* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download An Evaluation of a Gentamicin Dosing Protocol in the Treatment of
Survey
Document related concepts
Group development wikipedia , lookup
Maternal physiological changes in pregnancy wikipedia , lookup
Prenatal nutrition wikipedia , lookup
Fetal origins hypothesis wikipedia , lookup
Preterm birth wikipedia , lookup
Hypothermia therapy for neonatal encephalopathy wikipedia , lookup
Transcript
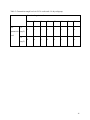
An Evaluation of a Gentamicin Dosing Protocol in the Treatment of Bacterial Infections in Neonates Johanna Ponnuthurai BHSc1; Nisha Varughese, PharmD2; Claire Laframboise, Rph3; Lena Lauzon, Rph2; Annie Pouliot, Ph.D2; Brigitte Lemyre, MD4,5 Affiliations: 1Clinical Research Unit, Children’s Hospital of Eastern Ontario Research Institute, Ottawa, Canada; 2Department of Pharmacy, Children’s Hospital of Eastern Ontario, Ottawa, Canada; 3Department of Pharmacy, The Ottawa Hospital, Ottawa, Canada; 4Department of Pediatrics, Children’s Hospital of Eastern Ontario, Ottawa, Canada; 5Department of Obstetrics, Gynecology and Newborn Care the Ottawa Hospital, Ottawa, Canada Running Title: Evaluation of a Gentamicin Dosing Protocol Key Words: gentamicin, newborn Corresponding author: Brigitte Lemyre, MD, Department of Pediatrics, Children’s Hospital of Eastern Ontario, 401 Smyth Road, Ottawa, Canada, K1H 8L1 Email: [email protected] Telephone: +1 (613) 737-8561 1 Abstract Objective: To determine the safety and efficacy of a revised gentamicin dosing regimen. Study Design: Retrospective study of neonates ≤ 1 month corrected gestational age and admitted to one of 3 Neonatal Units between June 1and December 31, 2012. The primary outcome was trough levels ≤2 mg/L. Result: 151 neonates (mean 33.5 weeks) were included. 80% of patients had a trough level ≤2 mg/L while 94% had peak levels of 5-10 mg/L. The majority of trough levels >2 mg/L were in infants 29-36 weeks corrected and ≤14 days and in term babies ≤7 days of age. Lower gestational age and lower 1 and 5 minute Apgar scores were associated with a high trough. Conclusion: Our gentamicin dosing regimen is suboptimal for moderately preterm neonates who are ≤14 days and term infants ≤7 days. Further research is needed to determine the most effective gentamicin dosing regimen, particularly in preterm neonates. 2 List of Abbreviations CHEO – Children’s Hospital of Eastern Ontario HIE - Hypoxic ischemic encephalopathy NICU – Neonatal intensive care unit TOH-CC – The Ottawa Hospital – Civic Campus TOH-GC – The Ottawa Hospital – General Campus 3 Introduction Gentamicin is an aminoglycoside antibiotic with bactericidal properties that is routinely used along with beta-lactam antibiotics to treat proven or suspected bacterial infections in term or preterm neonates.1-4 Gentamicin has concentration dependent bacterial killing and a postantibiotic effect, which can lead to toxicity if trough concentrations are too high.2, 4 To optimize therapeutic concentrations and to minimize toxicity, gentamicin serum levels are closely monitored. Low trough concentrations correspond with a reduced risk for toxicity, and a high peak concentration corresponds with therapeutic efficacy.8 Gentamicin dosing regimens in neonates at our centers are based on traditional dosing based on weight, gestational or corrected gestational age. The target trough level is ≤ 2mg/L and the target peak level is 5-10 mg/L, with the ideal peak level between 7 and 10 mg/L. Dosing can be challenging in neonates due to their increased volume of distribution and decreased renal clearance, compared to that of adults.2, 6 Factors that have been known to affect clearance and determine dosing include birth weight, gestational age, and postnatal age which also affect gentamicin clearance.1 Gentamicin is excreted by the kidneys un-metabolized through glomerular filtration, which can accumulate in neonates with immature renal function after birth, including those with hypoxic ischemic encephalopathy (HIE) due to perinatal asphyxia.4, 6, 7 The Pharmacists at our three institutions collectively revised our gentamicin dosing protocol (Figure 1) recently to optimize peak levels and minimize toxicity for term and preterm neonates. The objective of this study was to examine the safety and efficacy of the revised dosing regimen compared to the prior dosing regimen. Materials and Methods 4 A retrospective chart review of neonates admitted to the level 3c neonatal intensive care unit (NICU) of the Children’s Hospital