho-Ser63 c-Jun (pJun)
and total c-Jun protein. (B) Cortical neurons infected with 50 MOI of LacZ or p75NTR
adenovirus were treated with DMSO or 50, 200, or 500 nM CEP1347 for 1 hour as in
(A). Lysates were analyzed by immunoblot as indicated (pJun, c-Jun, LacZ, p75NTR).
(C) AdLacZ or Adp75NTR-infected cortical neurons were treated with 500 nM of
CEP 1347 [C] or DMSO [D] at the time of infection and lysates were prepared 48 hours
later and analyzed by immunoblot for levels of p75NTR, LacZ, phospho-Ser63 c-Jun
(pJun), and cleaved Caspase 3 (cl. Caspase 3).
Figure 5. Activation of the JNK pathway is required for p75NTR-mediated Caspase
activation. Immunoblots for phospho-Ser63 c-Jun (pJun), c-Jun, Flag-JIP, LacZ, p75NTR,
phospho-Thr183/Tyrl85-JNK (pJNK), JNK, and cleaved Caspase 3 were performed as
indicated on lysates from (A) U373 cells treated with TNF 20ng/ml that were either left
uninfected (0) or infected with JBD-JIP adenovirus (JBD) at 10 MOI, (B) cortical
neurons infected with 50 MOI of LacZ or p75NTR adenovirus together with increasing
amounts (0, 0.05, 0.5, 2.5 MOI) of JBD-JIP adenovirus, and (C) PC12 cells infected with
50 MOI of LacZ or p75NTR adenovirus supplemented with LacZ or JBD-JIP (JBD)
adenovirus (both at 5 MOI).
Figure 6. p75NTR-induced Caspase 3 cleavage does not correlate with
phosphorylation of c-Jun. PC 12 cells were infected with 50 MOI of AdLacZ (Lz),
Adp75NTR (p75), or AdMLK3 (MLK) recombinant adenovirus and lysates were
prepared at 30 hours post-infection (A and C), or at 12, 18, 24, and 30 hours postinfection (B). Lysates normalized for protein content were analyzed for LacZ, p75NTR,
cleaved Caspase 3, phospho-Thr183/Tyr185-JNK (pJNK), total JNK, phospho-Ser63 c-Jun
(pJun) and total c-Jun protein levels by immunoblot as indicated.
113
Figure 7. p75NTR does not transcriptionally regulate BH3-domain-only proteins.
Cortical neurons were infected with 0, 50, or 200 MOI of LacZ or p75NTR (p75)
adenovirus and 24 hours later mRNA was isolated as described in Materials and
Methods. RT-PCR was performed using primers directed against Bim, Bmf, Hrk, Bik,
Puma, Noxa, p75NTR and Actin as indicated.
Figure 8. p75NTR activates JNK-dependent phosphorylation and oligomerization of
Bad. (A) U373 cells were infected with 0, 50, 100, or 200 MOI of LacZ or p75NTR
adenovirus and lysates were analyzed by immunoblot for LacZ, p75NTR, phospho-Ser128
Bad, and Bad (C-20 - shown; N19 - data not shown). (B) PC 12 cells were left uninfected
(0) or were infected with LacZ (Lz) or p75NTR (p75) adenovirus aqt 100 MOI and
lysates were analyzed by immunoblot for LacZ, p75NTR, phospho-Ser128 Bad, and Bad
(C-20). (C) PC 12 cells were infected with nothing (0), LacZ (Lz), or p75NTR (p75)
adenovirus together with either 5 MOI of LacZ or JBD-JIP (JBD) adenovirus. Lysates
were compared for expression of Bad, cleaved Caspase 3, LacZ, p75NTR, and Flag-JIP
(Flag) by immunoblot as indicated.
