Chemical Equations
... e) remember the rules for writing formulas for molecular compounds (______________) • Only NONMETALS! f) remember the formula for water, ________ • HOH = hydrogen hydroxide 3. Write a balanced chemical equation by adding_____________________, NOT subscripts (this will require trial and error, the f ...
... e) remember the rules for writing formulas for molecular compounds (______________) • Only NONMETALS! f) remember the formula for water, ________ • HOH = hydrogen hydroxide 3. Write a balanced chemical equation by adding_____________________, NOT subscripts (this will require trial and error, the f ...
5-17_MICROBES_AND_ INDUSTRY
... year that results in more pollutional load on to the environment. But as the research focuses to reduce this environmental pollution load by limiting the chemical use, new options emerged from degrading capabilities of microbes & their enzymes and then various chemical process were replaced by biote ...
... year that results in more pollutional load on to the environment. But as the research focuses to reduce this environmental pollution load by limiting the chemical use, new options emerged from degrading capabilities of microbes & their enzymes and then various chemical process were replaced by biote ...
5-17_MICROBES_AND_ INDUSTRY
... year that results in more pollutional load on to the environment. But as the research focuses to reduce this environmental pollution load by limiting the chemical use, new options emerged from degrading capabilities of microbes & their enzymes and then various chemical process were replaced by biote ...
... year that results in more pollutional load on to the environment. But as the research focuses to reduce this environmental pollution load by limiting the chemical use, new options emerged from degrading capabilities of microbes & their enzymes and then various chemical process were replaced by biote ...
Synthesis Reaction
... I can write chemical reactions by interpreting word equations I can classify reaction types (synthesis, decomposition, single replacement, double replacement, combustion) I can predict the products of chemical reactions in writing complete chemical equations (synthesis, decomposition, single replace ...
... I can write chemical reactions by interpreting word equations I can classify reaction types (synthesis, decomposition, single replacement, double replacement, combustion) I can predict the products of chemical reactions in writing complete chemical equations (synthesis, decomposition, single replace ...
Document
... I can write chemical reactions by interpreting word equations I can classify reaction types (synthesis, decomposition, single replacement, double replacement, combustion) I can predict the products of chemical reactions in writing complete chemical equations (synthesis, decomposition, single replace ...
... I can write chemical reactions by interpreting word equations I can classify reaction types (synthesis, decomposition, single replacement, double replacement, combustion) I can predict the products of chemical reactions in writing complete chemical equations (synthesis, decomposition, single replace ...
Diversity-oriented synthesis - David Spring
... analysis of various databases. It was found that the number of chiral centers, on average, from combinatorial chemistry, natural products, and drugs was 0.4, 6.2, and 3.3 per molecule, respectively.17 Although the compound archives of pharmaceutical companies (which will include many combinatorial l ...
... analysis of various databases. It was found that the number of chiral centers, on average, from combinatorial chemistry, natural products, and drugs was 0.4, 6.2, and 3.3 per molecule, respectively.17 Although the compound archives of pharmaceutical companies (which will include many combinatorial l ...
Balancing a Chemical Equation
... 4. Balance the elements one at a time by using coefficients. When no coefficient is written, it is assumed 2. Write the skeleton equation by to be 1. Begin by balancing placing the formulas for the elements that appear only once on reactants on the left and the each side of the equation. Never formu ...
... 4. Balance the elements one at a time by using coefficients. When no coefficient is written, it is assumed 2. Write the skeleton equation by to be 1. Begin by balancing placing the formulas for the elements that appear only once on reactants on the left and the each side of the equation. Never formu ...
Siegfried`s One Stop Shop for partners in the pharmaceutical industry
... For many years Siegfried is a well known ...
... For many years Siegfried is a well known ...
Section 1 The Nature of Chemical Reactions
... • The law of definite proportions states that a compound always contains the same elements in the same proportions, regardless of how the compound is made or how much of the compound is formed. • Because the law of definite proportions holds true for all chemical substances in all reactions, mole ra ...
... • The law of definite proportions states that a compound always contains the same elements in the same proportions, regardless of how the compound is made or how much of the compound is formed. • Because the law of definite proportions holds true for all chemical substances in all reactions, mole ra ...
H 2
... In this section you will analyze common chemical reactions. The goal is to give you the background information necessary to determine the type of product when given the reactants. There are seven of these reactions. You have already had experience with some of them. The following slides will ...
... In this section you will analyze common chemical reactions. The goal is to give you the background information necessary to determine the type of product when given the reactants. There are seven of these reactions. You have already had experience with some of them. The following slides will ...