of Eastern Ontario (CHEO), the level 3a NICU of The Ottawa Hospital – General Campus (TOH-GC) or the level 2c neonatal unit at The Ottawa Hospital – Civic Campus (TOH-CC) between June 1, 2012 and December 31, 2012 was conducted. The study was approved by the Research Ethics Boards at CHEO and TOH. Neonates up to one month corrected gestational age who had at least one serum gentamicin level measured were included in the study. Gentamicin was administered by intermittent intravenous infusion over 30 minutes. A trough sample was obtained 30 minutes before administration of the third dose and a peak sample was obtained 30 minutes after the end of the third infusion. Data was collected using electronic patient records and individual patient charts. Demographic data collected included gestational age, postnatal age, gender and birth weight. All data regarding gentamicin administration (dose, interval, trough and peak levels) were also collected. Apgar scores at 1, 5, and 10 minutes, admission diagnosis, diagnosis of HIE (defined as a cord pH ≤7.1 and an Apgar score of ≤5 at 10 minutes) and whether therapeutic hypothermia was received were also collected. Urine output from 24hrs prior to 72hrs after the first dose of gentamicin, creatinine and urea levels pre-gentamicin were also collected if available. Participants were categorized into different age groups according to the gentamicin dosing protocol: 1) less than 29 weeks and more than 14 days; 2) less than 29 weeks and 0-14 days; 3) 29-36 weeks and more than 14 days; 4) 29-36 weeks and more than 14 days; 5) 37 weeks or more and more than 7 days; and 6) 37 weeks or more and 0-7 days. (Figure 1) Outcomes 5 The primary outcome of the study was the proportion of patients with an acceptable trough level (≤2 mg/L). Secondary outcomes included the proportion of patients with an acceptable peak level (5-10 mg/L) and proportions with both acceptable trough and peak by age group category. Analysis Statistical analysis was conducted using SPSS version 20.0. P values <0.05 were considered significant. Descriptive statistics were used for all categorical variables and summarized using frequencies and percentages. Normally distributed continuous variables were summarized using means and standard deviations. Non-normally distributed continuous variables were summarized using medians and quartiles. Analyses were performed for each of the age subgroups in the new dosing regimen. Results 151 neonates were included in the study. Their mean gestational age was 33.5 +/- 5.3 weeks, mean postnatal age was 5.4 +/- 11.9 days and mean birth weight was 2.3 +/- 1.1 kg. (Table 1) The majority of patients were either 29-36 weeks gestational age and <14 days old or ≥37 weeks in their first week of life. (Table 1) A trough gentamicin level was measured in 149 neonates. 119 neonates (79.9%) had an adequate trough level and 30 neonates (20.1%) had an inadequate trough level (Table 2). A peak gentamicin level was measured in 122 neonates. Target peak levels (5-10 mg/L) were found in 118 neonates (96.7%). Peak levels of 7-10 mg/L were found in 84 neonates (68.9%). Trough levels were found to be acceptable in all age groups, with the exception of neonates 29-36 weeks 6 gestational age and 0-14 days old, where 33.3% had a high trough and term babies 0-7 days old, where 17.5% had a high trough (Table 2). We performed exploratory analyses, to identify risk factors that could be associated with trough levels >2 mg/L in these 2 subgroups. In the 29-36 weeks gestational age and 0-14 days subgroup, the lower the gestational age, the greater the likelihood of a trough level >2mg/L (Pearson correlation coefficient, 2 tailed, p=0.007) (Table 3). Apgar score at 1 and 5 minutes also correlated inversely with a high trough level (p=0.05 for Apgar score at 1 minutes and p=0.01 for Apgar score at 5 minutes). Low urinary output (<2 mL/kg/h) after the first dose of gentamicin or concomitant treatment with ibuprofen or indomethacin were not associated with a high trough level. In the 0-7 day-old term neonate sub-group, a diagnosis of HIE was associated with a greater risk of having a high gentamicin trough level (p=0.007); 4 out of 6 infants (including 3 out of 4 babies who received therapeutic cooling) with HIE on admission had a high trough level. Apgar scores at 5 minutes were also inversely correlated with a high trough level (p=0.01). As well, a higher gestational age in this category was associated with a lower likelihood of a high trough level (p=0.045). We assessed whether