Figure 9. Bad is required for p75NTR-induced apoptosis. PC 12 cells were transfected
with GFP plasmid alone or with GFP plasmid together with plasmids encoding DN-Bad
(SI28A) or expressing Bad-RNAi. Cells were infected 48 hours later with LacZ or
p75NTR adenovirus and, at 24 hours post-infection, were fixed and immunostained for
cleaved Caspase 3 as described in Materials and Methods. Transfected cells were scored
for Caspase 3 cleavage by a blind observer (n =300 cells/condition). '**' indicates a
difference of p<0.001 between GFP/Mock (Bar 1) and GFP/p75NTR (Bar 5) and '*'
indicates a difference of p<0.001 between GFP/p75NTR (Bar 5) and both DNBad/p75NTR (Bar 6) and with Bad RNAi/p75NTR (Bar 7), indicated by ANOVA.
114
Cortical Neurons
0.045
0.035
w
wo
< en
0.025
LacZ
.p75NTR
£ in
0.015
li
0.005
0
50
250
B
PC12
0.8
>.
I 8 0.6
>
o
; in
LacZ
.p75NTR
£ m 0.4
t~
*
0.2
I i
0
50
100
U343
%i 1
I1
1l j
s
0.5
oo
*
< cr> 0.4
l£>
to
> O
0.3
£ in
w
Q
0.2
I
:*
i
0.1
0
D
i
0
10
.p75NTR
1
JUL
50
100
250
U373
1.6
to
IA
„
IB o
<
0>
s <°
56
1.4
1.2
1
n
•
£ m 0.8
0.6
;
0.4
Figure 1
Bhakar et al.
I, 1
I
0.2
0
LacZ
.p75NTR
*
10
50
100
*
250
115
LacZ
10
50
p75NTR
100
10
50
100
LacZ
mm% p75NTR
mM
Caspase 9
~m*
CI Caspase 3
CI PARP
—
B
LacZ
0
p75NTR
50
100
50
TSF
*^
Z^t
M
100
Cortical neurons
mm PC12
—
Bhakar et. al.
+
Lz p75
—
Figure 2
CI Caspase 6
U373
116
A
B
LacZ
0
p75NTR
C
rj
50 100200 50 100200
Lz p75
JH
mm
«•
m mmmm
-
Figure 3
Bhakar et al.
-r
JNK
*
• » • » • •
LacZ
-,
LacZ
10
50
150
p75NTR
10
50
150
P75NTR
pJNK
JNK
- • -
mm,
— —
—
P75NTR
pJun
117
MLK3
0
DMSO CEP
C-Jun
« M » W
B
LacZ
DMSO
500
50
CEP
200
p75NTR
DMSO
500 500
50
m
CEP
200 500
pJun
-
c-Jun
,.,
LacZ
^^^m
ymmim
•^"*
^^^
p75NTR
LacZ p75NTR
C D C D C
—
pJun
—
CI Caspase 3
LacZ
fcaE~-
Figure 4
Bhakar et al.
— *•
118
0
TNF
0 10 JBD
—mm
pJun
Flag-JBD
B
LacZ
0
p75NTR
0 0.05 0.5 2.5 0 0.05 0.5 2.5 JBD
Sm —
pJun
Jun
LacZ
p75NTR
Flag-JBD
LacZ
JBD
Lz p75 Lz p75
0
*•»
pJNK
JNK
pJun
CI Caspase 3
w «
LacZ
p75NTR
Flag-JBD
Figure 5
Bhakar et al.
119
Lz p75 MLK
a s
pJNK
JNK
pJun
mm - ~
CI Caspase 3
B
12
18
24
Lz p75 Lz p75 Lz p75
«•
I
•
"""*•
<•»•
30
hours
Lz p75
« M
m^-IB
LacZ
P75NTR
- — C I Caspase 3
*""* ~ " «2! PJNK
— pJun
Figure 6
Bhakar et al.
120
LacZ
p75
0 50 200 50 200
Figure 7
Bhakar et al
121
B
LacZ
0
p75NTR
0
50 100 200 50 100 200
—
Lz p75
mm
LacZ
LacZ
Wmf H
•~m~
—
•
- -•
—
«l m m
p75NTR
LacZ
JBD
0 Lz p75 Lz p75
Bad
•
"
*
•
CI Caspase 3
pS128-Bad
m
- * - pS 128-Bad
LacZ
p75NTR
Bad
- * - Bad
Flag-JBD
Figure 8
Bhakar et al.
