Design of Thermoresponsive Polymers with
... respond only within a specific range of an environmental variable rather than just beyond a critical value. Reversible deactivation radical polymerization (RDRP) techniques have been applied to produce block copolymers comprising LCST- and UCST-type blocks in which the different parts of the macromole ...
... respond only within a specific range of an environmental variable rather than just beyond a critical value. Reversible deactivation radical polymerization (RDRP) techniques have been applied to produce block copolymers comprising LCST- and UCST-type blocks in which the different parts of the macromole ...
doc_296
... substances may be placed. Technologies for microencapsulating materials have been around for several years, primarily for applications involving minimisation of hygroscopy and chemical interactions, elimination of oxidation, and controlled release of nutraceuticals. The Use of Man-Made Liposomes Man ...
... substances may be placed. Technologies for microencapsulating materials have been around for several years, primarily for applications involving minimisation of hygroscopy and chemical interactions, elimination of oxidation, and controlled release of nutraceuticals. The Use of Man-Made Liposomes Man ...
Tuning the Molecular Properties of Polybenzimidazole by
... solubility of TPA in PPA being very low34 when the monomer concentration is high (4.83 wt % in Table 1), more and more TPA is precipitating out from the reaction mixture as insoluble solid destroying the stoichiometric balance. This in turn disfavors the reaction equilibrium and yields low IV polyme ...
... solubility of TPA in PPA being very low34 when the monomer concentration is high (4.83 wt % in Table 1), more and more TPA is precipitating out from the reaction mixture as insoluble solid destroying the stoichiometric balance. This in turn disfavors the reaction equilibrium and yields low IV polyme ...
THE ROLE OF FeSO4 IN THE OBTAINING OF
... the grafted polymerization. Unreacted PVP may be washed out during hydration that affects physicomechanical properties of copolymers. Therefore from the scientific and practical points of view it is interesting to determine the amount of reacted PVP. One can see from Table 2 that copolymer compositi ...
... the grafted polymerization. Unreacted PVP may be washed out during hydration that affects physicomechanical properties of copolymers. Therefore from the scientific and practical points of view it is interesting to determine the amount of reacted PVP. One can see from Table 2 that copolymer compositi ...
Physical Chemistry of Polymers: Entropy
... 1.C. Recent Developments. Synthesis. Conventional polymerization methods, either of the step-growth (e.g., polycondensation) or chain-growth (e.g., free radical) class, produce broad molecular weight distributions and offer little control over long-chain architectural characteristics such as branchi ...
... 1.C. Recent Developments. Synthesis. Conventional polymerization methods, either of the step-growth (e.g., polycondensation) or chain-growth (e.g., free radical) class, produce broad molecular weight distributions and offer little control over long-chain architectural characteristics such as branchi ...
Self-Assembly of Narrowly Distributed Carboxy
... swollen by the hydrophobic monomers presented in the dispersion so that a further reaction inside these micelle-like nanostructures could lead to stable polymeric latex particles. Therefore, it is important for us to understand how these short polystyrene chains are selfassembled in water in the ini ...
... swollen by the hydrophobic monomers presented in the dispersion so that a further reaction inside these micelle-like nanostructures could lead to stable polymeric latex particles. Therefore, it is important for us to understand how these short polystyrene chains are selfassembled in water in the ini ...
SYNTHESIS OF HIGH MOLECULAR WEIGHT POLY (METHYL
... Re-initiation: The radical produced from RAFT adduct propagates with the monomer and form another reactive polymer chain. Main-RAFT equilibrium: In an ideal situation, the radicals have the same opportunity to propagate with the species, which have not been terminated through a rapid exchange proces ...
... Re-initiation: The radical produced from RAFT adduct propagates with the monomer and form another reactive polymer chain. Main-RAFT equilibrium: In an ideal situation, the radicals have the same opportunity to propagate with the species, which have not been terminated through a rapid exchange proces ...
The role of chain length and conformation in stress
... fracture plane. In thermoplastic solids, where chains cohere through van der Waals forces, this involves several terms, which all depend on the stress transfer through and the breaking of secondary bonds. The relative contributions of the different mechanisms are well reflected by the fracture energ ...
... fracture plane. In thermoplastic solids, where chains cohere through van der Waals forces, this involves several terms, which all depend on the stress transfer through and the breaking of secondary bonds. The relative contributions of the different mechanisms are well reflected by the fracture energ ...
Nugget
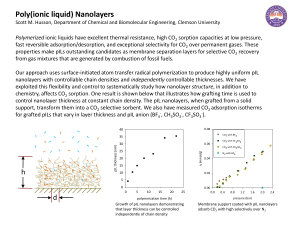
... Polymerized ionic liquids have excellent thermal resistance, high CO2 sorption capacities at low pressure, fast reversible adsorption/desorption, and exceptional selectivity for CO2 over permanent gases. These properties make pILs outstanding candidates as membrane separation layers for selective CO ...
