* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download CH12 Page 1-3 - Pharmacotherapy
Survey
Document related concepts
Transcript
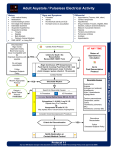
12-1 SECTION 2 CARDIOVASCULAR DISORDERS 12 CARDIAC ARREST Staying Alive . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Level II Jennifer McCann, PharmD, BCPS, BCCCP Sarah Hittle, PharmD, BCPS, BCCCP CASE SUMMARY A 68-year-old female presents to the emergency department (ED) with shortness of breath after missing a hemodialysis session and was subsequently intubated due to respiratory failure. Shortly afterward, the patient develops pulseless electrical activity (PEA). Resuscitation efforts are initiated, and the patient eventually achieves return of spontaneous circulation (ROSC). The reader is asked to apply the advanced cardiac life support (ACLS) guidelines to evaluate the code team response in the absence of a pharmacist for both non-pharmacologic and pharmacologic therapy administered during the arrest.1 An appropriate pharmacotherapeutic plan is then needed to maintain the patient’s stability while optimizing neurological recovery. QUESTIONS Problem Identification 1.a. What actual and potential drug therapy problems does this patient have just prior to the development of pulseless electrical activity (PEA)? Actual problems: • Patient with end-stage renal disease (ESRD) requires hemodialysis for fluid management and electrolyte balance. Patient missed last dialysis session and presents with electrolyte imbalances and metabolic derangements. • Patient is mechanically ventilated; therefore, she is at risk for the development of deep vein thrombosis and stress-related mucosal disease. • Pain and agitation management may be necessary to facilitate mechanical ventilation while minimizing adverse effects. • All critically ill patients require appropriate nutrition support, including an assessment of caloric and protein needs and fluid goals. Potential problems: • Anemia of chronic disease may have played a role in the patient’s symptom development; current treatment may be inadequate. • Needs appropriate diet for kidney disease, dyslipidemia, and diabetes management. • Presents with hypoglycemia; possible need to reassess current treatment of type 2 diabetes mellitus. • Dyslipidemia treated with atorvastatin—unknown long-term control. 1.b.Discuss the possible causes for the development of PEA. PEA is defined as an organized rhythm on electrocardiogram with no palpable pulse detected. Causes need to be identified quickly to prevent death and can be remembered via the mnemonic of H’s and T’s. These include: hypovolemia, hypoxia, hydrogen ion (acidosis), hypo- or hyperkalemia, hypoglycemia, hypothermia, toxins (ie, drug overdose and ingestion), tamponade (cardiac), tension pneumothorax, thrombosis (coronary or pulmonary), and trauma. Although hypoglycemia was not included in the H’s and T’s assessment in the 2015 ACLS algorithm, it remains an important assessment piece for PEA as well as in any patient with a change in level of consciousness. Trauma was removed from the algorithm and replaced with its own specific treatment recommendations among the ACLS guidelines.2 Changes to the care of the trauma patient in cardiac arrest were not updated in the 2015 ACLS publication.1 Possible causes of PEA in this particular patient include: • Hyperkalemia: The patient has ESRD and missed her scheduled hemodialysis, which may have contributed to the elevated potassium on presentation. In addition, ongoing lisinopril administration prior to admission may have played a role. • Hypoglycemia: The patient presented with a blood glucose (BG) of 55 mg/dL. Based on the patient’s history of poor oral intake, ongoing insulin therapy may have played a role in her cardiac arrest. • Hypoxia: Her respiratory failure may have been precipitated by fluid retention including pulmonary edema, as evidenced by clinical exam findings of pitting edema and rales. Her missed hemodialysis session may have contributed to fluid accumulation. Desired Outcome 2.What are the short-term goals of pharmacotherapy for this patient? • Return to and maintain spontaneous circulation. • Prevent additional adverse outcomes (ie, anoxic injury). Therapeutic Alternatives • Close monitoring is needed to maintain cardiovascular stability. 3.a. What nonpharmacologic maneuvers should be taken immediately in a patient with PEA? • Electrolyte abnormalities may be anticipated, given the patient’s kidney disease; warrants close monitoring. • Basic life support (BLS) measures are the immediate first steps in caring for a patient with PEA. In a simultaneous fashion, the Copyright © 2017 by McGraw-Hill Education. All rights reserved. 