Brand-Name vs Generic Finasteride Jan-Feb 2016
... is the central ingredient. The second is known as the excipient, which is the inactive substance that serves as the vehicle for the API itself. If the drug is in a syrup form, then the excipient is the liquid that has been used to make it as such.² APIs are commonly referred to as “bulk pharmaceutic ...
... is the central ingredient. The second is known as the excipient, which is the inactive substance that serves as the vehicle for the API itself. If the drug is in a syrup form, then the excipient is the liquid that has been used to make it as such.² APIs are commonly referred to as “bulk pharmaceutic ...
Clinical Actions of Specific Agents
... United States Pharmacopeial Convention (USP) reviewed the MRDs for local anesthetics and no longer adjusts them for inclusion of a ...
... United States Pharmacopeial Convention (USP) reviewed the MRDs for local anesthetics and no longer adjusts them for inclusion of a ...
Norepinephrine
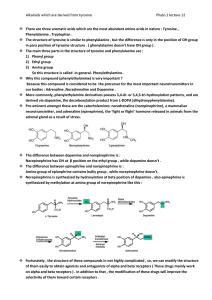
... bulkiness than agonist, so they said if we change the hydroxyls in catechol to something more hydrophobic then we make it antagonist. We extended the alkyl chain by one atom and we try to put many other atoms than carbon, the only atom that gives us the antagonist effect is O (ether linkage) . ...
... bulkiness than agonist, so they said if we change the hydroxyls in catechol to something more hydrophobic then we make it antagonist. We extended the alkyl chain by one atom and we try to put many other atoms than carbon, the only atom that gives us the antagonist effect is O (ether linkage) . ...
March 8, 2012 RADM William S Stokes Director, National Toxicology Program
... FDA agrees that the accuracy and reliability of the BG ILuc ER T A test method support its use as a screening test to identify substances with in vitro ER agonist or antagonist activity. However, FDA notes that with respect to use ofthe BGILuc ER TA test method and FDA-regulated products, FDA does n ...
... FDA agrees that the accuracy and reliability of the BG ILuc ER T A test method support its use as a screening test to identify substances with in vitro ER agonist or antagonist activity. However, FDA notes that with respect to use ofthe BGILuc ER TA test method and FDA-regulated products, FDA does n ...
A “Compare and Contrast” of International Guidelines - IPAC-RS
... Division 8, Part C of the Food and Drug Regulations. Abbreviated New Drug Submissions (ANDS) and Supplemental New Drug Submissions (SNDS) in support of these drug products are required to contain evidence of safety and effectiveness under the proposed conditions of use, in conformity with the applic ...
... Division 8, Part C of the Food and Drug Regulations. Abbreviated New Drug Submissions (ANDS) and Supplemental New Drug Submissions (SNDS) in support of these drug products are required to contain evidence of safety and effectiveness under the proposed conditions of use, in conformity with the applic ...
PDF - Dechert LLP
... Zenith have given rise to numerous class actions filed on behalf of putative classes of direct and indirect purchasers of terazosin hydrochloride. Noteworthy decisions in the Hytrin litigation include: Abbott Labs. v. Zenith Labs., Inc., 934 F. Supp. 925 (N.D. Ill. 1995), holding, inter alia, that t ...
... Zenith have given rise to numerous class actions filed on behalf of putative classes of direct and indirect purchasers of terazosin hydrochloride. Noteworthy decisions in the Hytrin litigation include: Abbott Labs. v. Zenith Labs., Inc., 934 F. Supp. 925 (N.D. Ill. 1995), holding, inter alia, that t ...
Oxcarbazepine (TrileptalTM)
... The Epilepsy Foundation is seriously concerned about mandatory substitution of generic antiepileptic drugs without prior approval of the patient and treating physician. Because changing from one formulation of an AED to another can usually be accomplished, and risks minimized, if physicians and pati ...
... The Epilepsy Foundation is seriously concerned about mandatory substitution of generic antiepileptic drugs without prior approval of the patient and treating physician. Because changing from one formulation of an AED to another can usually be accomplished, and risks minimized, if physicians and pati ...
510(k) - Indiana Medical Device Manufacturers Council
... that a device is exempt from premarket notification regulations only if there is already a similar device on the market with the same intended use and fundamental scientific technology • Limitations of exemptions from section 510(k) of the Federal Food, Drug, and Cosmetic Act – If not, manufacturer ...
... that a device is exempt from premarket notification regulations only if there is already a similar device on the market with the same intended use and fundamental scientific technology • Limitations of exemptions from section 510(k) of the Federal Food, Drug, and Cosmetic Act – If not, manufacturer ...
