* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Reactions of Alkenes
Survey
Document related concepts
Asymmetric induction wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Homoaromaticity wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
2-Norbornyl cation wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Petasis reaction wikipedia , lookup
Ene reaction wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
Vinylcyclopropane rearrangement wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Marcus theory wikipedia , lookup
Transcript
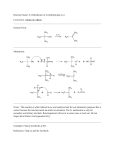
Reactions of Alkenes H C + HCl C C + C C C Cl hyd roch lorination (h yd rohalogenation ) C OH hydration H H+ H2 O C H + H Br C H C C C H C C C + Br 2 H+ + CCl4 OsO4 O3 C C (CH3 ) 2 S C O H O C H H H C C H H C + H 1. BH3 2. HO , H2O2, H2O O3 (CH3 ) 2 S H redu ction (h yd rogenation) C Br C addition of radicals C H oxymercuration/demercuration OCH3 C H C OH H Br O H oxidation H HO H Ozonolysis 1. Hg(OAc)2, CH3OH - Br H2 O Addition of HOX; X: Cl, Br peroxide 2. NaBH4 C H Br2 H bromination (h alogenation) OH OH+ (hydrogenation) Ozonolysis + H2 + HBr C C H C C H H H hydroboration-oxidation H O H + H H Reaction Mechanism • A reaction mechanism describes how a reaction occurs – which bonds are broken and which new ones are formed – the order in which bond-breaking and bondforming steps take place – the role of the catalyst (if any is present) – the energy of the entire system during the reaction Energy Diagram • Energy diagram: a graph showing the changes in energy that occur during a chemical reaction • Reaction coordinate: a measure of the change in position of atoms during a reaction • Heat of reaction, H: the difference in energy between reactants and products – exothermic: products are lower in energy than reactants; heat is released – endothermic: products are higher in energy than reactants; heat is absorbed Energy Diagram • Transition state: an energy maximum – represents an unstable species formed during the course of a reaction • Activation Energy, Ea: the difference in energy between reactants and the transition state – determines the rate of reaction – if Ea is large, only a few molecular collisions occur with sufficient energy to reach the transition state, and the reaction is slow – if Ea is small, many collisions generate sufficient energy to reach the transition state, and the reaction is fast Activation Energy • Activation energy – Is the difference in energy between the reactants and the transition state. – Is the minimum energy required to a reaction to occur or happen – This will determine the rate of the reaction • How fast the reaction occurs Energy Diagram – an energy diagram for a two-step reaction involving formation of an intermediate • Nucleophile (nucleus loving): An electron rich species that seeks a region of low electron density. • Electrophile (electron loving): A low electrondensity species that seeks a region of high electron density. Addition of HX (X=halogens) Hydrohalogenation • Carried out with pure reagents or in a polar solvent such as acetic acid CH2 =CH2 + HCl Ethylene H Cl CH2 -CH2 Chloroeth ane • Addition is regioselective – regioselective reaction: a reaction in which one direction of bond-forming or bond-breaking occurs in preference to all other directions CH3 CH=CH2 + HCl Prop ene Cl H H Cl CH3 CH-CH2 + CH3 CH-CH2 2-Chloropropane 1-Ch loroprop ane (not ob served ) – Markovnikov’s rule: in additions of HX, H adds to the carbon with the greater number of hydrogens HCl + 2-Butene • A two-step mechanism – Step 1: formation of the sec-butyl cation, a 2° carbocation intermediate + CH3 CH-CHCH3 + : Cl : : CH3 CH=CHCH3 H : : : slow , rate + determinin g + H-Cl : sec-Butyl cation (a 2° carbocation intermediate) – Step 