122
GFP
Mock
Figure 9
Bhakar et al.
GFP
DN-Bad
LacZ
Bad RNAi
GFP
DN-Bad
p75NTR
Bad RNAi
123
Supplementary Figure 1
BAD
U6
US-Ri
Bad
Actin
Validation of Bad RNA Interference vector. Bad RNAi was validated by transfection of 293 cells
with a Bad expression plasmid in the absence or presence of the U6-driven Bad RNAi plasmid,
followed by lysis and analysis by immunoblot Upper panel was analyzed with an anti-Bad
antibody (N-20) and lower panel with an antibody directed against actin. M = Mock transfection,
V = pcDNA3, U6 = pcDNA3+U6 promoter. U6-R1I = pcDNA3+ U6 promoter drivingBad RNAi
Guide ine;; ror completing the fo.'rn are available at wwyv mcgiil sa/rjo/animal
ssr^ss
Protccc! : *
McGili University
Animal Use Protocol -- Research
!nvestica:cr:?:
Approval End Date
Facility Ccmr.iiee
F i l l ; : MAGE gt-nes arid autism
(musi t.iatek ike til. e of the funding source application)
X] New Application
Q] Renewal of Protocol # _
Category (see section llj
Pilot
I. Investigator Data:
Principal Investigator
Dr Phi! A Barker
Neurology arid Neurosurgery'
Bepiirmienii:
FaT,i:
M.N.I. - :;810 Universilv St.
Iddi-ess:
Email:
Jy«-5iH
phil.barker'axicgil] c£
I. Emergency C o n t a c t s : i u o peoj le must be designated to handle err erge: >aes.
830-3243
Same:
Phi Barkei
Work ft:
393-3064
3
Name:
Kathleen E'icisoi
YV'ork#:
393-3212
Emergency it:
3, F u a d i o g Source:
Esters ai
[^
Internal
ioma
Source (s):
(s):
Stat is :
[K; A-warded
~'^J
827-2919
For Office Use Only:
C l.F.R. - MOP-62^-99
Peer Reviewed: £>3 YES
..>*..< j ^ , .
L ] NO**
f j Pending
Funding period: Ai:r 1. 2003-Mar. 3 1 .
200-i
| |
Peer Reviewed: Q
Status:
YES
' 1 Awarded
L NO**
Q Pending
Funding period:
** Ail ]:rojecis ihi.t have not been peer reviewed for scientific nKrit bv tfce funding source require 2 Pee:- !iev:ev Fo ins to be
comsjeiafi e.;>. Projects f jnded from .ndiistriai sources. Pee:* Review Jonas are avaiiabie at www tncgiH.Cii/rgD/animai
i
Proposed Start But of Anima! Use (d/m/v):
jane 16. 2003
or ongoing
i
Expect:d Date of 'Completio.i of Animal Use (d/m/y):
Mar. 3 ?. 2005
9r ongoing 1 1
1
i
i Investigator's Statement: The information in this application js^oact ^:fS comptoe. I assure that all care and Lse of animals ir tus
i proposal will be in accordance with the guidelines and policies of' thejtanacBan Council on Animal Care and tlicse of McG 11 Universi>• I shall
I request the Aaimal Care Committee's approval prior to any deviatiG5<s^frd"ii this/protocol as approved. I understand that this approval is valid
i for one year and mjst be approved on an annual basis.
/// /
!
I
"
I
y
-'
/
j Principal Investigator's signature:' '
I
,-•>- /
/
Approvedby:
1
Chair, Facility Ankaai C.sre Committee:
•
]
Til .> r - .
i L^aje.
University Veterinarian:
i
! Date;
1
C h a r , Ethics Subcommittee (as perDACCpolio):
4pp r eved Animal list
Beginning:
[ j This protocol has fceer approved with the modiikations noted in Section 13.
October 2002
|
Dare:
! Endin<*:
S3
i