a serum creatinine ≥60 µmol/L was associated with a higher likelihood of a high trough level, but it was not (p=0.054). Discussion Dosing gentamicin in neonates to obtain maximal efficacy with minimal toxicity is challenging. This study found that our revised gentamicin dosing regimen did not achieve this goal for moderately preterm infants who are less than 2 weeks old and term babies who are in their first week of life. Our dosing regimen thus requires adjustment. Our data suggests that 7 gestational age as well as postnatal age significantly influence gentamicin clearance, likely due to delayed kidney maturation. This is consistent with previous studies on gentamicin pharmacokinetics.1, 2, 7 Overall, babies less than 34 weeks gestational age and less than 2 weeks of life were more likely to have high trough levels. Currently, the dosing regimen for neonates between 29-36 weeks gestational age and 0-14 days old is 3-3.5mg/kg/dose every 18h; this may be too broad in terms of dose and age range, particularly for the moderately preterm neonate. Consideration should be given as to whether lowering the dose or increasing the interval would allow maximal efficacy and minimal toxicity. Term babies 0-7 days old with high trough levels were of particular interest because they were all admitted to the 3c NICU at CHEO. This may be explained by higher acuity of illness in these patients, who were referred from community hospitals; however we were not able to demonstrate that retrospectively. Many of these term neonates had HIE or were cooled for HIE, but not all of them, which may be associated with poorer renal function and lower creatinine clearance. Recent literature has found that therapeutic hypothermia can alter gentamicin pharmacokinetics, prolonging its half-life.3, 6, 12, 13 Neonates with HIE often have renal dysfunction, which results in an inability to properly excrete gentamicin.7 Frymoyer et al. noted that gentamicin clearance was 25-50% lower in term neonates with HIE than their term, nonasphyxiated counterparts. While our institutions use traditional dosing, there exists debate in the literature that extended interval dosing may be more effective. Previous studies 8, 9, 10 on term neonates suggest that extended interval dosing is superior to traditional dosing, in achieving higher peak levels and lower trough levels. Much of the literature regarding extended interval dosing looks at late preterm or term neonates.8, 9, 10, 14, 15, 17 A daily dose of 4mg/kg is recommended for term babies 8 ≥34 weeks gestational age.8, 9, 10, 15, 16, 17 There is limited data available confirming the efficacy of this regimen in early preterm neonates, born <34 weeks.2, 16, 17 Available dosing recommendations for preterm neonates states that 3mg/kg is optimal.16 Even less research is available for early preterm infants as they are often not considered in these studies. We acknowledge the following limitations for our study: 1) the retrospective nature of this study has intrinsic limitations, mostly availability of all data; 2) small numbers of some subgroups prevented meaningful analysis and allows for the possibility of type 2 errors; 3) analysis of the influence of renal function was limited by the age of the majority of infants, as most had not established good urine output yet. Due to the above findings, we have used pharmacokinetic modeling to modify the dosing of gentamicin for these 2 subgroups. (Data not shown) The modifications include a precise dose (3.5 mg) as opposed to a range of dosing (3.0 to 3.5 mg) and dosing every 24h in infants 29-36 weeks and 0-14 days instead of every 18h. For neonates with a low cord pH and low Apgar scores or those with risk factors for abnormal renal function, we also recommend obtaining a serum trough level 24h after the first dose of gentamicin and giving further doses only if the level is ≤2 mg/L. We are currently evaluating the effects of these revisions in our dosing regimen. In conclusion, a dose of 3-3.5mg/kg/dose every 18h was found to be sub-optimal in neonates 29-36 weeks gestational age and 0-14 days old. Furthermore, a dose of 33.5mg/kg/dose every 18h was also found to be sub-optimal in term neonates 0-7 days old. Ongoing quality assurance projects to ensure optimal dosing regimens in the neonatal population are important, particularly after modifications. The literature is inconclusive regarding whether 9 extended interval dosing is the best method of gentamicin dosing for preterm neonates, therefore, further research is warranted. 