* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Eyedrops can affect more than the eye
Survey
Document related concepts
Transcript
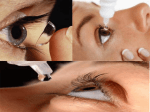
ESONT 7 Eyedrops can affect more than the eye Stefanie Petrou-Binder MD in Nürnberg While ophthalmic eye drops may contain a very small amount of a given drug, nurses and staff should remember that these medications can still have significant systemic side effects, said Sabine Menkhaus MD, University Eye Clinic Magdeburg, Germany. "When administered, eye drops come into contact with the conjunctiva and the secretory membranes of the tear canals, eventually reaching the circulatory system through the mucosa of the nose and throat. One should not underestimate the potential systemic side effects that ocular medications may precipitate," Dr Menkhaus told the 17th Congress of the German Ophthalmic Surgeons (DOC) in Nürnberg, Germany. A good example of a very commonly applied ophthalmic medication that can cause unwanted systemic side effects is atropine. Atropine drops are administered to achieve mydriasis, which allows retinal examination, refractive measurement, and even the dissolution of adhesions between the iris and the lens. Side effects to watch out for include: fever, headache, facial erythema, dry mouth, high blood pressure, difficulty in concentration, cramps (in children), and blurry vision. Patients should be advised not to drive until this effect subsides. Also, closed angle glaucoma patients should not receive mydriatic drugs, to avoid a glaucoma attack. Glaucoma medications represent another group of ophthalmic drugs that can bring about unwanted systemic reactions. For instance, beta blockers (Tilomann, Betamann, Vistagan, Arutimol), which decrease the production of aqueous humour, may cause bradycardia, reduced blood pressure, bronchial asthma, and dry eye. Cholinergic drugs such as pilocarpine are used in glaucoma therapy to increase the flow of aqueous humour and induce a narrow pupil. They may also, in turn, worsen vision in patients with cataracts and reduce visual acuity at night. Both alpha-sympathotonics (Iopidine and Alphagan) and carbonic anhydrase blockers (Trusopt, Azopt, Cosopt), on the other hand, cause a reduction in aqueous humour production. They can be the cause of allergy, fatigue and headaches, with carbonic anhydrase blockers leaving the patient with a bitter, metallic taste. Other carbonic anhydrase blockers like Diamox and Glaupax tablets affect the excretion of potassium in the kidney, causing a low blood potassium level (hypokalemia), heart arrhythmias, exsiccation, paraesthesias in the extremities, and nausea. Xalatan and Travatan are prostaglandin analogues used in glaucoma therapy. These agents can cause an increase in aqueous humour emptying and a dark discoloration of the iris. As these drugs may bring about increased eyelash growth, Dr Menkhaus suggested applying it to both eyes to achieve a better cosmetic result. Drugs that "get the red out" function by causing a vasoconstriction of the conjunctival vessels. Dr Menkhaus urged ophthalmic professionals to remind their patients not to abuse these drugs as the eye can get used to them over time. So much so, that the eye may appear red unless they are applied. These drugs may also cause a rise in blood pressure, daze and even hallucinations. Local, as opposed to systemic, drug reactions are varied. Reactions to ophthalmic medications may include eczema, for instance, which results from eye drops that come into contact with the skin of the eyelids or cheeks. The skin exerts an allergic reaction from exposure to the active ingredient or conservation medium. Antibiotic eye drops and eye ointments are used for the treatment of inflammations of the lids, conjunctiva, cornea, and lacrimal pathways. These drugs may cause irritation in the eye, but systemic side effects are unlikely. Dr Menkhaus pointed out that local anaesthetic drops such as Novesine or Propakarakain POS, should best be administered by the doctor or staff during treatments. She warned that if used at home as a remedy for chronic pain, a disruption in the nutrition of the cornea may occur, which could ultimately lead to corneal ulcerations. She asserted that eye drops that contain corticosteroids are a well-known cause of increased intra-ocular pressure (and glaucoma) and may further catalyse the development of lens opacification (and cataract). Also, patients with a healed herpetic inflammation of the eye may suffer a reactivation of the viral disease when receiving cortisone-containing medications, unless anti-viral eye ointment is administered concomitantly. Even iodine, which is applied to the skin and conjunctiva pre-operatively for disinfection, may provoke an allergic skin reaction or exacerbate pre-existent hyperthyroidism. Because even the smallest amount of an active substance of a drug can cause systemic side effects, Dr. Menkhaus recommended a simple trick to side-step potential adverse reactions. "After administering the eye drops, the doctor should apply slight pressure with his finger on the descending tear canal for approximately two minutes. Any excess drops should be carefully wiped away and cleaned from the eyelids and cheeks. This way one can ensure that only a minimal drug dose enters the circulatory system, and reduce the possibility of systemic