File - Fidaa`s Level 2 Portfolio
... carboxylic acids, ketones, ethers, amines, and benzenes. They all have different properties that make them unique and are very important especially for this specific lab. Esters, the main topic for this lab, are part of the artificial flavors we will be making. Ester synthesis is when a chemical rea ...
... carboxylic acids, ketones, ethers, amines, and benzenes. They all have different properties that make them unique and are very important especially for this specific lab. Esters, the main topic for this lab, are part of the artificial flavors we will be making. Ester synthesis is when a chemical rea ...
Chapter 11 * Chemical Reactions
... Chemical Reactions • Chemical equations • Balancing Chemical Equations • According to the Law of Conservation of Mass, the number of atoms on the reactant side of the equation must equal the number of atoms on the product side. • We can adjust the number of atoms on each side of the equation by pla ...
... Chemical Reactions • Chemical equations • Balancing Chemical Equations • According to the Law of Conservation of Mass, the number of atoms on the reactant side of the equation must equal the number of atoms on the product side. • We can adjust the number of atoms on each side of the equation by pla ...
How do we predict chemical change?
... bottles will react with the liquids it contains to produce toxic substances. We may be interested in predicting whether the combustion products of a new type of gasoline will react with compounds in the atmosphere. We may want to identify the types of chemical reactions that led to the formation of ...
... bottles will react with the liquids it contains to produce toxic substances. We may be interested in predicting whether the combustion products of a new type of gasoline will react with compounds in the atmosphere. We may want to identify the types of chemical reactions that led to the formation of ...
Chemistry Merit Badge
... 5) List the four classical divisions of chemistry. Briefly describe each one, and tell how it applies to your everyday life. 6) Do EACH of the following activities: A) Name two government agencies that are responsible for tracking the use of chemicals for commercial or industrial use. Pick one agenc ...
... 5) List the four classical divisions of chemistry. Briefly describe each one, and tell how it applies to your everyday life. 6) Do EACH of the following activities: A) Name two government agencies that are responsible for tracking the use of chemicals for commercial or industrial use. Pick one agenc ...
press release
... programs, AIChE continues to be a focal point for information exchange on the frontiers of chemical engineering research in such areas as energy, sustainability, biological and environmental engineering, nanotechnology, and chemical plant safety and security. More information about AIChE is availabl ...
... programs, AIChE continues to be a focal point for information exchange on the frontiers of chemical engineering research in such areas as energy, sustainability, biological and environmental engineering, nanotechnology, and chemical plant safety and security. More information about AIChE is availabl ...
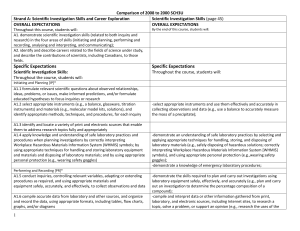
Comparison of 2008 to 2000 SCH3U_ud
... compounds can be released into the environment, including cadmium, arsenic, sulphur dioxide, and mercury, all of which can endanger the health and safety of local populations. Sample questions: What are some chemical reactions used in the manufacture of paper? How might the reactants or products of ...
... compounds can be released into the environment, including cadmium, arsenic, sulphur dioxide, and mercury, all of which can endanger the health and safety of local populations. Sample questions: What are some chemical reactions used in the manufacture of paper? How might the reactants or products of ...
Comparing Free Energies
... absorption (DHrxn > 0) should limit the number of states in which these particles may exist. Energy exchange at high T has a smaller impact on the entropy of the surroundings as its particles already have access to many different states. On the other hand, an energy transfer at low T should affect t ...
... absorption (DHrxn > 0) should limit the number of states in which these particles may exist. Energy exchange at high T has a smaller impact on the entropy of the surroundings as its particles already have access to many different states. On the other hand, an energy transfer at low T should affect t ...
CHEM MINI-COURSE SERIES M1.2___
... each type of atoms must appear on both sides of an equation. The atoms merely rearrange or regroup into different elements or compounds; they will not change into other atoms or be lost through a chemical reaction. For example, hydrogen gas and oxygen gas react to produce water. The following equati ...
... each type of atoms must appear on both sides of an equation. The atoms merely rearrange or regroup into different elements or compounds; they will not change into other atoms or be lost through a chemical reaction. For example, hydrogen gas and oxygen gas react to produce water. The following equati ...
Lecture on Air Pollution Prevention and Control
... established by the EPA in quantities greater than the threshold level, will be required to comply with rules for the "use, operation, repair, replacement, and maintenance of equipment to monitor, detect, inspect, and control releases." Such regulations must also require owners and operators to "prep ...