2: reaction of the sec-butyl cation (an electrophile) with chloride ion (a nucleophile) completes the reaction : + : : Cl : + : CH3 CHCH 2 CH 3 Ch loride ion sec-Butyl cation (a nucleop hile) (an electrop hile) fast :Cl : CH3 CHCH2 CH3 2-Chlorobutan e Carbocations • Carbocation: a species in which a carbon atom has only six electrons in its valence shell and bears positive charge • Carbocations are: – classified as 1°, 2°, or 3° depending on the number of carbons bonded to the carbon bearing the positive charge – electrophiles; that is, they are electron-loving – Lewis acids; that is, they are electron-pair acceptors Carbocations – a 3° carbocation is more stable than a 2° carbocation, and requires a lower activation energy for its formation – a 2° carbocation is, in turn, more stable than a 1° carbocation, and requires a lower activation energy for its formation – methyl and 1° carbocations are so unstable that they are never observed in solution H H C+ H Meth yl cation (methyl) CH3 H C+ CH3 CH3 C+ CH3 CH3 C+ H H CH3 Ethyl cation Is op ropyl cation tert -Butyl cation (1°) (2°) (3°) Increas ing carb ocation stability Addition of H2O • Addition of H2O is called hydration – acid-catalyzed hydration of an alkene is regioselective; hydrogen adds preferentially to the more substituted carbon of the double bond CH3 CH=CH2 + H2 O Prop ene CH3 CH3 C=CH2 + H2 O 2-Methylprop ene H2 SO4 H2 SO4 OH H CH3 CH-CH2 2-Prop anol CH3 CH3 C-CH2 HO H 2-Methyl-2-propanol Step 1: proton transfer to the alkene gives a carbocation + CH3 CH=CH2 + H O H slow , rate determining + CH3 CHCH3 A 2o carb ocation intermediate H + O H H Step 2: a Lewis acid/base reaction gives an oxonium ion + CH3 CHCH3 + O-H fast CH3 CHCH3 O+ H H H An oxonium ion Step 3: proton transfer to solvent gives the alcohol CH3 CHCH3 + H-O-H + O H H fas t + CH3 CHCH3 + H-O-H O H H Carbon Rearrangements + + HCl Cl expected product; 40% Cl rearrangement product; 60% • Rearrangement: change in connectivity of atoms in a product compared to starting material • 1,2-shift • Hydride ion is migrating group • More stable carbocation is formed HCl slow, rate determining + CH H H + fast CH + C H H + C Cl H - fast Cl + Cl - Addition of Cl2 and Br2 • Carried out with either the pure reagents or in an inert solvent such as CH2Cl2 Br Br CH3 CH=CHCH3 2-Butene + Br2 CH2 Cl2 CH3 CH-CHCH3 2,3-Dibromobutane Addition of Cl2 and Br2 – addition is stereoselective – stereoselective reaction: a reaction in which one stereoisomer is formed or destroyed in preference to all others that might be formed or destroyed – addition to a cycloalkene, for example, gives only a trans product Br + Br2 Cyclohexen e CH2 Cl2 Br t rans -1,2-D ib romocycloh exane Addition of Cl2 and Br2 – Step 1: formation of a bromonium ion intermediate Br Br C Br C C Br C C C + A bridged b romon ium ion intermediate – Step 2: halide ion opens the three-membered ring : : : Br : + : Br : :Br : : C C C C : : Br : A nti (cop lan ar) orien tation of ad ded b romin e atoms Br - Addition of Cl2 and Br2 • Anti coplanar addition to a cyclohexene corresponds to trans-diaxial addition Br + Br2 Br Br tran s diaxial (les s stable) Br tran s diequ atorial (more stable) Ozonolysis • Cleavage of a Carbon-Carbon double bond • Two Carbonyl groups in place 1. O3 2. (CH3)2S O O + H Reduction of Alkenes • Alkenes react with H2 in the presence of a transition metal catalyst to give alkanes – the most commonly used catalysts are Pd, Pt, and Ni – the reaction is called catalytic reduction or catalytic hydrogenation + H2 Cyclohexene Pd 25°C, 3 atm Cyclohexane Reduction of Alkenes – the most common pattern is syn addition of hydrogens; both hydrogens add to the same side of the double bond CH3 + H2 CH3 1,2-D imeth ylcycloh exene Pt CH3 CH3 cis-1,2-D imeth ylcyclohexane