* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download here
Survey
Document related concepts
Transcript
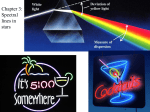
NatSci102 Due Feb. 14 Professor G. Rieke Homework on spectroscopy, colors, and light. Answers should be entered on a Scantron form given out in class. This exercise is worth 30 points (25 questions plus 5 for the spectrum drawings) 1. Is the spectrum below a. an absorption line one d. Doppler shifted b. a continuum e. unresolved c. an emission line one 2. If a photon of energy E has a wavelength of 0.6 microns, what energy photon will have a wavelength of 0.3 microns? a. 2 times E b. Half of E c. 4 times E d. ¼ of E e. the energy cannot be determined accurately 3. The figure on the left illustrates electronic transitions from 3 higher levels to a lower one (52, 42, or 3 2) in the hydrogen atom. Which transition produces a photon with the highest energy? a. 5 -> 2 b. 4 -> 2 c.3 -> 2 4. Which transition produces a photon in the reddest portion of the spectrum? a. 5 -> 2 b. 4 -> 2 c. 3 -> 2 An energy level diagram showing the 3 hydrogen transitions which give the strongest lines in the visible spectrum. 5. What type of spectrum (emission, or absorption) does the observer see in the figure on the right? a. absorption b. emission c. continuous 6. How are the electrons in the atoms in the hydrogen cloud in this figure behaving? 1 a. They are jumping from a lower to a higher level b. they are jumping from higher to lower c. They are leaving the cloud d. We cannot know how they are moving e. They are jumping from a lower to a higher level and then eventually falling back to the lower level 7. The young, hot star in picture to the left heats up the gas in the cloud. Assume that the observer can only see radiation emitted by the hydrogen cloud and cannot see the star. What type of spectrum (emission, or absorption) does the observer see? a. absorption b. emission c. continuous 8. How are the electrons in the atoms in the hydrogen cloud in this figure behaving? a. They are jumping from a lower to a higher level b. they are jumping from higher to lower c. They are leaving the cloud d. we cannot know how they are moving e. They are jumping from a lower to a higher level and then eventually falling back to the lower level 9. Look at the spectrum of the Sun below. What type of spectrum (continuous, emission line or absorption line spectrum) does the Sun produce? a. absorption b. emission c. continuous The next few problems will be much easier if you review how to use scientific notation (see also http://www.ifa.hawaii.edu/~barnes/ast110_06/sosat.html#[20] ) 10. Hydrogen has a spectral line that is observed at a wavelength of 656 nm (656x10-9 meters). Recalling that the speed of light is 3x108 meters/sec and the relationship between wavelength and frequency, what is the frequency of the photons observed as this spectral line? a. 4.6x1014 Hz b. 2.2x10-15 Hz c. 100 MHz d. 4.6 Hz e. Cannot be computed 11. The photons producing the hydrogen line in question 10 also have energy. Review the relationship between energy and frequency given in the lectures. Use h=Planck’s constant = 6.6x10-34 Joules/Hz to compute the photon’s energy which is ___________ Joules. a. 3 b. 3.0x10-19 c. 24 d. 24x1019 e. 7x1047 2 12. If you observed the hydrogen line discussed in questions 10 and 11, what color would it have? a. purple b. green c. white d. all the colors of the rainbow e. red A. B. Examine the two spectra above. Go to http://www.amateurspectroscopy.com/color-spectra-ofchemical-elements.htm and compare the spectra shown there with these spectra. 13. The spectrum labeled A. above is produced by which element? a. lithium b. hydrogen c. helium d. carbon e. no way to tell 14. The spectrum labeled B. above is produced by which element? a. lithium b. hydrogen c. helium d. carbon e. no way to tell A. B. C. D. 3 15. The spectra of four stars are shown above. Which star’s spectrum is dominated by spectral lines from hydrogen? A. B. C. D. E. none are 16. The spectrum plotted above is from a real star, Vega. The spectrum is also shown in color as it might appear to your eye. What element’s absorption lines are prominent in Vega’s spectrum? a. lithium b. hydrogen c. helium d. carbon e. no way to tell 410.2165 410.4216 The plot at the right shows a magnified section of the Vega spectrum in black. You may assume that Vega is not moving. The red plot is part of the spectrum from another star very similar to 4 Vega except for its Doppler shift. The numbers show the location of the spectral line. Use c=300,000 km/sec 17. In what direction is the star producing the red spectrum moving? a. Toward us b. Away from us c. Cannot be figured out from this plot 18. How fast is the star producing the red spectrum moving? a. 300,000 km/sec b. 150 km/sec c. 15000 km/sec d. 0.205 km/sec e. 61,500 km/sec 19. This figure is similar to the previous one but the blue plot is the spectrum from a third star with a different Doppler shift. In what direction is the star producing the blue spectrum moving? a. Toward us b. Away from us c. Cannot be figured out from this plot 20. How fast is the star producing the blue spectrum moving? a. 300,000 km/sec b. 210 km/sec c. 21000 km/sec d. 0.287 km/sec e. 86,160 km/sec 21. The graph below shows the output of a black body at a temperature of 5000K, against the wavelength. The peak is at 0.58 microns. How would the output against wavelength change if the temperature were 2500K? a. it would stay the same b. it would get fainter but otherwise stay the same c. the shape of the curve of output against wavelength would change d. the peak of the output would appear at 1.16 microns e. the peak of the output would appear at 0.29 microns 5 409.9293 410.2165 1.4 1.2 1 flux 0.8 0.6 0.4 0.2 0 0 0.2 0.4 0.6 0.8 1 1.2 1.4 1.6 1.8 2 wavelength (microns) 22. In the situation described in problem 21, with the temperature going down by a factor of two, how will the total output over the entire range of wavelengths (the luminosity) change? a. stay the same b. get 2 times smaller c. get 4 times smaller d. get 8 times smaller e. get 16 times smaller 23. How low would the temperature have to be for the peak of the output to be in the far infrared, at 100 microns? a. 250K b. near absolute zero c. 29K d. -57C e. None of the above 24. Of the two spectra you observed in class, which had emission lines? a. Incandescent light b. Fluorescent light c. Both d. Neither 25. From the spectra below, can you identify a material in one of the types of light? a. neon b. mercury c. sodium d. hydrogen e. no, none of these materials are in either light 6 7