PURDUE UNIVERSITY PHYS221 FINAL EXAM
... 15. If you wish to use a step down transformer on a trip to Belgium (since Belgians still drive on the proper side of the road unlike the English) to reduce the local voltage by a factor of 2 to suit your U.S. electrical gadgets, and the primary side of the transformer has 100 turns of wire, how man ...
... 15. If you wish to use a step down transformer on a trip to Belgium (since Belgians still drive on the proper side of the road unlike the English) to reduce the local voltage by a factor of 2 to suit your U.S. electrical gadgets, and the primary side of the transformer has 100 turns of wire, how man ...
6. Magnetism
... 1 mm in length and normally random in direction Each acts like tiny magnet Generally, domains cancel – no magnetic effects An external field aligns domains (non-random) A strong magnetic field can make other ferromagnetic materials into permanent magnets ...
... 1 mm in length and normally random in direction Each acts like tiny magnet Generally, domains cancel – no magnetic effects An external field aligns domains (non-random) A strong magnetic field can make other ferromagnetic materials into permanent magnets ...
The Movement of Charged Particles in a Magnetic Field
... In order to find the force of the magnetic field, we must first calculate its strenghth. Since F=qvB and, according to Newton’s second law, F=m•v²/r, we can deduce that ...
... In order to find the force of the magnetic field, we must first calculate its strenghth. Since F=qvB and, according to Newton’s second law, F=m•v²/r, we can deduce that ...
Department of Natural Sciences
... and into a magnetic field 0f 0.100 T. Each has a speed of 1.00 x 106 m/s. The nucleus of the first atom contains one proton and has a mass of 1.67 x 10-27 kg, and the nucleus of the second atom contains two neutrons and has a mass of 3.34 x 10-27 kg. Find their distance of separation when they strik ...
... and into a magnetic field 0f 0.100 T. Each has a speed of 1.00 x 106 m/s. The nucleus of the first atom contains one proton and has a mass of 1.67 x 10-27 kg, and the nucleus of the second atom contains two neutrons and has a mass of 3.34 x 10-27 kg. Find their distance of separation when they strik ...
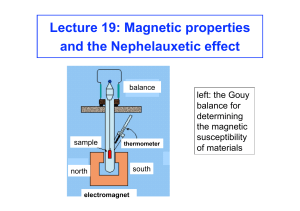
Lecture 19: Magnetic properties and the Nephelauxetic effect
... Spin and Orbital contributions to Magnetic susceptibility The value of λ is negligible for very light atoms, but increases with increasing atomic weight, so that for heavier d-block elements, and for f-block elements, the orbital contribution is considerable. For 2nd and 3rd row dblock elements, λ ...
... Spin and Orbital contributions to Magnetic susceptibility The value of λ is negligible for very light atoms, but increases with increasing atomic weight, so that for heavier d-block elements, and for f-block elements, the orbital contribution is considerable. For 2nd and 3rd row dblock elements, λ ...
Neutron magnetic moment

The neutron magnetic moment is the intrinsic magnetic dipole moment of the neutron, symbol μn. Protons and neutrons, both nucleons, comprise the nucleus of atoms, and both nucleons behave as small magnets whose strengths are measured by their magnetic moments. The neutron interacts with normal matter primarily through the nuclear force and through its magnetic moment. The neutron's magnetic moment is exploited to probe the atomic structure of materials using scattering methods and to manipulate the properties of neutron beams in particle accelerators. The neutron was determined to have a magnetic moment by indirect methods in the mid 1930s. Luis Alvarez and Felix Bloch made the first accurate, direct measurement of the neutron's magnetic moment in 1940. The existence of the neutron's magnetic moment indicates the neutron is not an elementary particle. For an elementary particle to have an intrinsic magnetic moment, it must have both spin and electric charge. The neutron has spin 1/2 ħ, but it has no net charge. The existence of the neutron's magnetic moment was puzzling and defied a correct explanation until the quark model for particles was developed in the 1960s. The neutron is composed of three quarks, and the magnetic moments of these elementary particles combine to give the neutron its magnetic moment.