12-2 SECTION 2 patient is checked for responsiveness, the emergency response service or ACLS team is activated, and the “CAB” interventions are initiated. The “CAB” mnemonic was first adopted by the 2010 ACLS guidelines and refers to1,3: ✓Circulation: Check for a carotid pulse for no more than 10 seconds, and begin cardiopulmonary resuscitation (CPR) immediately starting with chest compressions at least 2 inches in depth and at a rate of 100–120 compressions/min (30:2 ratio). Cardiovascular Disorders ✓Open Airway: In this patient, ensure endotracheal tube is placed properly. ✓Breathing: Deliver rescue breaths via manual ventilation with bag valve-delivered breaths over 1 second. Breaths should be effective enough to see visible chest rise and at a compression to ventilation ratio of 30:2. • Once the automated external defibrillator (AED) arrives along with the ACLS team, assess the rhythm and deliver a shock when indicated to do so (eg, ventricular fibrillation and pulseless ventricular tachycardia). The energy used depends on the type of defibrillator available, monophasic or biphasic. • After these BLS interventions have been initiated, identification of potential causes of the arrest, including the H’s and T’s mentioned above, is critical to survival. Changes to the “CAB” interventions as part of the ACLS measures will include: ✓Circulation. Chest compressions are to be delivered in a continuous fashion with limited interruptions no sooner than every 2 minutes for pulse checks. Additionally, obtaining intravenous (IV) or intraosseous (IO) access to administer fluids and medications without interruptions in CPR is needed. ✓Airway. Endotracheal intubation (already done in this case). Must weigh the benefits of advanced airway placement against consequences of interrupting chest compressions for endotracheal tube placement. Bag-mask may be more appropriate in some patient situations. ✓Breathing. Continue manual ventilation with bag valvedelivered breaths at a rate of 1 rescue breath every 6–8 seconds. 3.b.What pharmacotherapeutic agents are available for the acute therapy of this patient’s condition? • Epinephrine is the drug of choice for PEA. Its primary effect is peripheral vasoconstriction that leads to increased cerebral and cardiac perfusion. The standard dose is 1 mg (10 mL of a 1:10,000 solution) given by IV push every 3–5 minutes. • Vasopressin was once considered an alternative to epinephrine in the treatment of pulseless arrest. It functions similarly to epinephrine by causing peripheral vasoconstriction but with less constriction in the coronary and renal vascular beds. Since the 2010 ACLS guideline publication, large, randomized, controlled studies have reported no difference in survival to hospital discharge, return of spontaneous circulation, or neurologic outcomes in patients treated with vasopressin compared to epinephrine.3–6 As a result, the 2015 ACLS guidelines no longer recommend the use of vasopressin either in combination with or as a substitute to epinephrine in cardiac arrest.1 However, its use in combination with steroids may be considered in some patients and is described here. • Amiodarone is considered an alternative to epinephrine in those patients refractory to defibrillation in the setting of ventricular fibrillation (VF) and pulseless ventricular tachycardia (pVT). It has multiple mechanisms of action including Copyright © 2017 by McGraw-Hill Education. All rights reserved. effects on sodium, potassium, and calcium channels as well as adrenergic-blocking properties. These unique pharmacologic actions make amiodarone a useful agent in both atrial and ventricular arrhythmias and for ventricular rate control. For VF and pVT, amiodarone is given as a 300-mg rapid IV bolus in between epinephrine dosing cycles. • Corticosteroid administration with vasopressin and epinephrine has been evaluated for in-hospital and out-of hospital arrest. Although not clearly understood, corticosteroids are thought to improve the hemodynamic response to vasopressin and epinephrine, resulting in shorter CPR time. As a result, neurological outcomes may be more favorable due to reduction in cerebral ischemia time related to CPR. Studies have reported an increase in the number of patients surviving to hospital discharge with improved neurological outcomes in those receiving methylprednisolone, vasopressin, and epinephrine during the arrest period followed by hydrocortisone post resuscitation.7 Further studies are warranted, therefore ACLS guidelines suggest this strategy may be considered in in-hospital cardiac arrest, but routine use is not recommended at this time. Optimal Plan 4.a. A pharmacist was not available to participate in this resuscitation effort. Assess the appropriateness of the treatment used to obtain a cardiac conversion in this patient (see Table 12-1). • Treatment of PEA: ✓BLS treatment: When the patient became unresponsive, activation of the ACLS team, pulse check, and immediate initiation of CPR was conducted; this aligns well with ACLS guideline recommendations. However, an interruption in CPR was noted before a full 2 minute cycle had been completed to check for a pulse and rhythm. Optimizing the use of effective chest compressions with limited interruptions is an area for further ACLS team education. ✓ACLS treatment: Patient had an advanced airway, receiving mechanical ventilation prior to the PEA. Manual ventilation with bag valve-delivered breaths upon arrival of the ACLS code team would have been appropriate in this patient, but this is not clearly documented in the code record. IO catheter placement occurred upon arrival of the ACLS team, allowing quick access for drug therapy administration. The differential for the PEA arrest appeared to be partially addressed with