... established by the EPA in quantities greater than the threshold level, will be required to comply with rules for the "use, operation, repair, replacement, and maintenance of equipment to monitor, detect, inspect, and control releases." Such regulations must also require owners and operators to "prep ...
Chemical Properties - Michigan State University
... substances, which have different properties, compared to the old substances. The composition is a chemical change is altered. This occurs when you burn a substance, mixing an acid and a base, or when you observe rusting or rotting. The process that produces a chemical change is known as a chemical ...
... substances, which have different properties, compared to the old substances. The composition is a chemical change is altered. This occurs when you burn a substance, mixing an acid and a base, or when you observe rusting or rotting. The process that produces a chemical change is known as a chemical ...
___Mg + ___O ___MgO • Mole : Mole ratio
... 2) What is the percent yield when 2.37 grams of silver nitrate reacts with sodium hydroxide to produce water, sodium nitrate and 1.55 grams of silver oxide? ...
... 2) What is the percent yield when 2.37 grams of silver nitrate reacts with sodium hydroxide to produce water, sodium nitrate and 1.55 grams of silver oxide? ...
Multi-Criteria Decision Making in Product
... chemical engineers to assist the development of chemical engineering. In tying science to engineering, PSE provides engineers with the systematic design and operation methods, tools that they require to successfully face the challenges of today's industry (Grossmann and Westerberg, 2000). One such m ...
... chemical engineers to assist the development of chemical engineering. In tying science to engineering, PSE provides engineers with the systematic design and operation methods, tools that they require to successfully face the challenges of today's industry (Grossmann and Westerberg, 2000). One such m ...
Lecture 14
... 1. Write the correct symbols and formulas for all of the reactants and products. 2. Count the number of each type of atom on BOTH sides of the equation. 3. Insert coefficients until there are the equal numbers of each kind of atom on both sides of the equation. ...
... 1. Write the correct symbols and formulas for all of the reactants and products. 2. Count the number of each type of atom on BOTH sides of the equation. 3. Insert coefficients until there are the equal numbers of each kind of atom on both sides of the equation. ...
Chemical Glossary - Wacker Chemie AG
... K12 bacterial strain which can flush proteins into the culture medium via the outer cell membrane. The result is an efficient, inexpensive way of manufacturing pharmaceutical proteins. The ESETEC® E. coli secretion technology lends itself to antibody fragment production. The use of ESETEC® E. coli s ...
... K12 bacterial strain which can flush proteins into the culture medium via the outer cell membrane. The result is an efficient, inexpensive way of manufacturing pharmaceutical proteins. The ESETEC® E. coli secretion technology lends itself to antibody fragment production. The use of ESETEC® E. coli s ...
Fine chemical

Fine chemicals are complex, single, pure chemical substances, produced in limited quantities in multipurpose plants by multistep batch chemical or biotechnological processes. They are described by exacting specifications, used for further processing within the chemical industry and sold for more than $10/kg (see the comparison of fine chemicals, commodities and specialties). The class of fine chemicals is subdivided either on the basis of the added value (building blocks, advanced intermediates or active ingredients), or the type of business transaction, namely standard or exclusive products.Fine chemicals are produced in limited volumes (< 1000 tons/year) and at relatively high prices (> $10/kg) according to exacting specifications, mainly by traditional organic synthesis in multipurpose chemical plants. Biotechnical processes are gaining ground. The global production value is about $85 billion. Fine chemicals are used as starting materials for specialty chemicals, particularly pharmaceuticals, biopharmaceuticals and agrochemicals. Custom manufacturing for the life science industry plays a big role; however, a significant portion of the fine chemicals total production volume is manufactured in house by large users. The industry is fragmented and extends from small, privately owned companies to divisions of big, diversified chemical enterprises. The term ""fine chemicals"" is used in distinction to ""heavy chemicals"", which are produced and handled in large lots and are often in a crude state.Since their inception in the late 1970s, fine chemicals have become an important part of the chemical industry. The total production value of $85 billion is split about 60 / 40 among in-house production by the main consumers, the life science industry, on the one hand, and the fine chemicals industry on the other hand. The latter pursues both a “supply push” strategy, whereby standard products are developed in-house and offered ubiquitously, and a “demand pull” strategy, whereby products or services determined by the customer are provided exclusively on a “one customer / one supplier” basis. The products are mainly used as building blocks for proprietary products. The hardware of the top tier fine chemical companies has become almost identical. The design, lay-out and equipment of the plants and laboratories has become practically the same all over the world. Most chemical reactions performed go back to the days of the dyestuff industry. Numerous regulations determine the way labs and plants have to be operated, thereby contributing to the uniformity.