Slide 1
... SkyePharma’s business which could cause our actual results and developments to differ materially from those forwardlooking statements are discussed in SkyePharma’s SEC filings under the caption “Risk Factors”. SkyePharma reports under UK GAAP. Where US dollar equivalents have been provided for conve ...
... SkyePharma’s business which could cause our actual results and developments to differ materially from those forwardlooking statements are discussed in SkyePharma’s SEC filings under the caption “Risk Factors”. SkyePharma reports under UK GAAP. Where US dollar equivalents have been provided for conve ...
Bristol-Myers Squibb Pharmaceutical Research Institute
... subsequent meeting when the drug sponsor lacks ready answers to one of the group’s questions. In general, minutes of the SAT meetings are not sufficiently detailed, making it difficult at times to recreate how a particular decision was made. Thus, there are inconsistencies in decision making among d ...
... subsequent meeting when the drug sponsor lacks ready answers to one of the group’s questions. In general, minutes of the SAT meetings are not sufficiently detailed, making it difficult at times to recreate how a particular decision was made. Thus, there are inconsistencies in decision making among d ...
The Pharmaceutical Industry Presented to Geoffrey Poitras Bus 417
... Upon desirable results from Phase III, New Drug Application (NDA) will be submitted NDA contains data supporting the efficacy and safety of the drug Approval can take 2 month to several years, but on average, it takes around 18 to 24 months Drugs are subject to ongoing review, making sure no adverse ...
... Upon desirable results from Phase III, New Drug Application (NDA) will be submitted NDA contains data supporting the efficacy and safety of the drug Approval can take 2 month to several years, but on average, it takes around 18 to 24 months Drugs are subject to ongoing review, making sure no adverse ...
r-allergytoNSAID
... comparable to aspirin 650 mg for immediate pain relief, but tended to provide a longer duration of pain relief than aspirin. Cataflam 50 mg t.i.d. (three times a day) was comparable in efficacy to ibuprofen 800 mg t.i.d. The dose of Cataflam in children over 14 years of age is up to 75 mg daily in d ...
... comparable to aspirin 650 mg for immediate pain relief, but tended to provide a longer duration of pain relief than aspirin. Cataflam 50 mg t.i.d. (three times a day) was comparable in efficacy to ibuprofen 800 mg t.i.d. The dose of Cataflam in children over 14 years of age is up to 75 mg daily in d ...
Slide 1
... and the patients willing to participate, to advance therapies. Clinical trials often tell us unexpected things, both positive and negative, about new medical products, which is why carefully designed and conducted trials are an essential part of the pre-market process for demonstrating that new drug ...
... and the patients willing to participate, to advance therapies. Clinical trials often tell us unexpected things, both positive and negative, about new medical products, which is why carefully designed and conducted trials are an essential part of the pre-market process for demonstrating that new drug ...
Branded extended-release metformin, dru424
... The most common adverse events associated with metformin use are gastrointestinal, but can often be managed through dose titration, dose reduction, and/or use of an extended-release product. Glumetza is an alternative formulation of long-acting metformin that uses patented technology to delay the re ...
... The most common adverse events associated with metformin use are gastrointestinal, but can often be managed through dose titration, dose reduction, and/or use of an extended-release product. Glumetza is an alternative formulation of long-acting metformin that uses patented technology to delay the re ...
Counterfeit, Black-Market and Off-Label Drug Use
... vanish from a UM pharmacy? Technician is charged with stealing $14 million in expensive cancer drugs over three years before he was discovered. ...
... vanish from a UM pharmacy? Technician is charged with stealing $14 million in expensive cancer drugs over three years before he was discovered. ...
Weighing up the costs of switching AEDs
... Why generics? The development of any new drug is extremely costly as it can take a decade to be rigorously tested and evaluated before it is approved for manufacture and distribution. To help recoup this enormous investment, the pharmaceutical company that developed the drug has the sole rights, or ...
... Why generics? The development of any new drug is extremely costly as it can take a decade to be rigorously tested and evaluated before it is approved for manufacture and distribution. To help recoup this enormous investment, the pharmaceutical company that developed the drug has the sole rights, or ...
ΚΑΤΑΣΤΑΣΗ ΤΩΝ ΜΕΓΑΛΥΤΕΡΩΝ GENERIC ΦΑΡΜΑΚΕΥΤΙΚΩΝ
... expanded further in renal therapies when it acquired the hemofiltration (renal replacement therapy) product line of Edwards Lifesciences in a $65 million deal in 2009. The Renal division is also a leading supplier of heparin, an anticoagulant used during dialysis and in critical care situations such ...