hyperkalemia treatment initiated, including regular insulin, sodium bicarbonate and calcium chloride. However, no documentation for dextrose 50% (D50) was noted in the record, and prior to arrest, the patient was noted to have a BG of 55 mg/dL. Since hypoglycemia is in the differential for causes of PEA, and because the patient required insulin therapy for hyperkalemia management, the lack of dextrose administration to this patient puts her at a high risk for death. • ROSC achieved: The administration of epinephrine IVP to a bradycardic patient with a pulse was not indicated. This may have contributed to the development of ventricular fibrillation. • Ventricular fibrillation: The team appropriately recognized the shockable rhythm, delivering a timely shock with immediate resumption of chest compressions. However, vasopressin was also administered, and this is no longer recommended by the ACLS guidelines. Furthermore, since epinephrine was administered just 2 minutes prior to the VF arrest, no medication therapy was indicated at that time. 12-3 • With return of spontaneous circulation, the focus should be on stabilizing the patient’s respiratory status, achieving/ maintaining hemodynamic stability, and assessing neurological function. ✓Hemodynamics: If the patient becomes hemodynamically unstable (ie, mean arterial pressure <65 mm Hg), evaluation of intravascular volume and if indicated, treatment with a 0.9% sodium chloride fluid bolus, would be appropriate. Since this patient is in sinus tachycardia, hemodynamically stable and has an advanced airway in place, treatment can be centered on minimizing the recurrence of arrhythmias as well as optimizing neurological recovery and assessment. ✓Neurological: If the patient were unresponsive upon ROSC, it may be appropriate to consider initiation of a hypothermia protocol as part of the post-cardiac arrest care plan as a means of optimizing neurological function. Close monitoring of blood glucose to prevent recurrence of hypoglycemia and subsequent neurologic sequelae, will be necessary. Pain management may be needed in this patient who experienced chest compressions. Selection of a short-acting analgesic that is also safe for a patient with ESRD (eg, fentanyl) on an intermittent basis may be considered. In addition, if agitation occurs despite adequate pain management strategies, a short-acting sedative (eg, propofol) may be preferable to allow more rapid neurological checks to occur during interruptions in its administration.8 • Obtain electrolytes (Na, K, Cl, CO2), BUN, SCr, and glucose every 6–8 hours. • Obtain iCa, Mag, and PO4 daily. • Obtain CBC with differential every 24 hours. REFERENCES 1. Link MS, Berkow LC, Kudenchuk PJ, et al. 2015 American Heart Association update for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation 2015;132(Suppl 2):S444–S464. 2. Vanden Hoek TL, Morrison LJ, Shuster M, et al. Part 12: Cardiac Arrest in Special Situations: 2010 American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation 2010;122:S829–S861. 3. Neumar RW, Otto CW, Link MS, et al. Part 8: Advanced Life Support: 2010 American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation 2010;122:S729–S767. 4. Ong ME, Tiah L, Leong BS, et al. A randomized, double-blind, multicentre trial comparing vasopressin and adrenaline in patients with cardiac arrest presenting to or in the Emergency Department. Resuscitation 2012;83:953–960. 5. Gueugniaud PY, David JS, Chanzy E, et al. Vasopressin and epinephrine vs. epinephrine alone in cardiopulmonary resuscitation. N Engl J Med 2008;359:21–30. 6. Mukoyama T, Kinoshita K, Nagao K, et al. Reduced effectiveness of vasopressin in repeated doses for patients undergoing prolonged cardiopulmonary resuscitation. Resuscitation 2009;80:755–761. 7. Mentzelopoulos SD, Malachias S, Chamos C, et al. Vasopressin, steroids, and epinephrine and neurologically favorable survival after in-hospital cardiac arrest: a randomized clinical trial. JAMA 2013;310:270–279. 8. Barr J, Fraser GL, Puntillo K, et al. Clinical practice guidelines for the management of pain, agitation, and delirium in adult patients in the intensive care unit. Crit Care Med 2013;41:263–306. Outcome Evaluation 5.How should the patient be monitored to prevent or detect adverse effects? In order to prevent recurrence of PEA, the patient’s vital signs, neurological status and electrolytes should be monitored closely: • Monitor ECG (HR and rhythm) continuously. • Check vital signs hourly. Copyright © 2017 by McGraw-Hill Education. All rights reserved. Cardiac Arrest ✓Respiratory failure: Since an advanced airway has been placed in this patient, ventilator management under the direction of the critical care physician team will occur. Since the missed hemodialysis session (leading to hyperkalemia and pulmonary edema/hypoxia) is the most likely cause of the initial arrest, hemodialysis may be needed urgently in this patient to assist with rapid correction of fluid and electrolyte imbalance. • Conduct neuro checks using Glasgow coma scale (GCS) at least every 4 hours. CHAPTER 12 4.b.After achieving return of spontaneous circulation (ROSC), what is your pharmacotherapeutic plan to maintain the patient’s stability and optimize neurological function?