10 Acknowledgements The authors would like to acknowledge Samantha Somers for editing the manuscript. Conflict of Interest The authors declare no conflicts or financial interest in any product or service mentioned in the manuscript including grants, medications, employment, gifts, and honoraria. 11 References [1] Mohamed A, Nielsen E, Cars O, Friberg L. Pharmacokinetic-Pharmacodynamic model for gentamicin and its adaptive resistance with predictions of dosing schedules in newborn infants. Antimicrobial Agents and Chemotherapy 2012; 56(1): 179-188. [2] Alshaikh B, Dersch-Mills D, Taylor R, Akierman A, Yusuf K. Extended interval dosing of gentamicin in premature neonates ≤ 28-week. Acta Pædiatrica 2012; 101: 1134–1139. [3] Liu X, Borooah M, Stone J, Chakkarapani E, Thoresen M. Serum gentamicin concentrations in encephalopathic infants are not affected by therapeutic hypothermia. Pediatrics 2009; 124(1): 310-315. [4] Nielsen EI, Sandström M, Honoré PH, Ewald U, Friberg LE. Developmental pharmacokinetics of gentamicin in preterm and term neonates. Clinical Pharmacokinetics 2009; 48(4): 253-263. [5] Tessin I, Bergmark J, Hiesche K, Jagenburg R, Trollfors B. Renal function of neonates during gentamicin treatment. Archives of Disease in Childhood 1982; 57(10): 758-760. [6] Mark L, Solomon A, Northington F, Lee C. Gentamicin pharmacokinetics in neonates undergoing therapeutic hypothermia. Ther Drug Monit 2013; 35(2): 217-222. [7] Frymoyer A, Meng L, Bonifacio S, Verotta D, Guglielmo BJ. Gentamicin pharmacokinetics and dosing in neonates with hypoxic ischemic encephalopathy receiving hypothermia. Pharmacotherapy 2013; 33(7): 718-726. 12 [8] Hagen I, Oymar K. Pharmacological differences between once daily and twice daily gentamicin dosage in newborns with suspected sepsis. Pharmacy World and Science 2009; 31: 18-23. [9] Thureen PJ, Reiter PD, Gresores A, Stolpman NM, Kawato K, Hall DM. Once versus twicedaily gentamicin dosing in neonates ≥34 weeks’ gestation: cost-effectiveness analysis. Pediatrics 1999; 103: 594. [10] Lundergan FS, Glasscock GF, Kim EH, Cohen RS. Once-daily gentamicin dosing in newborn infants. Pediatrics 1999; 103(6): 1228-1234. [11] Hitron AE, Yao S, Scarpace SB. Accuracy of empiric gentamicin dosing guidelines in neonates. The Journal of Pediatric Pharmacology and Therapeutics 2010; 15(4): 264-273. [12] Zhou J, Poloyac S. The effect of therapeutic hypothermia on drug metabolism and response: cellular mechanisms to organ function. Expert Opinion on Drug Metabolism and Toxicology 2011; 7(7): 803-816. [13] Bagna FC, Pitoni S, Andrews PJD. Therapeutic mild hypothermia and the pharmacokinetics of drugs in trauma brain injury (TBI) patients with a focus on sedation, anticonvulsant and antibiotic therapy. The Open Critical Care Medicine Journal 2013; 6: 31-38. [14] de Alba Romero C, Castillo EG, Secades CM, López JR, López LA, Valiente PS. Once daily gentamicin dosing in neonates. The Pediatric Infectious Disease Journal 1998; 17(12): 1169-1171. 13 [15] Krishnan L, George SA. Gentamicin therapy in preterms: a comparison of two dosing regimens. Indian Pediatrics 1997; 34: 1075-1080. [16] Hansen A, Forbes P, Arnold A, O’Rourke E. Once-daily gentamicin dosing for the preterm and the term newborn: proposal for a simple regimen that achieves target levels. Journal of Perinatology 2003; 23: 635-639. [17] Agarwal G, Rastogi A, Pytai S, Wilks A, Pildes RS. Comparison of once-daily versus twicedaily gentamicin dosing regimens in infants ≥2500g. Journal of Perinatology 2002; 22: 268-274. 14 Table 1: Demographics of Study Participants Demographics n=151 Gender [n (%)] Male 95 (62.9%) Female 56 (37.1%) GA, weeks (mean +/- SD) 33.5 +/- 5.262 BW, kg (mean +/- SD) 2.263 +/- 1.106 Postnatal age, days (mean +/- SD) 5.42 +/- 11.950 Subgroup [n (%)] ≤28 weeks CGA and >14 days 1 (0.7%) ≤28 weeks GA 0-14 days 26 (17.0%) 29-36 weeks CGA and >14 days 14 (9.3%) 29-36 weeks GA and 0-14 days 50 (33.1%) ≥37 weeks and >7 days 3(2.0%) ≥37 weeks and 0-7 days 57 (37.8%) 15 Table 2: Gentamicin trough levels by age group Age Group Pre- ≤2 ≤ 28 ≤ 28 29-36 29-36 ≥ 37 ≥ 37 weeks weeks weeks weeks weeks weeks CGA GA CGA GA GA GA >14 days 0-14 days >14 days 0-14 days > 7 days 0-7 days 1 24 12 32 3 47 0 2 2 16 0 10 1 26 14 48 3 57 gentamicin (mg/L) level >2 (mg/L) N (total) 16 Table 3: Gentamicin trough levels in 29-36 weeks and <14 days subgroup Gestational age in weeks Pre- 29 30 31 32 33 34 35 36 2 0 3 1 7 9 2 8 5 4 2 1 3 0 1 0 ≤2 gentamicin (mg/L) level >2 (mg/L) 17 Figure 1: Gentamicin Dosing Protocol 18