... expanded further in renal therapies when it acquired the hemofiltration (renal replacement therapy) product line of Edwards Lifesciences in a $65 million deal in 2009. The Renal division is also a leading supplier of heparin, an anticoagulant used during dialysis and in critical care situations such ...
increased risk of local anesthetic toxicity
... • Adrenaline us a potent dilator of the smooth muscle of the bronchiole , so it the drug of choice for management of acute asthma . ...
... • Adrenaline us a potent dilator of the smooth muscle of the bronchiole , so it the drug of choice for management of acute asthma . ...
Sympathomimetics
... Consequently, total peripheral resistance may actually fall, explaining the fall in diastolic pressure that is sometimes seen with epinephrine injection. Activation of 2 receptors in skeletal muscle contributes to blood flow increase during exercise. Under physiologic conditions, epinephrine functio ...
... Consequently, total peripheral resistance may actually fall, explaining the fall in diastolic pressure that is sometimes seen with epinephrine injection. Activation of 2 receptors in skeletal muscle contributes to blood flow increase during exercise. Under physiologic conditions, epinephrine functio ...
Adrenergic Receptor Agonists
... • These are agents which directly active the alpha1 adrenergic receptor. They are less potent than the endogenous agonists epinephrine or norepinephrine. However, because of structural modifications they are orally active and have longer plasma half-lives. There are 2 structural classes of alpha1 ag ...
... • These are agents which directly active the alpha1 adrenergic receptor. They are less potent than the endogenous agonists epinephrine or norepinephrine. However, because of structural modifications they are orally active and have longer plasma half-lives. There are 2 structural classes of alpha1 ag ...
About the National Institute of Allergy and Infectious Diseases
... the anterolateral aspect of the thigh [see Dosage and Administration (2) and Patient Counseling Information (17)]. • Do not inject intravenously. Large doses or accidental intravenous injection of epinephrine may result in cerebral hemorrhage due to sharp rise in blood pressure. Rapidly acting vasod ...
... the anterolateral aspect of the thigh [see Dosage and Administration (2) and Patient Counseling Information (17)]. • Do not inject intravenously. Large doses or accidental intravenous injection of epinephrine may result in cerebral hemorrhage due to sharp rise in blood pressure. Rapidly acting vasod ...
AACE, TES, ATA Joint Position Statement on the Use and
... Note: Drug I may be AB to Drug II and Drug II AB to Drug III. However, this does not mean that Drug I and III are AB to each other. This is because Drug II and Drug III may not have been compared with one another, rather than proven not to be equivalent. This point is illustrated by the following ex ...
... Note: Drug I may be AB to Drug II and Drug II AB to Drug III. However, this does not mean that Drug I and III are AB to each other. This is because Drug II and Drug III may not have been compared with one another, rather than proven not to be equivalent. This point is illustrated by the following ex ...
Anaphylaxis - SDACEP Conference
... Be sure to have two doses available, as the severe reaction can recur in about 20% of individuals There are no data to help predict who may need a second dose of epinephrine, so this recommendation applies to all patients with a food allergy If you are uncertain whether a reaction warrants epi ...
... Be sure to have two doses available, as the severe reaction can recur in about 20% of individuals There are no data to help predict who may need a second dose of epinephrine, so this recommendation applies to all patients with a food allergy If you are uncertain whether a reaction warrants epi ...
The Food and Drug Administration (FDA) - Overview
... contaminants. FDA also ensures the purity and effectiveness of biologicals (medical preparations made from living organisms and their products), such as insulin and vaccines. ...
... contaminants. FDA also ensures the purity and effectiveness of biologicals (medical preparations made from living organisms and their products), such as insulin and vaccines. ...
7100 Software Test System
... equipment Inspection and test status Control of non-conforming product Corrective and preventive action Handling, storage, packaging, preservation, and delivery Control of quality records Internal quality audits Training Servicing Statistical techniques ...
... equipment Inspection and test status Control of non-conforming product Corrective and preventive action Handling, storage, packaging, preservation, and delivery Control of quality records Internal quality audits Training Servicing Statistical techniques ...
Epinephrine autoinjector

An epinephrine autoinjector, is a medical device for injecting a measured dose or doses of epinephrine (adrenaline), by means of autoinjector technology; it is most often used for the treatment of anaphylaxis. The EpiPen is derived from the Mark I NAAK ComboPen, which was developed for the U.S. military for treating exposure to nerve agents in the course of chemical warfare. Trademark names for the autoinjector device include EpiPen, Emerade, Twinject, Adrenaclick, Anapen, Jext, Allerject, and Auvi-Q.