... Polymerized ionic liquids have excellent thermal resistance, high CO2 sorption capacities at low pressure, fast reversible adsorption/desorption, and exceptional selectivity for CO2 over permanent gases. These properties make pILs outstanding candidates as membrane separation layers for selective CO ...
Classification of polymers
... Gibbs free energy of mixing is ΔG = ΔH - TΔS The important points to be considered in thermodynamics of polymer solution are ...
... Gibbs free energy of mixing is ΔG = ΔH - TΔS The important points to be considered in thermodynamics of polymer solution are ...
Polymers
... Addition or chain growth polymerisation is the linking of molecules having mul- tiple bonds. These unsaturated monomers have extra internal bonds which are able to break and link up with other monomers to form repeating chain. All the addition polymers are chain growth polymers, but few examples of ...
... Addition or chain growth polymerisation is the linking of molecules having mul- tiple bonds. These unsaturated monomers have extra internal bonds which are able to break and link up with other monomers to form repeating chain. All the addition polymers are chain growth polymers, but few examples of ...
EXPERIMENT 28 Intrinsic Viscosity: Chain Linkage in Polyvinyl
... For the determination of very high molecular weights, freezing-point depressions, boiling-point elevations, and vapor-pressure lowerings are too small for accurate measurement. Osmotic pressures are of a convenient order of magnitude, but measurements are time-consuming. The technique to be used in ...
... For the determination of very high molecular weights, freezing-point depressions, boiling-point elevations, and vapor-pressure lowerings are too small for accurate measurement. Osmotic pressures are of a convenient order of magnitude, but measurements are time-consuming. The technique to be used in ...
Surface Polymerisation (Polymer) 1 Plasma Enhanced Chemical
... higher concentration of polymerization building blocks than in the conventional processes and initiation and chain termination reactions dominated here over chain propagation. Moreover termination does not necessary interrupt the plasma polymerization, since the neutrals formed in the termination st ...
... higher concentration of polymerization building blocks than in the conventional processes and initiation and chain termination reactions dominated here over chain propagation. Moreover termination does not necessary interrupt the plasma polymerization, since the neutrals formed in the termination st ...
Controlled Fabrication of Cross-Linked Nanoparticles/Polymer
... this problem by suspending the silicon wafer in the diluted solution under continuous stirring. Furthermore, the slow polymerization rate in the solution kept the concentration of insoluble polymers low in the whole synthetic fabrication of homogeneous, cross-linked PLDMA films so that no polymers d ...
... this problem by suspending the silicon wafer in the diluted solution under continuous stirring. Furthermore, the slow polymerization rate in the solution kept the concentration of insoluble polymers low in the whole synthetic fabrication of homogeneous, cross-linked PLDMA films so that no polymers d ...
Combustion and thermal degradation of polymers
... Depending on catalysts the chain growth in anionic polymerization can go through two, three four or more directions not only in one direction as shown in the above example. In cationic polymerization as catalysts are used Lewis acids and Friedel-Crafts catalysts such as BF3, AlCl3, and SnCl4 and str ...
... Depending on catalysts the chain growth in anionic polymerization can go through two, three four or more directions not only in one direction as shown in the above example. In cationic polymerization as catalysts are used Lewis acids and Friedel-Crafts catalysts such as BF3, AlCl3, and SnCl4 and str ...
Chemistry 595: Physical Chemistry of Polymers
... topic, go to literature and learn about the current state of knowledge in that topic. Write a ~6-8 pages paper describing what you learned, formulating the major challenges and important problems in this topic. You will also present a ~15 minutes talk on the material during the last weeks of classes ...
... topic, go to literature and learn about the current state of knowledge in that topic. Write a ~6-8 pages paper describing what you learned, formulating the major challenges and important problems in this topic. You will also present a ~15 minutes talk on the material during the last weeks of classes ...
POLYMERS
... If a molecule has ν branches, it has ν + 2 chain ends. Two limiting cases are combs and stars. If the concentration of junction points is high enough, a point can be reached at which the polymer molecule becomes a giant three dimensional network, and it is said to be cross-linked. However, it is als ...
... If a molecule has ν branches, it has ν + 2 chain ends. Two limiting cases are combs and stars. If the concentration of junction points is high enough, a point can be reached at which the polymer molecule becomes a giant three dimensional network, and it is said to be cross-linked. However, it is als ...
Polymer Properties and Structure
... C2H4 is stable – does not spontaneously change into polyethylene – requires severe conditions ...
... C2H4 is stable – does not spontaneously change into polyethylene – requires severe conditions ...