side-effects and the risk of contact allergy on eye lids and cheeks." [email protected] NOVEMBER 2004 MAY 2005 SEPTEMBER 2005 SEPTEMBER 2006 26 19-20 10-14 9-13 LONDON, UK XXIV Congress of the ESCRS & ESONT Programme Contact:ESCRS / ESONT Temple House,Temple Road,Blackrock, Co.Dublin,Ireland Tel:+ 353 1 209 1100, Fax:+ 353 1 209 1112 Email:[email protected] BRUSSELS, BELGIUM Annual Congress of BSONT (Belgian Society of Ophthalmic Nurses & Technicians) Contact:Gert Princen Email:[email protected] MALMÖ, SWEDEN Annual Meeting of the Swedish Ophthalmic Nurses Society Contact:Ulla Hurtig Email:[email protected] LISBON, PORTUGAL XXIII Congress of the ESCRS & ESONT Programme Contact:ESCRS / ESONT Temple House,Temple Road,Blackrock, Co.Dublin,Ireland Tel:+ 353 1 209 1100, Fax:+ 353 1 209 1112 Email:[email protected] calendar January 2005 28-29 VEJLE, DENMARK Annual Meeting of the Danish Ophthalmic Nurses Society Email:[email protected] Web:www.dsr.dk/fs20 JUNE 2005 16-18 NYÍREGYHÁZA, HUNGARY V Congress of the SHAO (Hungarian Ophthalmic Assistants Society) Contact:Dr.Kelenhegyi Csilla Tel:+ 36 303 492 058 Email:[email protected] DUOVISC ® Viscoelastic System VISCOAT® Ophthalmic Viscosurgical Device PROVISC® Ophthalmic Viscosurgical Device Device Description: DUOVISC® Viscoelastic System is designed to give two viscoelastic materials with different physiochemical properties that can be used differently and sequentially to perform specific tasks during a cataract procedure. VISCOAT ® (Sodium Chondroitin Sulfate Hyaluronate) Ophthalmic Viscosurgical Device Sodium Indications: VISCOAT® Ophthalmic Viscosurgical Device is indicated for use as a surgical aid in anterior segment procedures including cataract extraction and intraocular lens (IOL) implantation. VISCOAT® Ophthalmic Viscosurgical Device maintains a deep chamber during anterior segment surgeries, enhances visualization during the surgical procedure, and protects the corneal endothelium and other ocular tissues. The viscoelasticity of the solution maintains the normal position of the vitreous face and prevents formation of a flat chamber during surgery. Contraindications: At the present time, there are no known contraindications to the use of VISCOAT® Ophthalmic Viscosurgical Device when used as recommended. Warning: Failure to follow all of the assembly instructions in “Directions for Use” or use of an alternate cannula may result in cannula detachment and the possibility of serious injury. Precautions: Precautions are limited to those normally associated with the surgical procedure being performed. Although sodium hyaluronate and sodium chondroitin sulfate are highly purified biological polymers, the physician should be aware of the potential allergic risks inherent in the use of any biological material. This Product Contains Dry Natural Rubber. Adverse Reactions: VISCOAT® Ophthalmic Viscosurgical Device has been extremely well tolerated in human and animal studies. A transient rise in intraocular pressure may be expected due to the presence of sodium hyaluronate, which has been shown to effect such a rise (9.8% > 25mmHg during 1-3 days after surgery in human clinical trials). PROVISC ® (Sodium Hyaluronate) Ophthalmic Viscosurgical Device Indications: PROVISC® Ophthalmic Viscosurgical Device is indicated for use as an ophthalmic surgical aid in the anterior segment during cataract extraction and intraocular lens (IOL) implantation. Ophthalmic viscoelastics serve to maintain a deep anterior chamber during anterior segment surgery allowing reduced trauma to the corneal endothelium and surrounding ocular tissues. They help push back the vitreous face and prevent formation of a flat chamber postoperatively. Contraindications: At present there are no known contraindications to the use of PROVISC® Ophthalmic Viscosurgical Device when used as recommended; care should be used in patients with hypersensitivity to any components in this material. Precautions: Postoperative increases in intraocular pressure have been reported with sodium hyaluronate products. The IOP should be carefully monitored and appropriate therapy instituted if significant increases occur. It is recommended that PROVISC® Ophthalmic Viscosurgical Device be removed by irrigation and/or aspiration at the close of surgery. Do not overfill anterior chamber. PROVISC® Ophthalmic Viscosurgical Device is obtained from microbial fermentation by a purified proprietary process. Although precautions have been taken to make this device protein-free and it has been tested in animals for allergenic response, this device, used in susceptible persons, may produce allergenic responses. This Product Contains Dry Natural Rubber. Adverse Reactions: Postoperative inflammatory reactions such as hypopyon and iritis have been reported with the use of ophthalmic viscoelastics, as well as incidents of corneal edema, corneal decompensation, and a transient rise in intraocular pressure. Refer to the product insert for complete set of prescribing information. CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO THE SALE BY OR ON THE ORDER OF A PHYSICIAN. U.S. Patent Nos. 5,273,056; 5,876,379 and 6,051,560. June 2006 19-20 COPENHAGEN, DENMARK Congress of the Nordic Co-Operation of Ophthalmic Nurses Email:[email protected] A www.alconlabs.com ©2003 Alcon, Inc. VIS4423 ESONT EuroTimes Magazine October-December 2004